Summary
Background
Achalasia is characterized by esophageal aperistalsis and a failure of lower esophageal sphincter (LES) relaxation. Combined multichannel intraluminal impedance and manometry (MII-EM) allows the simultaneous recording of esophageal peristalsis and bolus transport patterns. The aim of this study was to evaluate the feasibility of MII-EM for the assessment of esophageal motility and to characterize patterns of esophageal bolus transport in patients with achalasia with or without Heller myotomy.
Materials and methods
A total of nine patients (2 men and 7 women, age range 25–46 years) were enrolled in this study. Two of the patients had previously undergone Heller myotomy. All patients underwent combined MII-EM with a nine-channel esophageal function testing catheter containing four impedance measuring segments and five solid-state pressure transducers. Each patient received 10 liquid and 10 viscous swallows in a sitting position. All tracings were recorded and analyzed for esophageal contractions and bolus transit.
Results
None of the patients with achalasia, whether they had undergone a Heller myotomy or not, had manometrically normal esophageal peristalsis during saline or viscous swallowing. They had a normal LES resting pressure, incomplete LES relaxation, and lower distal esophageal contraction. The LES relaxation percentages in the patients who had undergone Heller myotomy (97% and 51%) were higher than those of the untreated patients (mean 47%). All patients demonstrated a low baseline impedance level in the distal esophagus. Air trapping in the proximal esophagus was also detected in nearly all of the patients. None of the patients in either group had complete bolus transit with either saline or viscous swallows.
Conclusion
Patients with achalasia are characterized by poor esophageal contraction and absent esophageal bolus clearance and such abnormalities are still noticeable after Heller myotomy. Although combined MII-EM can provide additional information regarding esophageal bolus transit, a low baseline impedance level and air trapping in the proximal esophagus may limit its utility in the diagnosis of esophageal dysmotility in patients with achalasia.
Keywords
Achalasia ; Combined multichannel intraluminal impedance and manometry ; Esophageal bolus transit ; Esophageal motility
Introduction
Achalasia is an uncommon, but important, esophageal motor disorder characterized by esophageal aperistalsis and a failure of lower esophageal sphincter (LES) relaxation. It may occur at any age and has an incidence of 0.6 per 100,000 per year [1] . Although its etiology is unknown, current data suggest that achalasia is an immune-mediated disorder and is caused by a loss of inhibitory nitrinergic neurons in the esophageal myenteric plexus [2] . Dysphagia to solids and liquids, regurgitation of undigested food, and chest pain are the main symptoms in patients with achalasia.
The diagnosis of achalasia relies on endoscopy, barium esophagogram, and manometric parameters. A barium esophagogram can be highly suggestive of the diagnosis of achalasia, particularly when there is a combination of esophageal dilatation with retained barium and minimal LES opening with a “bird beak” appearance; however, in a previous series, the diagnosis of achalasia was suggested in only 64% of barium examinations [3] . Esophageal manometry has the highest sensitivity for the diagnosis of achalasia, although variations occur in manometric findings, especially in the early course of the disease [4] . Thus additional features and methodology by high-resolution manometry have been used to reinforce the diagnostic accuracy [5] ; [22] .
Multichannel intraluminal impedance (MII) measurement is a new technique for the assessment of esophageal bolus transport on the basis of differences in the resistance to alternating current between air, the esophageal mucosa, and an esophageal liquid bolus. The basic principles of impedance testing were first described by Silny [6] in 1991. The ability of the impedance to detect bolus movement within the esophagus has been validated with the use of combined video-fluoroscopy and impedance [7] ; [8] .
When combined with manometry, MII provides a simultaneous evaluation of esophageal contraction and bolus transit, thus revealing the functional aspects of esophageal motility. Studies in patients using combined intraluminal impedance and manometry (MII-EM) have shown functional details of bolus transit in patients with esophageal motility abnormalities [9] ; [10] . The aim of this study was to evaluate the clinical utility of combined MII-EM in the diagnosis of motility characteristics in patients with achalasia and those who had previously undergone Heller myotomy. We also investigated the feasibility of combined MII-EM in the diagnosis of esophageal dysmotility in patients with achalasia.
