Summary
Objective
Cholangiocarcinoma is common in Thailand. There are many palliative treatments available for patients with unresectable tumor, such as endoscopic retrograde cholangiopancreatography (ERCP) with stents, percutaneous transhepatic biliary drainage, or surgery. In cases in which ERCP has failed, we propose an alternative technique: the use of endoscopic ultrasound with fluoroscopy to perform hepaticogastrostomy for palliative drainage instead of percutaneous transhepatic biliary drainage.
Patients and methods
A case series study was conducted between December 2005 and December 2009 of 10 patients (4 male and 6 female, average age: 57 years) who presented with severe jaundice caused by advanced cholangiocarcinoma, who were treated with this procedure after failure to drain by ERCP. We used an electronic convex curved linear-array fluoroscopy-guided echoendoscope to drain the left dilated intrahepatic duct to the stomach by metallic wallstent. We performed the procedure with the first six patients under general anesthesia and with the other four under conscious sedation. Follow-up liver function tests were done, and clinical symptoms and survival times were recorded.
Results
Hepaticogastrostomy was unsuccessful on the first two patients (success rate = 8/10; 80%), and effective drainage was obtained in only seven patients. Average total bilirubin reduction was 14.96 mg/dL (58.75%) and 18.13 mg/dL (71.20%) after 2 weeks and 4 weeks, respectively, with good quality of life. One patient was not effectively drained because of malposition of the stent. There were two patients whose stent migrated into the stomach; one needed a second session with a second wallstent, and the other needed a double pigtail stent inside the second wallstent. Follow-up survival rates were 32–194 days (average: 123 days).
Conclusion
Endoscopic-ultrasound-guided hepaticogastrostomy is safe and can be a good palliative option for advanced malignant biliary obstruction because it drains internally and is remote from the tumor site, promoting a long patency period of prosthesis and better quality of life.
Keywords
advanced cholangiocarcinoma;endoscopic ultrasound;hepaticogastrostomy
1. Introduction
Cholangiocarcinoma is a common malignant tumor in the Thai population, especially in the northeastern part of Thailand; an area in which liver fluke is endemic. The curative treatment is surgery, but most patients are inoperable by the time they consult a specialist. The choice of treatment for patients with biliary obstruction caused by inoperable cancer is controversial. Usually, the first choice is endoscopic retrograde cholangiopancreatography (ERCP) with plastic or metallic stents. Another palliative treatment is percutaneous transhepatic biliary drainage (PTBD), including external drainage or internal drainage by radiologically placing a stent through the tumor into the common bile duct; however, ∼28% of these patients have recurrent obstruction and cholangitis.1 Palliative hepaticoenteric surgical bypass may offer longer-term patency and better quality of life compared with PTBD, but the patients will suffer from postoperative pain, complications and higher mortality. This proposed technique instead of PTBD, is also a palliative procedure that drains the biliary duct far from the site of tumor and avoids open surgery by using an endoscopic ultrasound (EUS)-guided method to perform hepaticogastrostomy as a minimally invasive option.2; 3; 4; 5; 6; 7 ; 8
2. Patients and methods
2.1. Patients
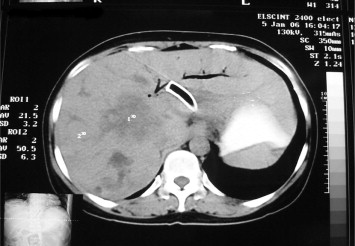
A case series study of minimally invasive trial EUS-guided hepaticogastrostomy was conducted from December 2005 to December 2009 on 10 patients with indications including obstructive jaundice from inoperable (advanced) hilar cholangiocarcinoma with failed ERCP and significant dilation of the left intrahepatic bile duct (Fig. 1). The exclusion criteria were shock, coagulopathy, ascites, and non-cooperation. Four of the patients were male and the other six were female, and their average age was 57 years. There were no pathologically proven cases of cholangiocarcinoma. All patients were diagnosed by clinical findings and imaging. We performed EUS-guided hepaticogastrostomy using the technique described below (Figs. 2 and 3); the first six patients received the procedure under general anesthesia and the other four under conscious sedation. The patients who drained successfully had follow-up progression of disease and blood level of liver function and alkaline phosphatase were followed 2 and 4 weeks after procedure and data of survival times were recorded.
