Abstract
Background
A significant proportion of patients admitted for acute decompensated heart failure (ADHF) that undergo volume reduction therapy are discharged with unchanged or increased bodyweight suggesting that the endpoints for these therapies are not optimally defined. We aimed to identify vectors that can help monitor changes in intravascular fluid volume, that in turn may more accurately guide volume reduction therapy.
Methods
Data from six different impedance vectors and corresponding changes in intravascular volume derived from changes in hematocrit were obtained from 132 clinical congestion events in 56 unique patients enrolled in a multisite trial of early detection of clinical congestion events (DEFEAT PE). Mixed effects regression models were used to determine the relation between changes in impedance derived from six different vectors and changes in intravascular plasma volume.
Results
Changes in impedance were negatively associated with changes in plasma volume. Two vectors, the right atrial ring to left ventricular ring and the left ventricular ring to the right ventricular ring, were most closely associated with changes in intravascular plasma volume.
Conclusion
Impedance vectors derived from a multivector monitoring system reflect changes in intravascular plasma volume. Two of these vectors most closely track changes in plasma volume and may be used to more accurately guide and optimize volume reduction therapy.
Keywords
Heart failure;Impedance;Diuretic therapy;Volume reduction therapy
1. Introduction
Heart failure (HF) is a leading cause of morbidity and mortality in the US and significantly contributes to national health care costs [1]. Due to the progressive and unstable natural history of HF, many patients develop recurrent episodes of acute decompensated heart failure (ADHF) requiring frequent hospital admissions [2]. The health care costs associated with heart failure are mainly due to these events and are expected to increase by 120% by 2030 [1]. ADHF and refractory congestive heart failure (CHF) are considered the end product of a vicious cycle of reduced cardiac output, impaired salt and water renal excretion and consequent neurohormonal activation. It often results in volume overload, body water redistribution and concomitant high cardiac filling pressures [3]. In the US, approximately 90% of all heart failure related admissions are due to symptoms of pulmonary congestion and fluid overload that are in turn associated with HF progression and increased mortality [4]. Estimation of total body water and redistribution of extra and intravascular volumes is crucial for assessment and treatment of these patients and has been the focus of research and care innovations [5] ; [6].
Although intrathoracic impedance monitoring has been extensively studied as a means for early detection of clinical congestion events resulting from the above mechanisms, there has been little attention given to its role in monitoring or guiding volume reduction therapies.Many implantable defibrillator systems and biventricular pacing devices have incorporated intrathoracic impedance measurement in their monitoring capabilities [7]; [8]; [9] ; [10]. Therefore, a large number of patients with ADHF could benefit from more accurate direction of volume reduction therapies. Strategies to effectively use this information should be developed.
It has been reported in the literature that as many as 50% of ADHF admitted patients treated with diuretics are discharged with unchanged or increased body weight compared to admission despite clinical improvement [11]. This suggests that clinical endpoints used to direct and terminate diuretic therapy might be inappropriate. One explanation for this observation is that effective volume reduction therapy ideally targets decreased extravascular volume without significant reduction in intravascular volume that elicits activation of neurohormonal mechanisms that promote fluid retention and a net increase in total body water. Therefore, effective volume monitoring must reflect not only total intrathoracic volume but must isolate the intravascular from the extravascular fluid compartment [12].
Recently developed volume monitoring algorithms have used the multiple impedance vectors that can be derived across different leads of pacing systems in an effort to more accurately detect volume changes [13]. It is possible that among these, specific vectors or their combinations more accurately report changes in plasma volume rather than extravascular or total intrathoracic fluid volume.
We therefore aimed to examine the relationship between different impedance vectors to changes in plasma volume and thus identify vectors that most accurately report changes in the intravascular compartment. Identification of vectors that specifically isolate the intravascular volume compartment will contribute to significant advances in the direction of volume reduction therapies and address an important unmet need in the treatment of acute decompensated heart failure.