Materials and methods
Patients
Nine patients with achalasia (2 men and 7 women, mean age 35.7 years, range 25–46 years) underwent combined esophageal MII recording and manometry. The patients were recruited from our outpatient clinic. The diagnosis of achalasia was made based on medical history, endoscopy, manometry, and barium esophagogram according to established criteria. Each of the patients gave written informed consent. The protocol was approved by the medical ethics committees of the Buddhist Tzu Chi Medical Center, Haulien, Taiwan.
Combined esophageal manometry and impedance recording
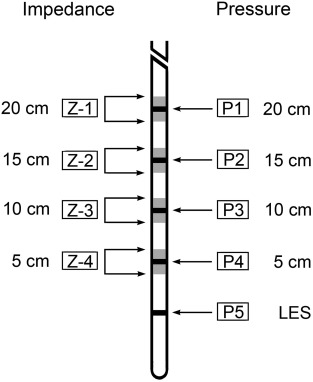
Each patient underwent esophageal function testing using combined MI-EM with a Koenigsberg nine-channel probe (EFT catheter; Sandhill Scientific, Highlands Ranch, CO, USA). This 4.5-mm diameter catheter incorporates five pressure (2 circumferential and 3 unidirectional) sensors and four impedance measuring segments. The two circumferential solid-state pressure sensors are located 5 cm and 10 cm from the tip and the three unidirectional pressure sensors at 15 cm, 20 cm, and 25 cm. The impedance measuring segments consist of pairs of metal rings placed 2 cm apart, centered at 10 cm, 15 cm, 20 cm, and 25 cm from the tip, i.e., across the four proximal pressure transducers (Fig. 1 ). In this system, four of the five pressure sensors are located within the impedance segments and the fifth pressure sensor is located 5 cm from the tip.
|
|
|
Figure 1. Nine-channel combined multichannel intraluminal impedance and manometry catheter. |
Study protocol
Prior to the study, the patients fasted for at least 6 hours. The EFT catheter was inserted transnasally into the esophagus to a depth of 60 cm. The intragastric position of the catheter was verified by the rise in pressure during deep inspiration and the intragastric pressure was set as the baseline pressure. The LES was identified using the stationary pull-through technique and the most distal sensor was placed in the high-pressure zone of the LES. Intraesophageal pressure sensors and impedance measuring segments were thus located at 5 cm, 10 cm, 15 cm, and 20 cm above the LES. The patients were positioned in a sitting position and allowed to accommodate to the assembly for 5–10 minutes. Each patient was then asked to swallow 10 5-mL boluses of normal saline and followed by 10 5-mL boluses of viscous solution (Sandhill Scientific). The swallows were spaced at least 20–30 seconds apart. Normal saline was used instead of regular water as it provides a better impedance change with a standardized ionic concentration. Manometric and impedance signals were recorded by a computerized combined manometry-impedance recording system and displayed and stored on a personal computer for subsequent analysis.
Data analysis
The manometric part of the combined impedance-manomerty tracings is analyzed in a similar way to conventional esophageal manometry. The manometric parameters used to characterize swallows included: (1) the contraction amplitude at 5 cm and 10 cm above the LES; (2) the distal esophageal amplitude as the average of the contraction amplitudes at 5 cm and 10 cm above the LES; and (3) the onset velocity of esophageal contractions in the distal part of the esophagus (i.e., the contraction velocity between 5 cm and 10 cm above the LES). Esophageal contractions are considered as: (1) normal, if contraction amplitude in the distal esophagus is >30 mmHg and the distal esophageal onset velocity is <8 cm/s; (2) ineffective, if the contraction amplitude in the distal esophagus is <30 mmHg; and (3) simultaneous, if the contraction amplitude is >30 mmHg and the distal onset velocity is >8 cm/second in the distal esophagus.