|
|
|
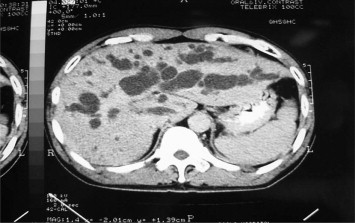
Figure 1. Computed tomography scan of the liver showed marked dilated bilateral intrahepatic bile duct in type 4 hilar cholangiocarcinoma. |
|
|
|
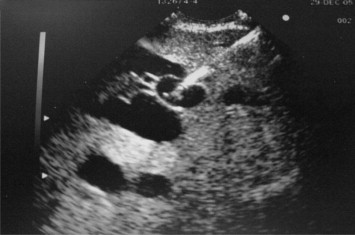
Figure 2. Endoscopic ultrasound view of needle punctured through the gastric wall into the dilated left intrahepatic bile duct. |
|
|
|
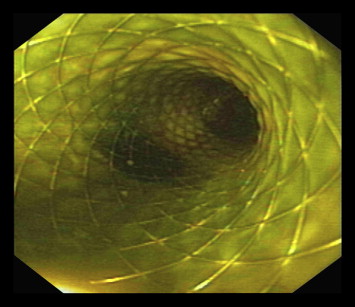
Figure 3. An 8 Fr 60-mm metallic wallstent in place of hepaticogastrostomy with bile flow. |
2.2. Technique of EUS-guided hepaticogastrostomy
The EUS-guided hepaticogastrostomy technique introduced by M. Giovannini consists of a two-step procedure which creates an anastomosis between the dilated left intrahepatic bile duct and the stomach.2 He placed a plastic stent in first step and then replaced it by a covered metallic stent a few weeks later. In this study, we used the Pentax EG 38 UT with EUB 6500 Hitachi Ultrasound with convex curved linear array echo-endoscope and C-arm fluoroscopy. This technique was applied from technique of EUS-FNA (fine needle aspiration) by using needle via the EUS endoscope to create tract from lesser curve of stomach to left intrahepatic duct under general anesthesia or conscious sedation (25 mg pethidine + 2.5 mg midazolam), the EUS scope was introduced through the mouth into the stomach. Through the lesser curvature of the stomach, the dilated left intrahepatic duct in segment II was identified and punctured with a 19-gauge EUS needle (Wilson–Cook, Winston-Salem, NC, USA). Under fluoroscopic control, a contrast agent was injected to show the intrahepatic bile duct. A guide wire (0.035 inch diameter) was inserted into bile duct and a cystotome was applied via the scope to create a tunnel at the lesser curvature of the stomach wall, then a needle knife was applied via the scope to puncture the liver into the bile duct by using cauterization. We preferred a single-step procedure, so instead of placing a plastic stent to create a fistula, after removing the needle knife, we placed a 10-mm diameter, 60-mm length covered, self-expanding metallic stent or wallstent (Boston Scientific Corporation, Boston, MA, USA) in one step and then removed the guide wire and EUS scope.
3. Results
EUS-guided hepaticogastrostomy was successful in eight of the 10 patients without any immediate complication such as massive pneumoperitoneum. Placement of stents was unsuccessful in the first two patients (success rate = 8/10; 80%). Effective drainage was obtained in seven patients while the stent position and drainage in the third patient was not so effective, because the penetration site of the stomach was at the esophagogastric junction. Follow-up observations after 2 weeks and 4 weeks found that average total bilirubin fell from 25.46 mg/dL to 10.50 mg/dL and 7.33 mg/dL (or reduction rate = 58.75% and 71.20%), respectively (Table 1). All successful patients had a good recovery, and started an oral diet on the day after the procedure. Follow-up computed tomography (CT) scans after 2 weeks showed metallic stents, with marked reduction of dilated intrahepatic bile duct (Fig. 4). There were two patients whose stents were dislodged and migrated into the stomach, detected by plain abdominal X-ray on the day after the procedure; one needed a second session with a second wallstent and the other required a second wallstent and a double pigtail stent placed inside the wallstent to prevent migration due to the tunnel being too large. Most of the followed up patients had much improved jaundice and a good quality of life, and the survival times were 32–194 days (average: 123 days) (Table 2). The causes of death were mostly advanced stage of the disease with liver or multiorgan failure.
