Abstract
Objectives
Although drug eluting stents (DES) have documented convenience in bifurcation lesions, possible unfavorable effects on small side branch ostium (SBO) remain a question. We aimed to explore the effects of DES on small jailed SBs (1.5–2.25 mm) which originated from the lesion on the main vessel and were not treated with either stenting or balloon dilatation.
Methods
Angiographic data of 107 consecutive patients (129 SB) with Medina 1,1,1 or 1,1,0 lesions were evaluated at the time of procedure and at the follow-up.
Results
Of all DES used, 70 (54.7%) was sirolimus-eluting, 39 (30.5%) was paclitaxel-eluting and 20 (14.8%) was zotarolimus-eluting. The diameter of SBs was 1.84 ± 0.41 mm with a stenosis of 20.7 ± 26.6% at SBO at baseline. The lesion at the SBO had progressed after the procedure when the pre vs postprocedure values and follow-up vs pre-procedure values are compared (20.7 ± 26.6% vs 29.4 ± 27.4%; p < 0.0001 and 25.4 ± 25.1 vs 20.7 ± 26.6%; p = 0.004 respectively). A significant reduction in stenosis was revealed over the follow-up (29.4 ± 27.4 vs 25.4 ± 25.1 respectively; p = 0.013). The severity of the disease at the SBO at baseline was the only parameter that affected the severity of SB stenosis in acute, longterm and follow-up. Additional parameters with influence on SB patency at different times were female gender, stent deployment with low pressure, cTFC of the main lesion, age, cTFC of the lesion, late loss index and the preprocedure TIMI flow grade of the SB.
Conclusions
Although there was a significant deterioration of SBO immediately after stenting, follow-up data showed that the lesion at SBO improved but remained worse than baseline.
Keywords
Drug eluting stents ; Bifurcation lesions ; Side branch
1. Introduction
Coronary bifurcation sites which are under pronounced endothelial shear stress, are particularly prone to atherosclerotic process [1] . Bifurcation lesions represent one fifth of all coronary interventions with higher complication and lower long term patency rates in which, both long term restenosis and acute occlusion/narrowing pose major problems for the treatment [2] . Current approach is treating the main branch (MB) with a stent and provisional stenting of the clinically significant side branch (SB) [2] and [3] . Consequently, some of the smaller side branches are left untreated to their fate on operators judgment or due to technical challenges. With bare metal stents, the SBs at the level of main lesion were frequently sacrificed. After introduction of drug eluting stents (DES), positive data have been accumulated on the stent behavior and low rates of restenosis, making DES as the stent of choice in most patients [2] . Yet, skepticism on the possible unfavorable effects of antiproliferative and anti-inflammatory properties of DES on endothelization of small SB with the risk of early or late thrombosis remains as a question.
Herein this study, we aimed to explore the immediate and long term (6 months) effects of DES on small jailed SBs (1.5–2.25 mm) which originate at the level of the lesion on the MB and do not receive any intervention.
2. Methods
In the study, a ‘bifurcation lesion’ represented a coronary lesion which resulted in narrowing of MB at the level where a SB originates, while the side branch ostium (SBO) may or may not be diseased. This definition included Medina 1,1,0 and 1,1,1 lesions. Only the small side branches with a diameter of 1.5 mm to 2.25 mm originating at the level of lesion and were not treated with dilatation or stenting are analyzed.
After approval from local Ethics Committee, 107 consecutive patients with stable coronary artery disease and bifurcation lesions in which a DES (Sirolimus eluting stent (SES), paclitaxel eluting stent (PES) or Zotarolimus eluting stent (ZES)) was implanted were involved to the study. All patients had control coronary angiography and they gave informed consent before inclusion. The study protocol complied with the Declaration of Helsinki.
Medical and angiographic records of 87 male and 20 female patients with a total of 129 SBs were evaluated. The traditional risk factors of the patients recorded were gender, age, the presence of diabetes mellitus, hypertension, family history of coronary artery disease, hyperlipidemia and smoking. Age was categorized in three categories as < 50, 50–65, > 65 years. The lesions were located on branching points of left anterior descending artery (LAD), circumflex artery (Cx), right coronary artery (RCA) and ‘protected’ left main coronary artery (LMCA).