Esophageal bolus transit is monitored by changes in impedance. A swallow is considered to have complete bolus transit if the bolus enters 20 cm above the LES (i.e., a drop in impedance baseline to less than 50% of the difference between baseline and nadir) and there is bolus exit in all three distal impedance measuring segments (i.e., 15 cm, 10 cm, and 5 cm above the LES). A swallow is considered to have an incomplete bolus transit if a bolus exit is not identified at any one of the three distal impedance measuring sites.
Diagnosis of manometric motility abnormalities were based on the criteria published by Spechler and Castell [11] . Achalasia was defined by the absence of distal esophageal body peristalsis supplemented by LES abnormalities (elevated resting pressure and incomplete relaxation) and increased esophageal body peristalsis when present. The bolus transit information complements esophageal peristalsis data, indicating which esophageal motility abnormality is associated with esophageal bolus transit impairment (esophageal function defect).
Results
Patient characteristics
All the studied patients had classical achalasia. None of the patients received botulinum treatment or pneumatic dilatation, but two of the patients had undergone Heller myotomy in the past (Case 1, 5 years previously; Case 2, 2 years previously; Table 1 ). The primary symptoms and signs for the patients were: dysphagia (100%); food or acid regurgitation (100%); chest pain (42%); loss of body weight (42%); heartburn (22%); and globus (11%). The average times between the occurrence of symptoms and diagnosis were 3.3 years (range 1.5–4.5 years; Table 1 ).
| Patient no.: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
|---|---|---|---|---|---|---|---|---|---|
| Sex | F | F | F | F | M | F | F | M | F |
| Age (y) | 42 | 29 | 25 | 41 | 35 | 40 | 33 | 46 | 30 |
| Dysphagia | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Chest pain | N | Y | N | N | N | Y | N | Y | Y |
| Food or acid regurgitation | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Weight loss | N | N | Y | Y | Y | Y | N | N | N |
| Time from symptom occurrence to diagnosis (y) | 4.5 | 3.5 | 2 | 4.5 | 3.5 | 4 | 3 | 3.5 | 1.5 |
| LES pressure (mmHg) | 17.5 | 39.2 | 36.3 | 50.5 | 56.5 | 34.1 | 37.2 | 38.1 | 25.6 |
| LES relaxation (%) | 97 | 51 | 47 | 54 | 47 | 43 | 46 | 40 | 52 |
| Distal esophageal amplitude (mmHg) | 19 | 24 | 25 | 16 | 22 | 31 | 37 | 23 | 11 |
| Peristaltic contractions (%) | 0 | 0 | 10 | 30 | 0 | 0 | 0 | 0 | 0 |
| Complete bolus transit (%) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Heller myotomy | Y | Y | N | N | N | N | N | N | N |
F = female; LES = lower esophageal sphincter; M = male; N = no; Y = yes.
Manometric characteristics
The LES pressure during the swallows of the patients with untreated achalasia ranged from 25.6 mmHg to 56.5 mmHg (normal 10–45 mmHg). The LES relaxation percentage was abnormal (range 40–54%; normal >90%) and the pressure of distal esophageal contraction was abnormally low (range 11–37 mmHg; normal 30–180 mmHg).
In the two patients who had undergone Heller myotomy, the pressures of the LES were 17.5 mmHg and 39.2 mmHg, respectively. The LES relaxation percentages, 97% and 51%, respectively, were higher than those of the untreated patients (mean 47%). A comparison of manometric patterns in the treated and untreated patients showed similar contraction amplitudes of the distal esophagus and onset velocities of the contraction waves.
The manometric and impedance characteristics are shown in Table 1 . None of the patients in either group had manometrically normal esophageal peristalsis during saline or viscous swallowing.
Impedance characteristics
None of the patients had a normal transit for liquid, viscous, or liquid and viscous swallows. All the analyzed impedance patterns from patients with achalasia were pathological.
