Abstract
Background
Levosimendan improves clinical and hemodynamic parameters exerting an anti-inflammatory and antiapoptotic effect in decompensated heart failure. The aim of this study was to evaluate the effects of levosimendan on LV torsion, plasma levels of NT-proBNP and on the balance between pro-inflammatory (TNF-α, IL-6) and anti-inflammatory cytokines (IL-10).
Methods
We enrolled 24 patients (age 62 ± 7 years) with acute HF, NYHA class IV and severe LV dysfunction. All patients underwent transthoracic echocardiography using two-dimensional speckle tracking analysis to detect LV twist angle (LVTA), at baseline and 1 week after treatment with levosimendan infusion. Biochemical parameters (pro-BNP, IL-6, IL-10, TNF-α) were determined by enzyme-linked immunosorbent (ELISA).
Results
After one week, we observed an improvement in LV function especially in LVTA (4.15 ± 2.54 vs 2.9 ± 2.1 p < 0.01), in LV ejection fraction (27.3 ± 8.04 vs 21.6 ± 6.8 p = 0.03) and also a significant reduction in BNP levels (1844 ± 560 vs 4713 ± 1050, p = 0.03). The multiple linear regression analysis showed a significant relation between a reduction of TNF α/IL-10 ratio (Δ > 20%) and BNP (Δ > 40%), LVEF (Δ > 10%) and LVTA (Δ > 20%) (O.R. 1.77, 95% C.I. 1.11–2.83; O.R. 1.49, 95% C.I. 1.08–2.67; O.R. 1.66, 95% C.I. 1.10–2.74, respectively, confirmed p, all < 0.01 by Hosmer–Lemeshov confirmation and the formal test for interaction).
Conclusions
Levosimendan exerts a hemodynamic effect by improving EF and LV torsion in patients with acute HF in association with a positive effect on the balance between pro and anti-inflammatory cytokines.
Keywords
Heart failure;Levosimendan;Strain echocardiography;Cytokines
1. Introduction
Levosimendan is a calcium sensitizing drug that improves cardiac contractility in systole without impairing relaxation in diastole. This drug may also have a vasodilator action which results in improved cardiac output without increasing myocardial oxygen consumption. An active metabolite of this drug (OR1896) maintains the clinical response for up to 9 days from the end of the continuous infusion [2].
The clinical effect of levosimendan, mainly in terms of reduced mortality or prolonged bridging window to non-medical therapies for advanced heart failure (HF), is still under debate but its efficacy in several settings of cardiovascular disease is likely to be proven in various studies [3].
Left ventricular (LV) torsion is a fundamental component of the normal LV systolic contraction that plays an important role to optimize LV ejection fraction (LVEF). It arises from the reciprocal rotation of the LV apex and base during systole, created by the contraction of its oblique spiral fibers. The evaluation of LV twist angle (LVTA) has been possible by speckle tracking echocardiography (STE) analyzing the net difference in mean rotation between the apex (counterclockwise) and the base (clockwise) [4]. STE has recently been proposed and validated as a feasible method for measuring LV rotation and torsion [5].
Levosimendan also seems to have anti-inflammatory and anti-apoptotic properties in decompensated heart failure [1]. Pro-inflammatory cytokines like IL-6 and TNF-α depress myocardial contractility, promote cardiomyocyte apoptosis and contribute to cardiac remodeling [6]; [7] ; [8].
The activity of inflammatory cytokines is also influenced by anti-inflammatory molecules such us interleukin (IL-10) which can downregulate the production of several inflammatory mediators from macrophages and other immune cells [9].
The aim of this study was to investigate the role of levosimendan on systolic function especially on myocardial torsional dynamics and on biochemical parameters like brain natriuretic peptide (BNP) levels and on pro/anti-inflammatory cytokine imbalance.