3. Angiographic data
Both of the lesion on the MB and lesion on the SB, if any, were evaluated quantitatively during coronary angiography. All quantitative angiographic measurements were derived from QCA-Telemedicine Technologies, MEDCON software. The measurements were made in diastole after calibration with the guiding catheter. The angiographic data were analyzed before the procedure, immediately after the procedure and at the follow-up coronary angiogram. At the time of follow-up CAG, segment within 5 mm proximal and distal ends of the stent were evaluated.
Among the angiographic parameters evaluated were lesion length, reference diameter of MB, percent stenosis on the MB, lesion type classified by ACC/AHA [4] , TIMI flow grade [5] , corrected TIMI frame count (cTFC) [6] of the MB and SB. Corrected TIMI frame count (cTCT), which is an index of coronary perfusion, is considered as a predictor of restenosis. It involves the frame count of contrast agent from the ostia to the standardized segments of coronary arteries. For LAD, which is longer when compared to other coronaries, this number is corrected by dividing the calculated number by 1.7 [6] . In the study, cTFC is calculated for both main and side branches. The cTFC of the side branches were calculated as the number of frames elapsed from the ostia to the end of the evaluated side branch. Whether a predilatation of the MB was employed or the maximal pressure achieved during stent deployment were also recorded. A pressure higher than 14 atmospheres was designated as high pressure inflation. Late loss index (late loss/acute gain) represents the percent loss of lumen diameter which was gained immediately after the procedure and was also evaluated to reveal the influence of restenosis on SBO. The SB involved was not treated with a stent or balloon due to small vessel size or difficult rewiring.
The difference between the severity of stenosis of SBO before and immediately after the procedure reflects the acute effect of the procedure and is designated as ‘acute effect ’ in the text and tables. The difference between the pre-procedure stenosis and stenosis of SB at the follow-up angiogram, on the other hand, represents the long term effect of the procedure and is entitled accordingly. Atherosclerosis is a progressive disease and the course of the disease at the SBO during follow-up is also evaluated by comparing the percent stenosis difference between the immediately post-procedure and follow-up angiogram of the side branches. For the purpose of the study this is designated as ‘disease course ’.
4. Statistical analysis
Both mean ± standard deviation and median (minimum–maximum) values are used in the descriptive statistics of the data. The distribution of the variables is tested by Kolmogorov–Simirnov test. The quantitative data are analyzed by Mann–Whitney U test. Repetitive measurements were analyzed by Wilcoxon test and Mc Nemar test. The correlation of the variables is evaluated by Spearman correlation analysis. All data is analyzed by SPSS 22.0 Software.
5. Results
A total of 107 patients, 20(18.7%) female and 87(81.3%) male with a mean age of 60.9 ± 10.1 years, are involved into the study. A follow-up CAG was carried out within 7.44 ± 3.1 months of index procedure. Of all DES used, 70 (54.7%) was SES, 39 (30.5%) was PES and 20 (14.8%) was ZES.
The demographic and angiographic characteristics of the study population are presented in Table 1 . Based on the demographic characteristics, one third of the patients were diabetic with relatively high risk criteria. Similarly, one third of the lesions possessed angiographically high risk characteristics (ACC AHA classification B2 and C). In half of the patients, predilatation with 13.76 ± 1.5 atmosphere was employed. The diameter of SBs was 1.84 ± 0.41 mm with a stenosis of 20.7 ± 26.6% at the SBO.