| Patient | Sex | Age (y) | Hb (g/dL) | WBC/μL | CEA (ng/mL) | Bismuth type | Pre-intervention | 2 wk post-intervention | 4 wk post-intervention | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bilirubin (TB/DB) (mg/dL) | AP (μL) | Bilirubin (TB/DB) (mg/dL) | AP (μL) | Bilirubin (TB/DB) (mg/dL) | AP (μL) | |||||||
| 1 | M | 51 | 9.3 | 7500 | 3.15 | 3A | 25.44/20.14 | 518 | 27.29/20.29 | 1269 | 27.46/18.78 | 1062 |
| 2 | F | 59 | 10.6 | 6800 | 6.22 | 4 | 20.50/17.48 | 488 | 3.42/2.16 | 358 | 1.25/0.64 | 615 |
| 3 | F | 44 | 10.2 | 10,900 | 5.38 | 4 | 38.37/29.45 | 494 | 10.42/7.87 | 501 | 5.02/3.69 | 1158 |
| 4 | M | 48 | 9.9 | 9300 | 7.90 | 4 | 22.00/16.09 | 521 | 12.77/8.80 | 489 | 5.20/2.50 | 720 |
| 5 | F | 62 | 11.5 | 11,000 | 3.06 | 3A | 22.62/16.78 | 655 | 4.22/3.19 | 218 | 0.72/0.38 | 60 |
| 6 | F | 56 | 10.8 | 5700 | 12.18 | 4 | 17.60/9.10 | 823 | 18.88/13.32 | 200 | 14.55/7.10 | 253 |
| 7 | M | 67 | 12.6 | 6300 | 9.05 | 4 | 26.31/18.99 | 1427 | 10.07/5.92 | 716 | 10.62/7.37 | 1863 |
| 8 | F | 60 | 10.3 | 5500 | 1.96 | 4 | 21.83/16.54 | 484 | 18.00/ 14.50 | 255 | 8.55/4.92 | 220 |
| 9 | M | 58 | 11.7 | 8800 | 3.44 | 3A | 66.27/40.46 | 2726 | 10.50/7.60 | 7.33/4.53 | ||
| 10 | F | 65 | 9.2 | 17,200 | 5.38 | 4 | 39.64/27.49 | 443 | 14.96 (58.75%) | 18.13 (71.2%) | ||
| Average | 57 | 10.6 | 7800 | 5.77 | 25.46/17.49 | |||||||
| Reduction of total bilirubin | ||||||||||||
AP = alkaline phosphatase; CEA = carcinoembryonic antigen; DB = direct bilirubin; F = female; Hb = hemoglobin; M = male; TB = total bilirubin; WBC = white blood cell count.
|
|
|
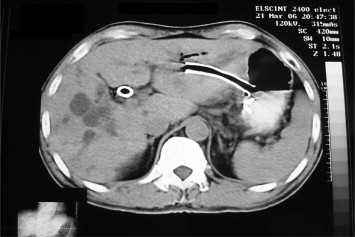
Figure 4. Follow-up computed tomography scan showed metallic stent drained at lesser curvature of the stomach and nearly normal size of the bile duct. |
| Patient | Result of procedure | Complications | Survival time | Cause of death |
|---|---|---|---|---|
| 1 | Failure | – | – | – |
| 2 | Failure | – | – | – |
| 3 | Success | Cholangitis | 1 m 2 d | Sepsis |
| 4 | Success | – | 6 m 10 d | Sepsis |
| 5 | Success | Stent migration | 7 m 24 d | Hepatic failure |
| 6 | Success | – | 3 m 4 d | Hepatic failure |
| 7 | Success | – | 4 m 10 d | Hepatic failure |
| 8 | Success | – | 1 m 11 d | Hepatic failure |
| 9 | Success | Stent migration | 5 m 2 d | Sepsis |
| 10 | Success | – | 5 m 1 d | Hepatic failure |
| Average | 123 d (4 mo) | |||
4. Discussion
Cholangiocarcinoma is a common malignant tumor of the bile duct in the Thai population and has the highest recorded incidence in the world in Northeast Thailand. For resectable cases, the treatment of choice is surgical resection but most patients present when the disease is at an advanced stage or inoperable. There are several options for treatment, with ERCP and endoprosthesis being the first choice. If ERCP fails or is not possible, PTBD or endoprosthesis trans-PTBD are other options but PTBD has a high risk of infection, obstruction, and subsequent poor quality of life because the stent is placed across the tumor and the catheter drains into a bag outside the body. Becker et al have demonstrated a higher rate of complications in patients with hilar involvement compared with those without hilar involvement, with significantly lower stent patency rates.1
There are many techniques for draining the biliary duct far from the tumor site, as described by Soulez et al using fluoroscopic, endoscopic and laparoscopic guidance to create a hepaticogastric anastomosis,9 and Tipaldi using CT guidance and fluoroscopy with two steps of percutaneous hepatogastric drainage.10 Warren indicated that diversion of bile from the liver into the stomach does not cause gastric ulcers, which is another advantage of hepaticogastrostomy,11 but Moss has indicated that gastritis could be a potential complication due to the contact of gastric mucosa with bile.12
Endoscopic ultrasound, which was developed >20 years ago, is currently a valuable diagnostic instrument and also a therapeutic intervention. Since the introduction of the curved linear array echoendoscope in 1990, many procedures such as cystogastrostomy for pancreatic pseudocyst, choledochoduodenostomy and hepaticogastrostomy can be performed by minimally invasive techniques.2; 3; 4; 5; 6; 7 ; 8 EUS-guided hepaticogastrostomy has the same level of safety as compared with PTBD, but higher effectiveness of drainage because of the larger diameter of the metallic stent than the PTBD catheter (10 mm vs. 3 mm).