2. Methods
2.1. Patient sample
The study sample was derived from participants in the pivotal trial (DEFEAT PE; ClinicalTrials.gov, NCT00916929) of a multivector algorithm for prevention of clinical volume overload events sponsored by St. Jude Medical. All subjects had heart failure with standard indications for either an ICD or CRTD device and were required to have at least one episode of decompensated congestive heart failure within six months of study enrollment. The participants in this sub-study were those who were admitted to a hospital for decompensated heart failure events for whom serial hematocrit data were available along with impedance vector data encompassing the time period of volume reduction therapy. Patients who received blood transfusions, had episodes of active bleeding during an admission or who had a history of a lead dislodgment were excluded from the analysis.
2.2. Estimation of percent plasma volume change
Percent change in plasma volume (ppchange) at any two points during a hospitalization was determined by using a formula that has been successfully used in clinical hemodialysis and ultrafiltration [14].
|
|
Where Hctpre is the first hematocrit measurement and Hctpost is a subsequent measure.
For this study, we chose two hematocrit values that were separated by the greatest time interval during volume reduction therapy.
Positive values of percent plasma volume change represent an increase in plasma volume from the time when the first hematocrit sample was drawn to the time of the repeated measurement.
2.3. Analysis of impedance vectors
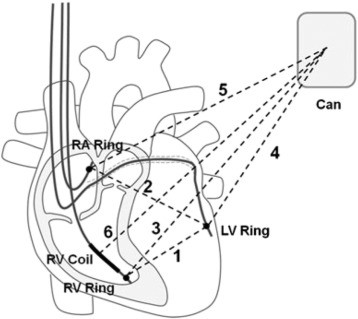
The CRTD device measures impedances obtained from six independent vectors formed by the following electrode configurations (Fig. 1): (1) LVr–RVr, LV ring electrode to RV ring electrode;(2) LVr–RAr, LV ring electrode to right atrium (RA) ring electrode; (3) RVr–Can, RVring electrode to device can;(4) LVr–Can, LV ring electrode to device can; (5) RAr–Can,RA ring electrode to device can; and (6) RVc–Can, RV coil electrode to device can. Impedance measurements were recorded every 2 h[15]. The ICD device only measures impedance vectors for configurations: (3) RVr–Can, RVring electrode to device can, (5) RAr–Can,RA ring electrode to device can; and (6) RVc-Can, RV coil electrode to device can.
|
|
|
Fig. 1. Diagram of CRDT impedance vectors. Electrode configurations used for measuring impedance: (1) LVr–RVr, (2) LVr–RAr, (3) RVr–can, (4) LVr–can, (5) RAr–can, and (6) RVc–can. LV: left ventricle, RA: right atrium, RV: right ventricle, c: coil electrode, r: ring electrode. Modified from Binkley et al. [15]. |
Impedance data from each vector obtained at times that most closely corresponded to acquisition of the hematocrits were selected for analysis. Therefore impedance measurements corresponding to the first and second of two hematocrit measures were analyzed for each of the six vectors. If the available impedance data were more than 2 h from a corresponding hematocrit, the observation was not included in the analysis. The change in impedance for each vector was calculated by subtracting the post-hematocrit related impedance (PostImp) from the pre-hematocrit related impedance (PreImp) values as follows:
|
|
Therefore, positive values of impedance difference represent an increase in impedance overtime, while negative values represent a decrease of impedance.
2.4. Statistical analysis
Changes in impedance and percent plasma volume were represented by the mean and standard deviation. The association between changes in impedance and plasma volume was analyzed using mixed linear models to account for inter-subject variability in measures and the different number of measurements made on each participant. Sequential models were constructed consisting of the mixed effects model with random intercept (defined by the patient identification number) and the mixed effecfs model with both random intercept and coefficient relating change in impedance and change in plasma volume. Models were constructed for each of the six impedance vectors. The likelihood ratio test was used to determine whether each step in the modeling process significantly improved the model compared to the previous step. Statistical significance was set at the 0.05 level for all analyses.
The Akaike information criterion (AIC) for non-nested models was used to determine which of the regression models showed the best fit between changes in impedance measures and change in percent plasma volume. All analyses were performed using Stata 13®.