| Number | Percent (%) | Mean ± SD | ||
|---|---|---|---|---|
| Demographic data | ||||
| Female | 20 | 18.7 | ||
| Age, yrs | 60.9±10.0 | |||
| Hypertension | 101 | 78.3 | ||
| Hyperchlosterolemia | 91 | 70.5 | ||
| Diabetes mellitus | 48 | 37.2 | ||
| Smoking | 41 | 31.8 | ||
| Family history | 30 | 23.3 | ||
| Angiographic data | ||||
| Main vessel | LAD | 76 | 58.9 | |
| Cx | 17 | 13.2 | ||
| RCA | 30 | 23.2 | ||
| Obtus | 3 | 2.3 | ||
| Saphenous | 1 | 0.8 | ||
| LMCA | 2 | 1.6 | ||
| Reference diameter, mm | 2.84±0.39 | |||
| Stent diameter, mm | 2.89±0.34 | |||
| Lesion length, mm | 13.53±7.5 | |||
| Lesion stenosis,% | 73.39±13.3 | |||
| Stent length, mm | 20.38±7.4 | |||
| Lesion type | A | 44 | 34.1 | |
| B1 | 40 | 31.0 | ||
| B2 | 21 | 16.3 | ||
| C | 24 | 18.6 | ||
| Predilatation | 73 | 56.6 | ||
| Maximum pressure, atm | 13.8±1.5 | |||
| Diameter of side branch, mm | 1.84±0.41 | |||
| Lesion at side branch ostium, % | 20.7±26.6 | |||
| TIMI flow of side branch | 2.63±0.7 | |||
| Open stent design | 25 | 19.4 | ||
| Drug type | SES | 70 | 54.3 | |
| PES | 39 | 30.2 | ||
| ZES | 20 | 15.6 | ||
When all the SBs are evaluated, 5 SBs (4%) were acutely lost after the procedure, while the follow-up CAG revealed that 3 out of 5 acutely occluded SBs were recanalized. Only 1 SB was seen to be occluded on the follow-up angiogram after good immediate result (0.008%).
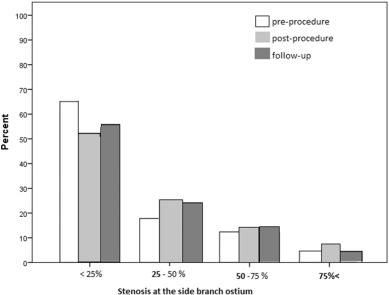
A more detailed evaluation of outcome of MB stenting on SBO is summarized in Table 2 and Table 3 and Fig. 1 . The lesion at the SBO had progressed severely after the procedure when the pre and post-procedural values for lesion at SBO are compared (21 ± 27.3 vs 29.4 ± 27.4 respectively; p < 0.0001). Similarly, there was a significant increase in stenosis when the follow-up and pre-procedure values were compared (25.4 ± 25.1 vs 21.5 ± 27.3 respectively; p = 0.004). Interestingly, when we compared the post-procedure and follow-up stenosis rates, a significant reduction in stenosis was revealed over the follow-up (29.4 ± 27.4 vs 25.4 ± 25.1 respectively; p = 0.013) (Table 2 ). Table 3 shows the acute effect of the procedure on the distribution of stenosis rates at SBO. There was a significant increase in the number of severely diseased SB immediately after the procedure (p = 0.007).
| Pre-procedure | Post-procedure | P* | Follow-up | Pα | P# | |
|---|---|---|---|---|---|---|
| Mean±SD | 20.7±26.6 | 29.4±27.4 | <0.0001 | 25.4±25.1 | 0.004 | 0.013 |
| Me (min–max) | 10 (0–100) | 23 (0–100) | 19 (0–100) |
Wilcoxon test.
P* — preprocedure vs postprocedure.
Pα — preprocedure vs follow-up.
P# — postprocedure vs follow-up.
p < 0.05 is considered statistically significant and represented in bold.
| Pre-procedure | Post-procedure | p⁎ | ||||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
| Lesion at SB ostium | <25% | 84 | 65% | 67 | 52% | 0.007 |
| 25–50% | 23 | 18% | 33 | 26% | ||
| 50–75 | 16 | 12% | 19 | 15% | ||
| 75< | 6 | 5% | 10 | 8% | ||
Mc Nemar test.
p < 0.05 is considered statistically significant and represented in bold.
|
|
|
Fig. 1. Evolution of disease at side branch ostium. |
The results of the study showed that the severity of the disease at the SBO before the procedure is the only parameter that affected the severity of SB stenosis in acute, longterm and follow-up results. Additional parameters with an influence on SB patency are as follows:
- Female gender (p = 0.011), stent deployment with low pressure (< 14 atm) (p = 0.008) and cTFC of the main lesion (p = 0.043) are shown to be the other determinants of the acute effects of MB stenting.