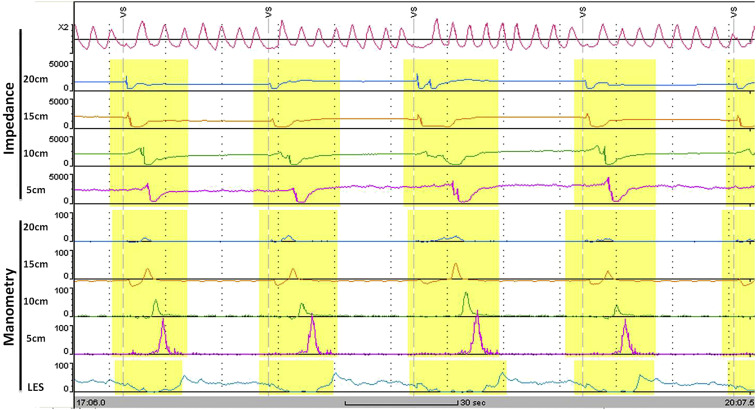
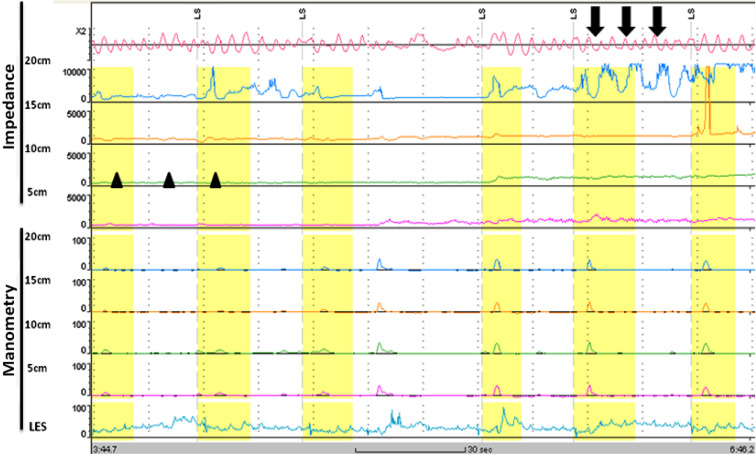
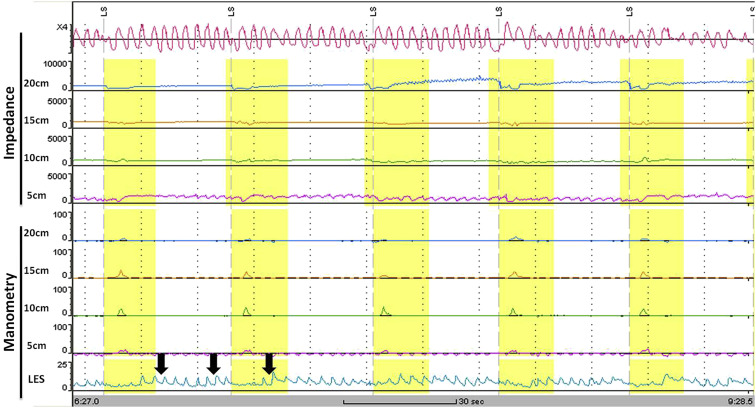
The analysis of the tracings revealed some important features. Firstly, the normal pattern of esophageal bolus transit (Fig. 2 ) was not shown in any patient. In all patients there was an absence of complete bolus transit through the esophagus with both liquid and viscous swallows. All patients had a low baseline impedance level throughout the esophagus, suggesting stasis of the ingested boluses. Secondly, some patients showed retrograde movement of esophageal content following a bolus transport. In addition, frequent abnormal movements of air within the proximal esophagus (air trapping) were found in nearly all of the patients. Fig. 3 shows an example of air trapping and low impedance baseline level. In the patients with achalasia after Heller myotomy, a low baseline impedance level and incomplete bolus transit were still seen. An example of an impedance tracing of a patient who had undergone Heller myotomy is shown in Fig. 4 .