2. Methods
2.1. Study population
We enrolled twenty-four consecutive patients (age 62 ± 7 years) with acute NYHA class IV heart failure and severe LV dysfunction (LVEF < 30%). Informed consent was obtained from all subjects and the protocol was approved by the ethics committee of the recruiting center as an extension of previous authorization for similar experimental models [12]. Exclusion criteria were: systolic blood pressure < 80 mm Hg, heart rate > 130 bpm, recent myocardial infarction (< 8 weeks) or active myocardial ischemia, serum creatinine levels higher than 2.5 mg/dl (or glomerular filtration rate less than 30 ml/min) or dialysis, hepatic failure, acute or chronic infectious or inflammatory diseases. All patients were on appropriate medical therapy including beta-blockers, ACE-inhibitors and diuretics and all patients were treated additionally with continuous 24-h infusion of levosimendan at 0.1 μg/kg/min without an intravenous loading dose (see Table 1 for details).
| Age (years) | 62 ± 7 |
| Sex, male | 8 (44%) |
| New York Heart Association | IV (100%) |
| Ischemic/Non ischemic patients | 12/12 (50%) |
| Medicaments | |
| Diuretics | 24 (100%) |
| Angiotensin converting enzyme inhibitors | 14/24 (58%) |
| Angiotensin receptor blockers | 10/24 (42%) |
| Beta-blocker | 8/24 (33%) |
| Statins | 12/24 (50%) |
| Antiaggregants | 12/24 (50%) |
| Spironolactone | 8/24 (33%) |
Qualitative values are expressed as %. No statistical significant differences were found between ischemic and non-ischemic dilated cardiomyopathy.
2.2. Standard echocardiography
Echocardiographic studies were performed using a high quality portable echocardiograph (Vivid i GE USA) equipped with a 2.5 MHz transducer. Subjects were studied in the left lateral recumbent position.
Echocardiography was performed by experienced physicians at baseline and one week after levosimendan treatment. Measurements of LV end diastolic and end systolic volumes (LV EDV and LV ESV respectively), left atrial dimensions, LVEF and diastolic LV filling were made in accordance with current recommendations of ASE [10]. LVEF, measured using the biplane modified Simpsons method, was employed as a standard index of global LV systolic function. LV diastolic function was estimated by pulsed wave (PW) Doppler on transmitral flow assessing peak velocities in early (E) and late diastole (A), E/A ratio.
2.3. Tissue Doppler imaging and M-mode annulus excursion analysis
Left ventricular longitudinal function was explored by pulsed tissue Doppler imaging, placing the sample volume at the level of mitral lateral annulus from the apical four chamber view. Peak systolic (Sm), early diastolic (Em), and late diastolic (Am) annular velocities were obtained. The E/Em ratio was also calculated and used as index of LV filling pressures. M-mode measurements of mitral annular plane systolic excursion (MAPSE) were performed by placing the cursor perpendicular to the lateral site of the annulus. Tricuspid annular plane systolic excursion (TAPSE) was also measured using two dimensional guided M-mode imaging [11].
2.4. Speckle tracking echocardiography
For the assessment of LVTA, LV short-axis planes were obtained at basal and apical levels at adequate frame rates (range 60–90 fps). The basal plane was defined at the level of the mitral valve, whereas the apical plane was acquired distally to the papillary muscles.
STE analysis was performed using a dedicated software (Echo Pac, GE USA). After manual delineation of LV endocardial border using a point-and-click approach a second epicardial tracing was automatically generated by the system. The software automatically segmented the LV circumference into six myocardial segments generating rotation curves for each segment. Two mean LV rotation curves representing basal and apical rotation were obtained. LVTA was calculated as the net difference in mean rotation between the apical and basal levels. LV untwisting rate was calculated as the early diastolic peak time derivative of the time-twisting angle curve [12].