- Among the parameters affecting the patency of SB ostia on the long term are; female gender (p = 0.001), age between 50 and 65 years (p = 0.002), cTFC of the lesion (p = 0.005).
- The factors affecting the disease course at SB ostium are shown to be late loss index of the stent on the MB (p = 0.043) and the preprocedure TIMI flow grade of the SB (p = 0.000).
The parameters evaluated but not shown to have influence on the patency of SBO at any time are the presence of cardiovascular risk factors (diabetes mellitus, hypertension, family history, smoking, hyperlipidemia), stent type (SES, PES or ZES), characteristics of the lesion on MB, the diameter of SB and the cTFC of the SB.
6. Discussion
The treatment of bifurcation lesions continues to be a challenge with no simple solution appropriate for every specific patient. When the SB is small, it is usually compromised for the sake of MB, sometimes leading to dramatic disappearance of the SB. Acute SB occlusion is the most common cause of periprocedural myocardial infarction and is associated with increased mortality [7] and [8] . Recurrence of anginal symptoms due to diseased SBO constitutes another problem. Therefore, the fate of small side branches that did not receive any treatment during stent implantation on main vessel is of considerable concern.
Herein this study, we wanted to explore the outcome of relatively small jailed SBs (1.5–2.25 mm) that arise from the diseased segment of the artery and received no treatment after DES implantation on the MB. The bifurcation lesions involved were Medina 1,1,0 or 1,1,1 and the DES used were SES, PES and ZES. We evaluated the disease at the SBO quantitatively and recorded the % luminal stenosis at SBO together with the acute total occlusion rates.
Our study revealed that MB stenting has unfavorable immediate and long term effects on small SBs assessed by % luminal stenosis at SBO. The deterioration of SBO immediately after stenting was shown to decrease on the follow-up but remained worse than the pre-intervention state.
In our study, the rate of acute occlusion of SB after DES implantation in stable coronary artery disease was shown to be 4%. Fischman et al. have documented in their study with BMS that acute SB occlusion occurred in 6% of patients while other investigators have proposed various results between 7 and 41% [9] , [10] , [11] and [12] . For SES, the rate of SB occlusion is shown to be 10% [13] . For everolimus eluting stents, on the other hand, the incidence of acute SB occlusion is reported as 4.1% [14] . This ratio defers depending on the clinical scenario with the higher rates of SB occlusion (22.5%) seen after stenting of the culprit lesion in acute myocardial infarction [15] .
The only predictor of SB occlusion in acute and chronic phase together with the disease course is the % stenosis of the SB ostium before the procedure [13] . This seems to be the result of plaque and carina shift towards SBO [16] and [17] . Plaque shift or ‘snowplow phenomenon’ includes the shift of the atherosclerotic plaque towards already diseased SB [16] . Previous BMS studies have also shown that the predictors of acute SB occlusion were stenotic SBO, the presence of SB originating from the lesion, TIMI flow lower than 3 on SB and balloon/ artery ratio [18] , [19] , [20] and [21] . In the RAVEL study, the only predictor of acute SB occlusion with SES implantation was shown to be the presence of severe lesion on the SBO [13] .
In our study, female gender was shown to have significant effect on both acute and long term results of DES implantation. This can be explained by the SB characteristics of female patients, since they tend to have smaller (1.7 mm in females vs 1.9 mm in males) and more severely diseased SBs (30% stenosis vs 18%). Another potential mechanism is higher incidence of elastic recoil, vasospasm, and dissection in female. These potentially reversible mechanisms of acute occlusion can also explain the 60% recanalization rate of acutely occluded SBs.
Interestingly, age between 50 and 65 years, which represented 51% of the study population, was associated with worse outcome of SB after DES implantation. Result was cross-checked by testing the correlation of age as a continuous numeric variable with stenosis rates on SBO which revealed no correlation (p = 0.149). This is obviously the result of the higher number of patients in this particular group and also the progressive nature of the disease.