|
|
|
Figure 2. Example of normal impedance tracing of complete bolus transit. |
|
|
|
Figure 3. Impedance of a patient with achalasia shows low baseline impedance level (arrowheads) and air trapping in the proximal esophagus (arrows). |
|
|
|
Figure 4. Impedance of a patient who underwent Heller myotomy showing low baseline impedance level, incomplete bolus transit and hypotensive lower esophageal sphincter pressure (arrows). |
Discussion
Recent studies support the concept that combined measurement of esophageal bolus transit and wave pressure provides important additional information about esophageal motility compared with conventional manometry, such as: (1) monitoring of bolus transport patterns without the use of radiation; (2) evaluation of bolus clearance; and (3) investigation of the relationships between bolus transit and LES relaxation [9] ; [10] ; [12] . We therefore evaluated the additional value of adding an MII recording to esophageal manometry in patients with achalasia using combined MII-EM. This study is the first observation study to apply MII-EM in the evaluation of esophageal dysmotility in Taiwanese patients with achalasia and in patients after myotomy.
The manometric criteria for diagnosing achalasia include aperistalsis of the distal esophageal body and incomplete or absent LES relaxation. Supportive features may include hypertensive LES and hypotensive esophageal peristalsis [13] . Furthermore, not all patients with achalasia have elevated LES pressure. In our study, only 29% of untreated patients had a hypertensive LES, whereas both patients who had undergone Heller myotomy had a normal LES resting pressure. In addition, all patients presented with incomplete LES relaxation and aperistalsis, regardless of whether they had had a myotomy.
In contrast with the uniform manometry tracings in patients with achalasia, the transport patterns showed much intra- and inter-individual variability. In previous studies, characteristics of achalasia have been described using multichannel intraluminal impedance. In a group of 350 patients, Tutuian and Castell [10] found abnormal bolus transit in all of their patients with achalasia. Nguyen et al [14] also demonstrated low baseline impedance level, regurgitation of luminal contents, and the presence of air in the proximal esophagus in patients with achalasia.
Because the intraluminal impedance is inversely related to the electrical conductivity of fluids and the cross-sectional area of the esophageal lumen, the bolus stasis of ingested materials and lumen dilatation results in a decrease in the impedance. In addition, the phenomenon of air trapping is very specific for achalasia. Previous radiological studies described air trapping as the separation of swallowed air from the bolus due to gravity prior to reaching the LES [16] ; [17] .
The explanation for the phenomenon of esophageal air trapping has been addressed by previous studies [18] ; [19] ; [20] . To spontaneously belch, there is a nearly simultaneous relaxation of both the upper and lower esophageal sphincter, immediately followed by upper esophageal sphincter relaxation, which leads to progressive air trapping and lumen dilatation inside the esophagus [18] . Second, patients with achalasia are characterized by a higher resting upper esophageal sphincter pressure [19] , which may interfere with air passage across the sphincter and thus increase the possibility of esophageal air trapping. Finally, air trapping is related to a one-way valve effect secondary to esophageal dilation and kinking behind the cricopharyngeous muscle [20] .
Therefore the main features identified as characteristics of achalasia are: (1) a lower baseline impedance value; (2) intermittent regurgitation of luminal contents; (3) disturbed esophageal bolus transit with failed bolus transport; and (4) pathological air movement within the lumen during deglutition. In our study, the impedance patterns are similar to those reported previously [14] ; [15] ; [25] . In addition, the MII findings of patients with and without Heller myotomy showed similar impedance characteristics in term of incomplete bolus transit.
Vigorous achalasia is a common variant of achalasia characterized by the presence of normal to high amplitude esophageal body contractions in the presence of a non-relaxing LES. In patients with vigorous achalasia, botulinum toxin has been reported to be effective [21] . A retrospective study subclassified achalasia into three subtypes using high-resolution manometry [22] . They were distinct in terms of their responsiveness to medical or surgical treatment. Type I (classic) patients did significantly better with Heller myotomy than with pneumatic dilatation, whereas type II patients did well with all available treatment modalities. Type III patients had a worse prognostic outcome than either of the other two subtypes [23] .