2.5. Biochemical measurements
Peripheral venous blood samples were collected by a 19 gauge needle without stasis at baseline and one week after treatment with levosimendan infusion to detect cytokine levels (IL-6, IL-10, TNF-α) measured by enzyme-linked immunosorbent assay (ELISA) and plasma BNP levels using the bedside device Triage BNP test (Biosite San Diego, CA) as previously described [12].
2.6. Statistical analysis
Statistical analysis was performed using the SPSS 12.0 statistical package for Windows. Quantitative values were expressed as mean ± standard deviation and qualitative values as %. The Wilcoxons paired test was used to compare values before and after drug administration. A two-tailed p value < 0.05 was considered significant.
A multiple logistic regression analysis was employed to evaluate the putative relations among studied variables once corrected for basic (age, BMI, pharmacological treatments and other established diagnosis) and measurable parameters changes evaluated during the study (PAS, PAD, heart rate, LVEF, LVTA). In particular we categorized a 5% growing differences range (both positive or negative trend, 0–4.9, 5–9.9, 10–14.9% and so on) in parameters of cardiac function (LVESV, LVEDV, LVEF, LVTA and BNP) as the primary end-point to relate with TNFα, IL6, IL10 and each to other ratios. Furthermore the statistical model consisting of a formal test for interaction was employed to determine the putative interaction for each single variable. Briefly, starting from logistic regression analysis in which Y was the parametric obtained modification of each single evaluated parameter; the variables X1 and X2 were the present or not (1 or 0) of categorized ranges of cytokine modifications and X3 the combination. The simplified formula for calculation was: Y = β0 + β1X1 + β2X2 + β3X3 and the null hypothesis was tested as H0:β3 = 0. Final validation of data was assessed by a resampling technique (Exact Tests in SPSS 2003 module) and discrimination analysis by the Hosmer–Lemeshow method [G2HL = ∑10J = 1 (Oj − Ej)2 / Ej (1 − Ej/nj) ~ X28] where nj = number of observations in the jth group, Oj = ∑1yij = observed number of positive cases in the jth group, Ej = ∑ pij = expected number of positive cases in the jth group.
3. Results
After one week, clinical, echocardiographic and neurohumoral controls were performed. The general characteristics and echocardiographic parameters of the studied patients are summarized in Table 1, Table 2 ; Table 3 respectively.
| Parameters | Baseline (N = 24) | One week (N = 24) | p value |
|---|---|---|---|
| LVEDD (mm) | 75.5 ± 11.52 | 75.25 ± 11.14 | p = ns |
| LVESD (mm) | 65.1 ± 10.7 | 64.62 ± 10.11 | p = ns |
| LVEDV (ml) | 314.12 ± 160 | 280.8 ± 96.95 | p = ns |
| LVESV (ml) | 226 ± 125 | 216.75 ± 82.10 | p = ns |
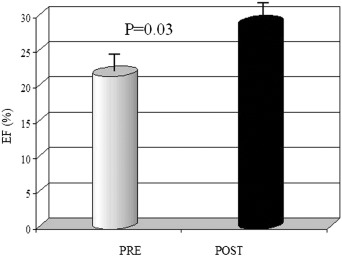
| LVEF (%) | 21.6 ± 6.8 | 27.3 ± 8.04 | p = 0.03 |
| E (m/s) | 0.78 ± 0.2 | 0.77 ± 0.2 | p = ns |
| A (m/s) | 0.3 ± 0.7 | 0.55 ± 0.2 | p = ns |
| Em (m/s) | 0.04 ± 0.01 | 0.05 ± 0.007 | p = ns |
| Am (m/s) | 0.07 ± 0.02 | 0.09 ± 0.04 | p = ns |
| MAPSE (mm) | 9 ± 2 | 11.25 ± 4.02 | p = ns |
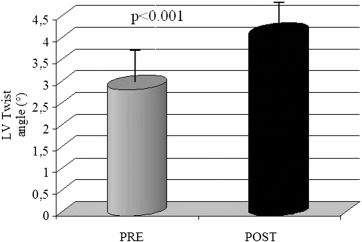
| LV twist angle (°) | 2.9 ± 2.1 | 4.1 ± 2.5 | p = 0.01 |
| LV untwisting | − 39 ± 36.2 | − 35.9 ± 22 | p = ns |
| Strain circumference apex (%) | − 5.76 ± 2.4 | − 5.98 ± 2.6 | p = ns |
| Strain circumference base (%) | − 4.29 ± 2.2 | − 5.35 ± 1.9 | p = ns |
| PVLS (%) | 6.3 ± 3 | 7.3 ± 3.3 | p = ns |
| PALS (%) | 9.3 ± 7.3 | 12 ± 4.7 | p = ns |
Data are expressed as mean ± standard deviation. Quantitative values were expressed as mean ± standard deviation. The Wilcoxons paired test was used to compare values before and after drug administration. A p value < 0.05 was considered significant.