Another predictor of acute SB occlusion was identified as the lower pressure stent (< 14 atm) deployment. The study revealed that stent deployment with a pressure lower than 14 atm is associated with worse acute outcome on SB. This is in contradiction to the studies of BMS that identified high pressure postdilatation (15.5 ± 3.3 atm) as a risk factor for SB occlusion [22] . For DES, on the other hand, the optimum pressure is the one that provides best stent apposition and strut opening [23] . In our study, in which the mean reference diameter was 2.84 mm, this optimum pressure seems to be more than 14 atm. Stent malapposition may result in delayed endothelization and formation of thrombi between the stent struts and SBO compromising the flow.
The cTFC of the main lesion reflects the functional severity and plaque burden of the lesion, and it is inevitable to see that lesions with higher cTFC have higher incidence of acute and chronic deterioration of the SB.
There has been no proven relationship between the open cell stent design and SB occlusion. Cho et al. could not demonstrate any effect of slotted tube, coil and hybrid stent design on acute or chronic SB occlusion [24] . In our study, results show that, open cell design may have possible favorable effects on SB patency. The low incidence of SB occlusion and predominance of stents with closed cell design may have precluded a statistically significant difference.
The main limitation of the study is the lack of clinical follow-up data. Although postprocedure evaluation of cardiac biomarkers such as CK, CK-MB or troponin could have demonstrated a periprocedural myocardial infarction; inclusion of such data would not provide further information on SB patency. The relatively small number of patients which may limit the statistical power of the study is also a limitation.
7. Conclusion
With coronary stenting, most of the small SB originating at the level of main lesion (Medina 1.1.0,1.1.1.) receive no intervention. In our study, we sought to explore the acute and long term outcome of such jailed SBs and found that; although there was a significant deterioration of SBO immediately after stenting, follow-up data showed that the lesion at SBO has decreased to some extent but remained worse than the original state. The acute occlusion, however, occurred in only 4% of SB with a recanalization rate of 60%. The late total occlusion rate was extremely low (0.008). The disease involvement of SBO was identified as the main predictor of all times outcome, together with Female gender, lower pressure stent deployment, cTFC of the main lesion and late loss index of the stent as the contributors to acute or long term outcome.
Conflicts of interest
The authors report no relationships that could be construed as a conflict of interest.
Acknowledgements
None.
References
- [1] J.J. Wentzel, Y.S. Chatzizisis, F.J.H. Gijsen, G.D. Giannoglou, C.L. Feldman, P.H. Stone; Endothelial shear stress in the evolution of coronary atherosclerotic plaque and vascular remodelling: current understanding and remaining questions; Cardiovasc. Res., 96 (2012), pp. 234–243
- [2] T.K. Steigen, M. Maeng, R. Wiseth, NordicPCI Study Group, et al.; Randomized study on simple versus complex stenting of coronary artery bifurcation lesions: the Nordic Bifurcation Study; Circulation, 114 (18) (2006), pp. 1955–1961
- [3] T. Lefevre, Y. Louvard, M.C. Morice, C. Loubeyre, J.F. Piechaud, P. Dumas; Stenting of bifurcation lesions: a rational approach; J. Interv. Cardiol., 14 (6) (2001), pp. 573–585
- [4] S.C. Smith Jr., J.T. Dove, A.K. Jacobs, American College of Cardiology, et al.; American Heart Association Task Force on practice guidelines. Committee to revise the 93 guidelines for percutaneous transluminal coronary angioplasty; J. Am. Coll. Cardiol., 37 (8) (Jun 15 2001), pp. 2215–2239
- [5] The TIMI Study Group; The Thrombolysis in Myocardial Infarction (TIMI) trial: phase I findings; N. Engl. J. Med., 33 (1984), pp. 523–530