In our study, the impedance results did not show better bolus clearance in patients treated with Heller myotomy compared with untreated patients. By contrast, Tatum et al [24] described some improvement in esophageal transit and bolus clearance in patients who had undergone a Heller myotomy. The reasons for the partial normalization of esophageal function after surgical treatment still remain unclear. As our patients did not have vigorous achalasia nor were they further classified into subtypes, they could belong to the subtypes which were less responsive to treatment. The lack of improvement after Heller myotomy could also be due to our small sample size, which was not able to achieve statistically significant differences. In addition, our patients were all Taiwanese, thus ethnical difference could have contributed to our different findings.
In addition, the low baseline impedance and air trapping in the proximal esophagus may prevent this modality being able to accurately assess esophageal emptying [14] . In a study of 40 patients with non-obstructive dysphagia, Conchillo et al [25] found that most of the swallows in patients with achalasia could not be quantified due to a low pre-swallow impedance baseline and almost all swallows had abnormal transit patterns. In another study, it was shown that in patients with achalasia, low baseline impedance levels and air entrapment in the proximal esophagus limit the value of MII monitoring as a test of esophageal emptying [15] .
Furthermore, our patients were in a sitting position during saline and viscous swallowing while the impedance parameters were measured. In the dilated esophagus, the swallowed bolus could transverse to the LES mainly by means of gravity, thus no change in resistance was detected as the bolus was not in contact with the impedance measuring sites. In future studies, patients could be placed in a supine position during MII-EM to assess whether impedance characteristics differ between different positions in patients with achalsia.
In conclusion, our study suggested that patients with achalasia are characterized by abnormal LES relaxation, esophageal aperistalsis, and incomplete bolus transit, regardless of Heller myotomy. Because of low baseline impedance levels and air trapping in the proximal esophagus by impedance, the clinical utility of combined MII-EM in the diagnosis of esophageal dysmotility related to achalasia appears to be limited.
Conflicts of interest
All authors declare no conflicts of interest.
References
- [1] J.F. Mayberry; Epidemiology and demographics of achalasia; Gastrointest Endosc Clin N Am, 11 (2001), pp. 235–248
- [2] W. Park, F. Vaezi; Etiology and pathogenesis of achalasia: the current understanding; Am J Gastroenterol, 100 (2005), pp. 1404–1414
- [3] P.J. Howard, L. Maher, A. Pryde, E.W. Cameron, R.C. Heading; Five year prospective study of the incidence, clinical features, and diagnosis of achalasia in Edinburgh; Gut, 33 (1992), pp. 1011–1015
- [4] I. Hirano, R.P. Tatum, G. Shi, Q. Sang, R.J. Joehl, P.J. Kahrilas; Manometric heterogeneity in patients with idiopathic achalasia; Gastroenterology, 120 (2001), pp. 789–798
- [5] P. Funch-Jensen, K. Aksglaede, P. Thommesen; A new method for the detection of incomplete lower esophageal sphincter relaxation in patients with achalasia; Scand J Gastroenterol, 35 (2000), pp. 349–352
- [6] J. Silny; Intraluminal multiple electric impedance procedure for measurement of gastrointestinal motility; J Gastrointest Motil, 3 (1991), pp. 151–162