Abbreviations: LVEDD and LVESD left ventricle end-diastolic and end-systolic diameter respectively, LVESV left ventricle end-systolic volume, LVEDV left ventricle end diastolic volume, E early rapid filling wave, A filling wave due to atrial contraction, Em early diastolic annular velocities, Am late diastolic annular velocities, PVLS peak longitudinal strain of left ventricle, and PALS peak longitudinal strain left atrium.
| Parameters | Baseline | One week | p value |
|---|---|---|---|
| IL-6 (pg/ml) | 11.6 ± 1.4 | 8.3 ± 0.9 | p < 0.05 |
| IL-10 (pg/ml) | 5.8 ± 0.5 | 8.8 ± 0.7 | p < 0.001 |
| TNF-α (pg/ml) | 13.1 ± 1.9 | 8.9 ± 0.8 | p < 0.05 |
| TNF- α/IL-10 ratio | 2.22 ± 1.6 | 1.01 ± 0.6 | p < 0.01 |
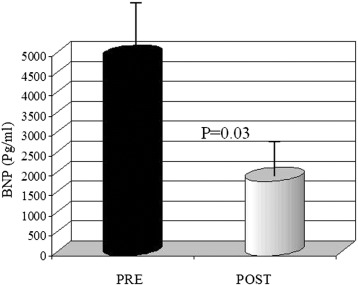
| BNP (pg/ml) | 4713 ± 1050 | 1844 ± 560 | p = 0.03 |
Data are expressed as mean ± standard deviation. The Wilcoxons paired test was used to compare values before and after drug administration. A p value < 0.05 was considered significant.
Abbreviations: BNP brain natriuretic peptide, IL interleukin.
Echocardiographic and speckle tracking evaluation after one week from the end of treatment demonstrated a significant improvement of LV systolic function resulting in an increase of LVEF (27.3 ± 8.04 vs 21.6 ± 6.8 p = 0.03) and LVTA (4.15 ± 2.54 vs 2.9 ± 2.1 p < 0.01) (Fig. 1 ; Fig. 2).
|
|
|
Fig. 1. Variation in LV ejection fraction after treatment with levosimendan. Quantitative values were expressed as mean ± standard deviation. The Wilcoxons paired test was used to compare values before and after drug administration. A p value < 0.05 was considered significant. |
|
|
|
Fig. 2. Increase of LV twist angle after levosimendan. Quantitative values were expressed as mean ± standard deviation. The Wilcoxons paired test was used to compare values before and after drug administration. A p value < 0.05 was considered significant. |
Even M-mode mitral annular plane systolic excursion (MAPSE), peak of left ventricle longitudinal strain (PVLS) and peak left atrium longitudinal strain (PALS) were not significantly increased (Table 2). We also observed a significant reduction in BNP levels after one week from the end of levosimendan infusion (1844 ± 560 vs 4713 ± 1050, p = 0.03). Furthermore, a significant decrease of TNF-α and IL-6 was observed after one week on treatment with levosimendan (8.9 vs 13.1 pg/ml and 8.3 us 11.6 respectively, both p < 0.05) whereas IL-10 was significantly increased (p < 0.001) (Fig. 3 and Table 3).