3. Results
Clinical characteristics of the study participants are shown in Table 1.
| Demographic variable | Number of patients (N = 132) |
|---|---|
| Age (years) | |
| Mean ± SD | 66 ± 14 |
| Range | (26, 88) |
| Gender, n (%) | |
| Female | 27 (20.5%) |
| Male | 105 (79.5%) |
| Race/ethnicity, n (%) | |
| Black or African American | 61 (46.2%) |
| Hispanic or Latino | 10 (7.6%) |
| White | 61 (46.2%) |
| NYHA class, n (%) | |
| I | 9 (6.8%) |
| II | 53 (40.2%) |
| III | 70 (53.0%) |
| LV ejection fraction (%) | |
| Mean ± SD | 25 ± 8 |
| Range | (10, 50) |
| Cardiomyopathy classification | |
| Ischemic, n (%) | 77 (58.3%) |
| Non-ischemic, n (%) | 55 (41.7%) |
| Cardiovascular history | |
| Coronary artery disease | 89 (67.4%) |
| Myocardial infarction | 72 (54.5%) |
| CABG | 53 (40.2%) |
| PTCA | 34 (25.8%) |
| Unstable angina | 40 (30.3%) |
| Arrhythmia history | |
| Ventricular | 71 (53.8%) |
| Non-ventricular | 69 (52.3%) |
| Pulmonary medical conditions | |
| Asthma | 12 (9.1%) |
| COPD | 42 (31.8%) |
| Pulmonary fibrosis | 2 (1.5%) |
| Pulmonary hypertension | 39 (29.5%) |
| Sarcoidosis | 6 (4.5%) |
| Sleep apnea | 15 (11.4%) |
| Partial left lower lobe resection | 1 (0.8%) |
| Pulmonary embolism | 3 (2.3%) |
| Cardiac medications at enrollment | |
| ACE inhibitors | 79 (59.8%) |
| Angiotensin receptor blockers (ARB) | 17 (12.9%) |
| Beta-blockers | 115 (87.1%) |
| Diuretics | 124 (93.9%) |
Hematocrits and corresponding impedance data were available for 136 congestion events on 59 unique admitted patients. Single observations from four of these patients were eliminated owing to requirement for blood transfusions (n: 3) or pacemaker lead fracture (n: 1). Accordingly, we analyzed data from 132 observations obtained on 56 unique patients. Twenty four patients had data available for only one admission, while the remainder had multiple admissions ranging from 2 to 11. Fifty seven percent of the patients (n: 32) had a CRDT device and forty-three percent had an ICD device only. Of all observations, there was an estimated 65% incidence of increase in plasma volume during the treatment period. The distribution of the number of hospital days over which hematocrit and impedance measures were obtained was positively skewed.The median number of days was 4 with an interquartile range of 2 to 7 days.There was no significant difference (p = 0.7) in the number of hospital days for those with no change or a decrease in plasma volume (4 days; interquartile range 2 to 6 days)and those with an increase in plasma volume (4 days; interquartile range 2 to 7 days).
The mean and standard deviations for the change in plasma volume and corresponding changes in the six impedance vectors for the entire group are shown in Table 2. The average changes in impedance for the six vectors in the group with an increase in estimated plasma volume and that with a maintained or decrease are shown on the right hand of Table 2. There was a decrease in mean impedance change for five of the six vectors in congestion events with an increase in percent plasma volume and an increase in the mean impedance change for all vectors in the group with a decrease in plasma volume. Therefore, other than the LV ring to can vector in the group with an increase in plasma volume, all impedance vectors exhibited the changes expected based on the known relation between electrical impedance and fluid filled media.