- [7] M. Simren, J. Silny, R. Holloway, J. Tack, J. Janssens, D. Sifrim; Relevance of ineffective esophageal motility during oesophageal acid clearance; Gut, 52 (2003), pp. 784–790
- [8] J. Silny, K. Knigge, J. Fass, G. Rau, S. Matern, V. Schumpelick; Verification of the intraluminal multiple electrical impedance measurement for the recording of gastrointestinal motility; J Gastrointest Motil, 5 (1993), pp. 107–122
- [9] R. Tutuian, D.O. Castell; Clarification of the esophageal function testing in patients with manometric ineffective esophageal motility: studies using combined impedance-manometry; Clin Gastroenterol Hepatol, 2 (2004), pp. 230–236
- [10] R. Tutuian, D.O. Castell; Combined multichannel intraluminal impedance and manometry clarifies esophageal function abnormalities: study in 350 patients; Am J Gastroenterol, 5 (2004), pp. 1011–1019
- [11] S.J. Spechler, D.O. Castell; Classification of oesophageal motility abnormalities; Gut, 49 (2001), pp. 145–151
- [12] E. Savarino, R. Tutuian; Combined multichannel intraluminal impedance and manometry testing; Dig Liver Dis, 40 (2008), pp. 167–173
- [13] M.F. Vaezi, J.E. Richter; Diagnosis and management of achalasia; Am J Gastroenterol, 94 (1999), pp. 3406–3412
- [14] H.N. Nguyen, G.R. Domingues, R. Winograd, F. Lammert, J. Silny, S. Matern; Impedance characteristics of esophageal motor function in achalasia; Dis Esophagus, 17 (2004), pp. 44–50
- [15] J.M. Conchillo, M. Selimah, A.J. Bredenoord, M. Samsom, A.J. Smout; Assessment of esophageal emptying in achalasia patients by intraluminal impedance monitoring; Neurogastroenterol Motil, 18 (2006), pp. 971–977
- [16] D.J. Ott, J.E. Richter, Y.M. Chen, W.C. Wu, D.W. Gelfand, D.O. Castell; Esophageal radiography and manometry: correlation in 172 patients with dysphagia; AJR, 149 (1987), pp. 307–311
- [17] E.T. Stewart; Radiologic evaluation of the esophagus and its motor disorders; Med Clin North Am, 65 (1981), pp. 1173–1194
- [18] B.T. Massey, W.J. Hogan, W.J. Dodds, R.O. Dantas; Alteration of the upper esophageal sphincter belch reflex in patients with achalasia; Gastroenterology, 103 (1992), pp. 1574–1579
- [19] F. Yoneyama, M. Miyachi, Y. Nimura; Manometric findings of the upper esophageal sphincter in esophageal achalasia; World J Surg, 22 (1998), pp. 1043–1047
- [20] E. Arcos, C. Medina, F. Mearin, J. Larish, L. Guarner, J.R. Malagelada; Achalasia presenting as acute airway obstruction; Dig Dis Sci, 45 (2000), pp. 2079–2083
- [21] P.J. Pasricha, R. Rai, W.J. Ravich, T.R. Hendrix, A.N. Kalloo; Botulinum toxin for achalasia: long-term outcome and predictors of response; Gastroenterology, 110 (1996), pp. 1410–1415
- [22] J.E. Pandolfino, M.A. Kwiatek, T. Nealis, W. Bulsiewicz, J. Post, P.J. Kahrilas; Achalasia: a new clinically relevant classification by high-resolution manometry; Gastroenterology, 135 (2008), pp. 1526–1533
- [23] J.O. Clarke, J.E. Pandolfino; Esophageal motor disorders: how to bridge the gap between advanced diagnostic tools and paucity of therapeutic modalities?; J Clin Gastroenterol, 46 (2012), pp. 442–448
- [24] R.P. Tatum, J.A. Wong, E.J. Figueredo, V. Martin, B.K. Oelschlager; Return of esophageal function after treatment for achalasia as determined by impedance-manometry; J Gastrointest Surg, 11 (2007), pp. 1403–1409
- [25] J.M. Conchillo, N.Q. Nguyen, M. Samsom, R.H. Holloway, A.J. Smout; Multichannel intraluminal impedance monitoring in the evaluation of patients with non-obstructive dysphagia; Am J Gastroenterol, 100 (2005), pp. 2624–2632
Document information
Published on 15/05/17
Submitted on 15/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?