|
|
|
Fig. 3. Decreased levels of BNP after treatment with levosimendan. Quantitative values were expressed as mean ± standard deviation. The Wilcoxons paired test was used to compare values before and after drug administration. A p value < 0.05 was considered significant. |
Moreover we found a linear correlation between reduction of TNF-α/IL-10 ratio and reduction on BNP levels at one week (r = 0.49, p = 0.0001 if increase of IL-10 was more of 20%).
It was also demonstrated a negative correlation between LVEF and TNF-α/IL-10 ratio (r = -0.44, p < 0.01 if increase of IL-10 > 20%) and a negative correlation between LVTA and TNF-α/IL-10 ratio (r = − 0.44 p < 0.0089). Finally there were no adverse effects of levosimendan treatment in our patient population.
The multiple linear regression analysis showed a significant relation only between a reduction of TNF α/IL-10 ratio (Δ > 20%) and BNP (Δ > 40%), LVEF (Δ > 10%) and LVTA (Δ > 20%) (O.R. 1.77, 95% C.I. 1.11–2.83; O.R. 1.49, 95% C.I. 1.08–2.67; O.R. 1.66, 95% C.I. 1.10–2.74, respectively, confirmed p, all < 0.01 by Hosmer–Lemeshov confirmation and the formal test for interaction).
4. Discussion
Levosimendan induced changes in hemodynamic and clinical parameters. Additionally the effect of this treatment in patients with heart failure has been shown to be reflected in change in plasma concentrations of neurohormonal and inflammatory markers [11].
In the present study a cohort of patients with decompensated heart failure was treated with the calcium sensitizer levosimendan and clinical, echocardiographic and laboratory parameters were evaluated to further assess the putative clinical benefit of this drug.
4.1. Echocardiographic and speckle tracking examination
Levosimendan improves echocardiographic indexes of systolic function after seven days from the end of treatment. Our study demonstrated an increase in LVEF and in LV torsional dynamics in patients with acute heart failure and treated with levosimendan. Previous studies have demonstrated that levosimendan improves LV systolic and diastolic function [13]. LV systolic contraction is a complex phenomenon resulting from interaction among different myocardial layers, leading to simultaneous longitudinal and circumferential shortening, radial thickening and twisting [14]. Left ventricular twisting is a component of the normal LV systolic contraction and represents the instantaneous rotational movement of the apex with respect to the base of the heart, able to generate a wringing motion of the LV that drives the blood out of LV cavity. Indeed during contraction the subendocardial and subepicardial layers oppose each others due to their oblique fiber angle orientations but greater force is generated in the epicardial layers as a result of the longer distance from the center of the ventricle. So the epicardial fibers govern the direction of LV twist determining a counterclockwise torsional movement during systolic ejection. In the dilated heart the left ventricle takes a spherical geometry and this determines the loss of oblique architecture of loop fibers and leads to impair LV torsional movement. The reduction of twisting motion would increase endocardial stress and, therefore, increase oxygen demand, thereby reducing the efficiency of LV systolic function. Levosimendan with its inotropic effect increases myocardial contractility of the subendocardial and subepicardial fibers and, in that way, determines an improvement in LV torsion dynamics that allows a reduction in oxygen consumption.
4.2. Neurohormonal response to levosimendan
In our patient population, levosimendan resulted in a significant reduction of BNP and pro-inflammatory cytokine (TNF-α, IL-6) circulating levels after one week from the end of treatment. This reduction was accompanied by a significant increase of LVEF and LV twisting.