| Variables | Entire group | > 0 ppchange (n:86) | ≤ 0 ppchange (n:46) | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| Estimated percent plasma change | ||||||
| Plasma change (%) | 7.13 | 16.12 | 15.73 | 12.69 | − 8.93 | 6.83 |
| Impedance vector differences | ||||||
| LVr to RVra (Ω) | 2.92 | 31.40 | − 0.68 | 32.13 | 11.63 | 29.10 |
| RAr to LVra (Ω) | − 1.23 | 32.05 | − 5.55 | 32.48 | 9.34 | 29.75 |
| RVr to can (Ω) | 1.70 | 15.28 | − 2.50 | 13.46 | 9.51 | 15.71 |
| LVr to cana (Ω) | 4.91 | 29.86 | 4.68 | 33.42 | 5.91 | 20.37 |
| RAr to can (Ω) | − 0.90 | 19.56 | − 6.16 | 17.94 | 8.66 | 19.23 |
| RVc to can (Ω) | 1.35 | 6.40 | − 0.12 | 5.28 | 4.12 | 7.45 |
Estimated percent change in plasma volume (ppchange) = (100 / (100 − Hctpre)) ∗ (100(Hctpre − Hctpost) / Hctpost).
Impedance vector differences were calculated by the subtraction of the post-value–pre-value.
a. These impedance vectors do not exist for ICD devices, and thus were not available for all observations.
3.1. Mixed effects models
The log likelihood and the overall model significance of the random intercept and the random intercept plus random slope models for each of the six vectors are shown in Table 3.
| I. Vector | Random intercept models | Random intercept + slope models | LR testa | ||||
|---|---|---|---|---|---|---|---|
| Log LH | p-value | Coef | Log LH | p-value | Coef | p-value | |
| LVr to RVr | − 339.5 | 0.0023 | − 0.168 | − 335.9 | 0.0034 | − 0.253 | 0.0246 |
| RAr to LVr | − 337.9 | 0.0002 | − 0.194 | − 332.0 | 0.0013 | − 0.299 | 0.0085 |
| RVr to can | − 533.0 | < 0.0001 | − 0.488 | − 523.7 | < 0.0001 | − 0.602 | < 0.0001 |
| LVr to can | − 342.4 | 0.0662 | − 0.108 | − 337.3 | 0.0336 | − 0.227 | 0.0132 |
| RAr to can | − 429.2 | < 0.0001 | − 0.469 | − 419.8 | < 0.0001 | − 0.586 | < 0.0001 |
| RVc to can | − 5435.3 | < 0.0001 | − 1.05 | − 530.7 | < 0.0001 | − 1.08 | 0.0132 |
Coef: fixed effects impedance vector slope coefficient.
Random intercept model: fixed effects + random intercept (patient ID).
Random intercept + slope model: fixed effects + random intercept + random slope.
a. LR test: likelihood ratio test, compares random slope model with the random intercept model.
Five of the six impedance models showed a significant relationship with percent change in plasma volume using a fixed effects only model. Although the LV ring to can vector did not show a significant relation with change in plasma volume in a fixed effect model, it and the remaining five vectors showed a progressive increase in model significance with addition of random intercept and random slope models. For each of the six vectors, hierarchical likelihood ratio tests showed an increase in model significance with the addition of a random slope to the random intercept model. In all vectors, the fixed effect coefficient became increasingly negative with the addition of the random slope. Therefore, when correcting for inter-subject variability, the direct inverse relation between change in impedance and change in percent plasma volume is strengthened for each of the vectors.
The impedance vector with the best model fit to the percent change in plasma volume was identified using the Akaike information criterion (AIC). This statistic is useful in comparing models that are not hierarchical, such as the different regression models for the different impedances, and is based on model log likelihood with a penalty for number of variables contained in the model. Because the models with the random intercept and slope were the most significant for each vector, these models were used for comparison using the AIC. The AIC for all models is shown in Table 4. The smaller the AIC, the better the model fit.
| Impedance vector models | AIC |
|---|---|
| RAr to LVr | 677.3251 |
| LVr to RVr | 683.36 |
| LVr to can | 686.77 |
| RAr to can | 851.61 |
| RVr to can | 1059.554 |
| RVc to can | 1073.491 |
Based on the AIC, the vector defined by the RAr to LVr had the best fit with change in percent plasma volume.
4. Discussion
This study provides foundation data for the use of intrathoracic impedance monitoring as a novel and effective means to guide volume reduction therapy. Given the absence of proven clinical endpoints for the administration of volume reduction therapies, the findings address a significant unmet need in the clinical management of acute decompensated heart failure.