With our first two patients, in the early stages when we had no previous experience, we failed to place the stent because of technical error and we consulted with a radiologist to perform PTBD in both cases. With the third patient, we succeeded in placing the stent, but the position of the stent was not favorable because it drained from the left intrahepaticduct through the esophagogastric junction, so that it was at the same level as the bile duct and the bile did not drain well by the natural effect of gravity. However, the CT scan showed much improvement in the dilated bile duct after 2 weeks (Fig. 5). With the other patients, with the benefit of more experience, we placed the stents more effectively, and bilirubin levels fell rapidly. At the 2-week follow-up observation, there was significant improvement in the patients' bile ducts, and they had a good appetite and a better quality of life.
|
|
|
Figure 5. Follow-up computed tomography scan showed metallic stent drained at the esophagogastric junction and much smaller size of the bile duct in the third patient. |
The complications and prognostic factors for mortality in these patients were advanced malignancy, impaired liver function, off and on cholangitis, bile leakage to the peritoneal cavity, stent obstruction or malposition, and poor drainage of bile. Two patients (Cases 3 and 8) that were not well drained had survival time of only ∼1 month because they quickly developed sepsis and hepatic failure. In other patients, with rapid reduction of bilirubin, appropriate positioning of the stent, and very good drainage of bile, longer survival times were achieved. There were no definite episodes of bile leakage and stent obstruction.
In conclusion, EUS-guided hepaticogastrostomy can be done safely, with short hospitalization, and a longer period of stent patency. It makes the patients more comfortable and results in a better quality of life. This technique should be an alternative to PTBD or surgical drainage for patients with advanced cholangiocarcinoma when palliative ERCP fails or is not possible.
Acknowledgments
Assoc. Prof. Sukij Panpimanmas, accepts full responsibility for the conduct of this study, collection and/or interpretation of data, and/or drafting the manuscript. Dr Thawee Ratanachu-ek has approved the final draft submitted.
References
- 1 C.D. Becker, A. Glatti, R. Maibach, H.U. Baer; Percutaneous palliation of malignant obstructive jaundice with the wallstent endoprosthesis: follow-up and reintervention in patients with hilar and non hilar obstruction; J Vasc Intervent Radiol, 4 (1993), pp. 597–604
- 2 M. Giovannini, M. Dotti, E. Bories, et al.; Hepaticogastrostomy by echo-endoscopy as a palliative treatment in a patient with metastatic biliary obstruction; Endoscopy, 35 (2003), pp. 1076–1078
- 3 M. Raj, R.Y. Chen; Interventional application of endoscopic ultrasound; J Gastroenterol Hepatol, 21 (2006), pp. 348–357
- 4 E. Bories, C. Pesenti, F. Caillol, C. Lopes, M. Giovannini; Transgastric endoscopic ultrasonography-guided biliary drainage: results of a pilot study; Endoscopy, 39 (2007), pp. 287–291
- 5 U. Will, A. Thieme, F. Fueldner, R. Geriach, I. Wanzar, F. Meyer; Treatment of biliary obstruction in selected patients by endoscopic ultrasonography (EUS)-guided transluminal biliary drainage; Endoscopy, 39 (2007), pp. 292–295
- 6 H. Park do, J.E. Koo, J. Oh, et al.; EUS-guided biliary drainage with one-step placement of a fully covered metal stent for malignant biliary obstruction: a prospective feasibility study; Am J Gastroenterol, 104 (2009), pp. 2168–2174
- 7 H. Park do, T.J. Song, J. Eum, et al.; EUS-guided hepaticogastrostomy with a fully covered metal stent as the biliary diversion technique for an occluded biliary metal stent after a failed ERCP (with videos); Gastrointest Endosc, 71 (2010), pp. 413–419
- 8 T. Nguyen-Tang, K.F. Binmoeller, A. Sanchez-Yague, J.N. Shah; Endoscopic ultrasound (EUS)-guided transhepatic anterograde self-expandable metal stent (SEMS) placement across malignant biliary obstruction; Endoscopy, 42 (2010), pp. 232–236
- 9 G. Soulez, M. Gagner, E. Therasse, et al.; Malignant biliary obstruction: preliminary results of palliative treatment with hepaticogastrostomy under fluoroscopic, endoscopic and laparoscopic guidance; Radiology, 192 (1994), pp. 241–246
- 10 L. Tipaldi; A simplified percutaneous hepatogastric drainage technique for malignant biliary obstruction; Cardiovasc Intervent Radiol, 18 (1995), pp. 333–336
- 11 K.W. Warren, J.W. Harmon; Hepaticogastrostomy: ulcerogenic preparation or therapeutic alternative; Ann Surg, 181 (1975), pp. 5–8
- 12 C.M. Moss, C.J. Schein; Nonanastomotic hepaticogastrostomy for palliative decompression; Surg Gynecol Obstet, 142 (1976), pp. 587–589
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?