It is well known that overexpression of circulating pro-inflammatory cytokines leads to increased oxidative stress and enhanced cardiomyocyte and endothelial cell apoptosis and plays an important role in the clinical and hemodynamic deterioration of heart failure [15]. At the same time overproduction of BNP in heart failure is a result of elevated end-diastolic cardiac wall stress. High levels of BNP and IL-6 are independent risk factors for morbidity and mortality in patients with heart failure, despite an improvement in LVEF and symptoms after medical therapy [16].
In the present study we demonstrated that levosimendan plays an important role in the imbalance between pro- and anti-inflammatory cytokines: it reduces the levels of IL-6 and TNF-α and increases the anti-inflammatory IL-10 levels that can downregulate the production of several inflammatory mediators from macrophages and other immune cells [8].
In our study we found an important reduction of TNF-α/IL-10 ratio that showed a significant correlation with LV twist, so we can confirm that levosimendan has an inotropic and anti-inflammatory role also in the setting of non-ischemic dilated cardiomyopathy (50% of our patients) with respect to post-ischemic dilated cardiomyopathy as previously demonstrated and actually confirmed [12]. The anti-inflammatory effects of levosimendan could be related to an indirect action on circulating cytokines mediated by the improvement of cardiac hemodynamics. However previous evidences have described as a relevant pathogenetic mechanism the imbalance between pro- and anti-inflammatory cytokines involved even before the development of HF in the initial stages of ischemic acute coronary syndromes [17].
Thus, a direct anti-inflammatory effect of levosimendan can be strongly suggested according to the previous demonstration of downregulation of apoptosis signaling mechanism through inhibition of TNF-α receptor superfamily and Fas/fas ligand system with subsequent positive effect on cardiomyocytes loss as a consequence of apoptotic cell death [12].
Taking into account that the effects of levosimendan in our study have been ascertained in a population of on top medical treated subjects, including several molecules known to exert an anti-inflammatory effect [18] (see Table 1), the incremental positive effect on cytokine imbalance is more likely due to a direct effect than to an indirect one linked to changes in cardiac hemodynamics.
The positive effect of levosimendan in neurohormonal balance has been suggested also for repeated infusions when a clinical improvement was established in terms of HF progression with a positive trend for 1-year mortality [18]. Furthermore the treatment with levosimendan has been suggested to reach an economically positive profile in acute HF [18].
5. Conclusions
Levosimendan exerts a positive effect on hemodynamic and clinical parameters improving myocardial contractility without increasing myocardial oxygen consumption. In patients with advanced heart failure the increase of LV torsional dynamics appears to be a compensatory mechanism of the failing heart.
Our study demonstrates that levosimendan also induces changes in neurohumoral markers having a beneficial influence in the balance between pro- and anti-inflammatory cytokines and in cardiac remodeling in patients with HF of ischemic or other etiology.
6. Limitations
An important limitation of the clinical use of STE is that to obtain correct endocardial border delineation an adequate echocardiographic view is recommended and furthermore STE is closely dependent from heart rate and it is not possible to conduct strain measurements in patients with non-sinus rhythms. Furthermore, the latter reduces the number of eligible patients in the setting of HF.
Sources of funding
The study was partially supported by a Grant from University of Siena (PAR 2007) to LP
Conflict of interest
The authors report no relationships that could be construed as a conflict of interest.