All six of the intrathoracic impedance vectors showed the expected inverse relationship between change in impedance and percent change in plasma volume in the mixed effects models. The inverse relationship between impedance vectors and percent change in plasma volume is consistent with the known increase in electrical conductivity through fluid filled media. To the authors' knowledge, this is the first in vivo demonstration in humans that intrathoracic impedance indeed follows the expected inverse relation. Furthermore, this study identifies impedance vectors that are most closely associated with changes in plasma volume and therefore may be the most informative vectors for monitoring volume reduction therapy.
The ability to specifically isolate changes in intravascular volume is of importance when considering the pathophysiology and treatment of congestion states [6]. It has been noted that the goal in congestion therapy is reduction of interstitial fluid volume with preservation of intravascular volume [3]; [4]; [6] ; [12]. When treatment for congestion states results in marked reduction of intravascular fluid volume, an iatrogenic cardiorenal syndrome can result with activation of the renin–angiotensin axis and other systems that promote fluid retention rather than volume loss. Indeed, over half of the instances of volume reduction therapy in our study were accompanied by an estimated increase rather than decrease in percent plasma volume. This is consistent with findings in the Acute Decompensated Heart Failure National Registry (ADHERE) in which 50% of patients were discharged from the hospital with either no change or an increase in weight following volume reduction therapy [4].
Two vectors, RA ring to LV ring and RV ring to LV ring, were found to be most closely associated with percent change in plasma volume. It is not surprising that these two vectors would isolate changes in the intravascular volume compartment based on their spatial and anatomic orientation (Fig. 1). These vectors traverse cardiac chambers and exclude interstitial fluid compartments. In contrast, vectors such as those including the pacer can will encompass the interstitial fluid compartments in the lungs and chest wall as well as the fluid volume contained in the cardiac chambers. Indeed, the LV ring to can vector did not show a significant correlation with change in plasma volume in the fixed effect and random intercepts models.In general, the vectors including the pacer can showed the weakest association with change in plasma volume in the random effects models. This weaker association is likely due to the influence of these vectors by multiple fluid compartments beyond the intravascular compartment alone.
The random slope models were superior to the random intercept only models since they account for an additional component of the expected intersubject variability in anatomy and orientation of pacemaker leads. That the random slope for the relationship between changes in plasma volume and impedance added significantly to the model indicates that the regression lines relating these measures for the different subjects are not parallel. It would in fact be surprising if the regression lines were parallel considering anatomic variation and orientation of pacemaker leads between subjects. Accounting for the random effects due to intersubject variability resulted in an even greater negative fixed effect coefficient, which represents the group mean of the relation between change in plasma volume and impedance. This further supports the inverse relation between changes in plasma volume and impedance.
4.1. Implications for treatment of congestion states
The capacity to isolate intravascular from extravascular volume compartments provides a unique opportunity to more effectively treat clinical congestion states. As noted above, overly aggressive congestion therapy can produce decreases in intravascular volume that induce a cardio-renal syndrome. As a result, total body water will increase rather than decrease. Optimal management of clinical congestion states requires an understanding of changes in intravascular volume and the delicate balance between excess intravascular volume and deleterious volume reductions in this compartment. Studies of ultrafiltration therapy have indicated that volume reduction therapy is ideally monitored by the capillary refill rate [12]. This measurement is determined by the net change in intravascular volume resulting from movement of extravascular fluid to the intravascular compartment and elimination of excess intravascular fluid by renal excretion. Ideally, this rate should maintain the net change in intravascular volume at zero. If this rate results in a decrease in intravascular volume, activation of neurohormonal mechanisms that promote increases in total body water may result. In fact, the failure to accurately monitor changes in intravascular volume may account for the maintenance or increase in weight noted in the ADHERE registry and the increases in intravascular volume noted in a subset of patients in the current study. The use of impedance vectors that most closely reflect changes in intravascular volume can prevent excessive volume reduction therapy. A small preliminary study has investigated the use of impedance monitoring to ascertain readiness for hospital discharge [16]. However, that study did not select an impedance vector that most accurately reflected changes in plasma volume for therapeutic monitoring.