- [6] TIMI 4 Study Group; TIMI Frame Count, a quantitative method of assessing coronary artery flow; Circulation, 93 (1996), pp. 879–888
- [7] J.Y. Hahn, W.J. Chun, J.H. Kim, et al.; Predictors and outcomes of side branch occlusion after main vessel stenting in coronary bifurcation lesions, results from the COBIS II Registry (COronary BIfurcation Stenting); J. Am. Coll. Cardiol., 62 (2013), pp. 1654–1659
- [8] D.W. Park, Y.H. Kim, S.C. Yun; Frequency, causes, predictors, and clinical significance of peri-procedural myocardial infarction following percutaneous coronary intervention; Eur. Heart J., 34 (2013), pp. 1662–1669
- [9] D.L. Fischmann, M.P. Savage, M.B. Leon, et al.; Fate of lesion related side branches after coronary stenting; J. Am. Coll. Cardiol., 22 (1993), pp. 1641–1646
- [10] T. Poerner, S. Kralev, et al.; Natural history of small and medium sized side branches after coronary stent implantation; Am. Heart J., 143 (2002), pp. 627–635
- [11] R. Ishiki, K. Hara, Y. Ikari, M. Yamasaki, T. Yamaguchi, T. Tamura; Patency of intermediate size side branch occlusion after coronary Palmaz Schatz stent implantation; Jpn. Heart J., 29 (1997), pp. 261–266
- [12] B. Meier, A.R. Gruentzig, S.B. King, et al.; Risk of side branch occlusion during coronary angioplasty; Am. J. Cardiol., 53 (1984), pp. 10–14
- [13] K. Tanabe, P.W. Serruys, M. Degertekin, et al.; Fate of side branches after coronary arterial sirolimus eluting stent implantation; Am. J. Cardiol., 90 (2002), pp. 937–941
- [14] T. Muramatsu, Y. Onuma, Garcı'a-Garcı'a, on behalf of the ABSORB-EXTEND Investigators, et al.; Incidence and short-term clinical outcomes of small side branch occlusion after implantation of an Everolimus-eluting Bioresorbable Vascular Scaffold; J. Am. Coll. Cardiol. Intv., 6 (2013), pp. 247–257
- [15] V. Khoo, L. Shen, V. Khoo, et al.; Incidence and predictors of side branch compromise in primary percutaneous coronary intervention for acute myocardial infarction; Invasive Cardiol. (2014), pp. 297–302
- [16] J.M. Ahmed, G.S. Mintz, N.J. Weissman, et al.; Mechanism of lumen enlargement during intracoronary stent implantation: an intravascular ultrasound study; Circulation, 102 (2000), pp. 7–10
- [17] B.K. Koo, K. Waseda, H.Y. Kang, et al.; Anatomic and functional evaluation of bifurcation lesions undergoing percutaneous coronary intervention; Circ. Cardiovasc. Interv., 3 (2010), pp. 113–119
- [18] T.C. Poerner, S. Kralev, W. Voelker, et al.; Natural history of small and medium sized side branches after coronary stent implantation; Am. Heart J., 143 (2002), pp. 627–635
- [19] F. Alfonso, C. Hernández, M.J. Pérez-Vizcayno, et al.; Fate of stent related side branches after coronary intervention in patients with in-stent restenosis; J. Am. Coll. Cardiol., 36 (2000), pp. 1549–1556
- [20] G.Y. Cho, C.W. Lee, M.K. Hong, J.J. Kim, S.W. Park, S.J. Park; Effects of stent design on side branch occlusion after coronary stent placement; Catheter. Cardiovasc. Interv., 52 (2001), pp. 18–23
- [21] B. Bhargava, R. Waksman, A.J. Lansky, R. Kornowski, R. Mehran, M.B. Leon; Clinical outcomes of compromised side branch after coronary stenting with the NIR stent; Catheter. Cardiovasc. Interv., 54 (2001), pp. 295–300
- [22] D. Aliabadi, F.V. Tilli, T.R. Bowers, et al.; Incidence and angiographic predictors of side branch occlusion following high pressure intracoronary stenting; Am. J. Cardiol., 80 (1997), pp. 994–997
- [23] O. Frobert, G. Sarno, S.K. James, N. Saleh, B. Lagerqvist; Effect of stent inflation pressure and post-dilatation on the outcome of coronary artery intervention. A report of more than 90 000 stent implantations; PLoS One, 8 (2) (2013), p. e56348
- [24] G.Y. Cho, C.W. Lee, M.K. Hong, J.J. Kim, S.W. Park, S.J. Park; Effects of stent design on side branch occlusion after coronary stent placement; Catheter. Cardiovasc. Interv., 52 (2001), pp. 18–23
Document information
Published on 19/07/16
Licence: CC BY-NC-SA license
Share this document
Keywords
claim authorship
Are you one of the authors of this document?