References
- [1] C.A. Innes, A.J. Wagstaff; Levosimendan: a review of its use in the management of acute decompensated heart failure; Drugs, 63 (2003), pp. 2651–2671 [Pubmed: 14636085]
- [2] G. Landoni, G. Biondi-Zoccai, M. Greco, et al.; Effects of levosimendan on mortality and hospitalization. A meta-analysis of randomized controlled studies; Crit Care Med, 40 (2012), pp. 634–646 [Pubmed: 21963578]
- [3] S. Mondillo, M. Galderisi, D. Mele, et al.; Speckle tracking echocardiography: a new technique for assessing myocardial function; J Ultrasound Med, 30 (2011), pp. 71–83 [Pubmed: 21193707]
- [4] T. Helle-Valle, J. Crosby, T. Edvardsen, et al.; New noninvasive method for assessment of left ventricular rotation: speckle tracking echocardiography; Circulation, 112 (2005), pp. 3149–3156 [Pubmed:16286606]
- [5] P. Aukrust, T. Ueland, E. Lien, et al.; Cytokine network in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy; Am J Cardiol, 83 (1999), pp. 376–382 [Pubmed:10072227]
- [6] A.M. Feldman, A. Combes, D. Wagner, et al.; The role of tumor necrosis factor in the pathophysiology of heart failure; J Am Coll Cardiol, 35 (2000), pp. 537–544 [Pubmed 10716453]
- [7] G. Plenz, Z.F. Song, T.D. Tjan, et al.; Activation of the cardiac interleukin-6 system in advanced heart failure; Eur J Heart Fail, 3 (2001), pp. 415–421 [Pubmed 11511426]
- [8] Z. Mallat, S. Besnard, M. Duriez, et al.; Protective role of interleukin 10 in atherosclerosis; Circ Res (1999), pp. 17–24 [Pubmed 10521249]
- [9] RM. Lang, M. Bierig, RB Devereux, et al.; Recommendations for chamber quantification: a report from the American Society of Echocardiographys Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a Branch of the European Society of Cardiology.; J Am Soc Echocardiogr, 18 (2005), pp. 1440–1463 [Pubmed 16376782]
- [10] C.M. Yu, J.E. Sanderson, T.H. Marwick, et al.; Tissue Doppler imaging a new prognosticator for cardiovascular disease; J Am Coll Cardiol, 49 (2007), pp. 1903–1914 [Pubmed 17498573]
- [11] S. Mondillo, M. Galderisi, D. Mele, et al.; Speckle tracking echocardiography; J Ultrasound Med, 30 (2011), pp. 71–83 [Pubmed 21193707]
- [12] S. Lunghetti, E. Palmerini, R. Urselli, et al.; Effects of levosimendan without loading dose on systolic and diastolic function in patients with end-stage heart failure; Cardiol J, 5 (2011), pp. 532–537 [Pubmed 21947989]
- [13] Y. Mizuguchi, Y. Oishi, H. Miyoshi, et al.; The functional role of longitudinal, circumferential and radial myocardial deformation for regulating the early impairment of left ventricular contraction and relaxation in patients with cardiovascular risk factors: a study with two-dimensional strain imaging; J Am Soc Echocardiogr, 21 (2008), pp. 1138–1144 [Pubmed 18926389]
- [14] S. Sasayama, A. Matsumori, Y. Kihara; New insights into the patho-physiological role for cytokines in heart failure; Cardiovasc Res, 42 (1999), pp. 557–564 [Pubmed 10533591]
- [15] S. Adamopoulos, J.T. Parissis, D.T. Kremastinos, et al.; A glossary of circulating cytokines in chronic heart failure; Eur J Heart Fail, 3 (2001), pp. 517–526 [Pubmed 11595599]
- [16] A.L. Pasqui, M. Di Renzo, A. Auteri, et al.; Cytokines in acute coronary syndromes; Int J Cardiol, 105 (2005), pp. 355–356 [Pubmed 16216352]
- [17] L. Puccetti, A.L. Pasqui, F. Scarpini, et al.; Statins discontinuation in compliant chronic users induces atherothrombotic profile despite baseline clinical settings and treatments; Int J Cardiol, 153 (2011), pp. 328–329 [Pubmed 22000266]
- [18] C. Lucioni, A. D'Ambrosi, S. Mazzi, P. Pollesello, M. Apajasalo, F. Fedele; Economic evaluation of levosimendan versus dobutamine for the treatment of acute heart failure in Italy; Adv Ther, 29 (Dec 2012), pp. 1037–1050 [Pubmed 23233357]
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?