Therefore, strategies to monitor volume reduction therapy for acute decompensated heart failure could consist of tracking impedance changes reported by the vectors that most accurately reflect changes in intravascular plasma volume. A persistent increase in impedance in these vectors, indicating a decreased intravascular volume, would signal the termination of volume reduction therapies.This would prevent excessive intravascular volume contraction and the stimulation of mechanisms that in fact promote volume retention. Conversely, persistent decreases in impedance in vectors most closely correlated with changes in plasma volume can indicate significant volume shifts not reflected by changes in body weight that may require initiation or resumption of volume reduction therapies [6]. Although not specifically tested in the current study, differential changes in impedance measured by those vectors closely correlated with intravascular volume alone and those that are reflective of total intrathoracic volume could provide important information regarding the relative changes in intravascular and extravascular volume compartments and further guide the course of volume reduction therapy.
4.2. Study limitations
These data were obtained from a subset of patients in a larger pivotal trial. Only patients who had a congestion event were included since hemoglobin and hematocrit data were available only in those subjects. Although it is plausible that the same relationships between change in impedance and intravascular volume would pertain in other study participants, this cannot be determined with certainty in the current analysis.
Changes in hematocrit constitute only an indirect measure of plasma volume. However, this measure has been used in numerous prior studies. We specifically excluded patients with acute bleeding or those who obtained blood transfusions that would result in rapid changes in hematocrit. This increases confidence that the hematocrit changes measured in the current study were indeed due to changes in intravascular volume.
The strongest correlations with changes in plasma volume were obtained with impedance vectors available in biventricular pacing systems. Therefore, the findings of this study may not be readily applied to those with non-CRT systems. A relatively strong relation between change in impedance and plasma volume was observed with the RAr to can vector suggesting that intravascular volume monitoring may still be possible in patients with devices having atrio-ventricular pacing as well as defibrillating capabilities. However, it is estimated that over 500,000 patients in the US have biventricular pacing devices (personal communication Hans Schafer, St. Jude Medical). Accordingly a significant proportion of patients with heart failure could benefit from this strategy for guiding volume reduction therapies.
4.3. Conclusions
It has been noted that monitoring of body weight may be misleading in either the detection of congestion or monitoring response to therapy since congestion may be due to volume compartment shifts rather than changes in absolute body water. Accordingly, the use of intrathoracic impedance vectors that accurately reflect changes in intravascular volume can significantly advance the guidance of volume reduction therapies with improved outcomes including the need for early hospital readmission. The current findings provide the foundation for future studies investigating the use of impedance vectors that most closely reflect changes in intravascular volume to accurately guide volume reduction therapy and advance the management of acute decompensated heart failure.
Conflict of interest
Dr. Heist has served as a consultant and has received research support and honoraria from both St. Jude Medical (<$10,000) and Biotronik (<$10,000). He has also served as a consultant and received honoraria for Boston Scientific and has received research support from Sorin (<$10,000). Dr. Pavri has received speaker honoraria from St. Jude Medical (>$10,000) and Medtronic (<$10,000). Dr. Van Bakel has received research funding from Medtronic. Dr. Herre has received research funding from St. Jude Medical. Dr. Binkley reports to have served as a consultant to St. Jude Medical (<$10,000). Dr. Qu is a research scientist for St. Jude Medical. Dr. Lara has no conflicts to report.
Acknowledgments
This work is a sub-study of the pivotal trial DEFEAT PE (ClinicalTrials.gov, NCT00916929), funded by St. Jude Medical.
References
- [1] A.S. Go, D. Mozaffarian, V.L. Roger, E.J. Benjamin, J.D. Berry, W.B. Borden, et al.; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2013 update: a report from the American Heart Association; Circulation, 127 (2013), pp. e6–e245
- [2] M.M. Givertz, J.R. Teerlink, N.M. Albert, C. Westlake Canary, S.P. Collins, Colvin-Adams, et al.; Acute decompensated heart failure: update on new and emerging evidence and directions for future research; J Card Fail, 19 (2013), pp. 371–389
- [3] I.S. Anand, R. Ferrari, G.S. Kalra, P.L. Wahi, P.A. Poole-Wilson, H. Pc; Edema of cardiac origin: studies of body water and sodium, renal function, hemodynamic indexes, and plasma hormones in untreated congestive cardiac failure; Circulation, 80 (1989), pp. 299–305
- [4] K.F. Adams Jr., G.C. Fonarow, C.L. Emerman, T.H. LeJemtel, M.R. Costanzo, W.T. Abraham, et al.; Characteristics and outcomes of patients hospitalized for heart failure in the United States: rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE); Am Heart J, 149 (2005), pp. 209–216
- [5] S. Di Somma, S. Navarin, S. Giordano, F. Spadini, G. Lippi, G. Cervellin, et al.; The emerging role of biomarkers and bio-impedance in evaluating hydration status in patients with acute heart failure; Clin Chem Lab Med, 50 (2012), pp. 2093–2105
- [6] M.E. Dunlap, P.a. Sobotka; Fluid re-distribution rather than accumulation causes most cases of decompensated heart failure; J Am Coll Cardiol, 62 (2013), pp. 165–166
- [7] C.M. Yu, L. Wang, E. Chau, R.H. Chan, S.L. Kong, M.O. Tang, et al.; Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization; Circulation, 112 (2005), pp. 841–848
- [8] W.T. Abraham, S. Compton, G. Haas, B. Foreman, R.C. Canby, R. Fishel, et al.; Intrathoracic impedance vs daily weight monitoring for predicting worsening heart failure events: results of the Fluid Accumulation Status Trial (FAST); Congest Heart Fail, 17 (2011), pp. 51–55
- [9] D.J. Van Veldhuisen, F. Braunschweig, V. Conraads, I. Ford, M.R. Cowie, G. Jondeau, et al.; Intrathoracic impedance monitoring, audible patient alerts, and outcome in patients with heart failure; Circulation, 124 (2011), pp. 1719–1726
- [10] V.M. Conraads, L. Tavazzi, M. Santini, F. Oliva, B. Gerritse, C.M. Yu, et al.; Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: the SENSE-HF trial; Eur Heart J, 32 (2011), pp. 2266–2273
- [11] G.C. Fonarow, ADHERE Scientific Advisory Committee; The Acute Decompensated Heart Failure National Registry (ADHERE): opportunities to improve care of patients hospitalized with acute decompensated heart failure; Rev Cardiovasc Med, 4 (Suppl. 7) (2003), pp. S21–S30
- [12] A. Boyle, P.a. Sobotka; Redefining the therapeutic objective in decompensated heart failure: hemoconcentration as a surrogate for plasma refill rate; J Card Fail, 12 (2006), pp. 247–249
- [13] D. Panescu, M. Naware, J. Siou, Y. Nabutovsky, N. Holmstrom, A. Blomqvist, et al.; Usefulness of monitoring congestive heart failure by multiple impedance vectors; Conf Proc IEEE Eng Med Biol Soc, 2008 (2008), pp. 5668–5670
- [14] W. Van Beaumont; Evaluation of hemoconcentration from hematocrit measurements; J Appl Physiol, 32 (1972), pp. 712–713
- [15] P.F. Binkley, J.G. Porterfield, L.M. Porterfield, S.L. Beau, R. Corbisiero, G.S. Greer, et al.; Feasibility of using multivector impedance to monitor pulmonary congestion in heart failure patients; J Interv Card Electrophysiol, 35 (2012), pp. 197–206
- [16] D.J. Whellan, C.J. Droogan, J. Fitzpatrick, S. Adams, M.M. McCarey, J. Andrel, et al.; Change in intrathoracic impedance measures during acute decompensated heart failure admission: results from the Diagnostic Data for Discharge in Heart Failure Patients (3D-HF) Pilot Study; J Card Fail, 18 (2012), pp. 107–112
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?