Abstract
The authors report a survey carried out in a cattle farm between January and April 2009 in Mateur region (Northern Tunisia). Seroprevalence by Microscopic Agglutination Test (MAT) was estimated to 81.4 ± 6% and 35 ± 2% in cows and calves, respectively. Seropositivity to more than one serovar was noticed in 91% (81/89) of infected animals. The examination of the distribution pattern of Leptospira serovars involved in this outbreak indicates that serovar Pomona was the predominant one (75.3%), followed by Autumnalis (59.5%), Bim (58.4%) and Munchen (55%). High titres (between 400 and 6400) were found in 68.7% of the tested animals and were correlated with clinical onset of leptospirosis. Leptospirosis is an underestimated pathogen in Tunisia; further investigations are needed to study the epidemiology both in man and animals and to implement effective control measures.
Introduction
Leptospirosis (Leptospira spp. infection) is a cosmopolitan spirochetal disease of humans and animal occurring especially in tropical and subtropical regions (Desvars et al. 2011). Despite the progress in molecular taxonomy, the subdivision of the genus in to serogroups and serovars remains widely used (Picardeau 2013). Each serovar has its own distribution area and its own host maintenance species. The prevalence of infection of rodents, which are the main reservoirs of leptospires, varies between 10.5 and 90.5% in Japan (Koizumi et al. 2008) and USA (Vinetz et al. 1996) respectively.
In cattle, leptospirosis induces decreased milk yield and reproductive failure due to infertility, abortions and stillbirths (Thiermann, 1984). The most important serovars, affecting cattle is L. Hardjo followed by Pomona (Bharti et al. 2003; Grooms 2006; Salgado et al. 2014). In Morocco, a survey carried out in North West regions reveals that 15% of cattle were seropositive to serovars Ballum, Sejroe and Australis (Benkirane et al. 2014). In Egypt the seroprevalence of bovine leptospirosis is estimated to be between 37.6 and 40% (Horton et al. 2014).
In Tunisia, the only study concerning bovine leptospirosis was in the Kef region (Northwest Tunisia) and was undertaken more than half a century ago by Durand & Loquerie (1960). The authors reported two clinical cases in a herd of 40 cattle. Fever, icterus and redwater were the main clinical signs exhibited by affected animals. Laboratory examination using culture and guinea-pigs inoculation confirmed Leptospira spp. infection.
In sheep, the seroprevalence in Central Tunisia has been estimated to be 25% (Khamassi Khbou et al. 2010). Serovar Copenhageni was present in 98% of sheep. As far as the authors know, there are no recent studies of the prevalence of leptospirosis in both humans and domestic animals in Algeria, Tunisia and Libya.
The Microscopic Agglutination Test (MAT) is the reference serological test for Leptospira diagnosis [Office International des Epizooties (OIE) 2014]. To be most relevant, the MAT should be performed using local live Leptospira strains; because the results are interfered with by occurrence of cross-reactivity between serovars. However, according to Levett (2003) and Blanco et al. (2016) the highest titres in MAT correlate to the cultured serogroup, making this test a good indicator of the infecting serovars.
In this study we performed a cross-sectional serological survey in a cattle farm in Northern Tunisia where an outbreak of leptospirosis occurred in 2009. The authors examine distribution pattern of Leptospira serovars and the multiple serovars cross-reactions.
Materials and methods
Animals
This study was carried out in an intensive dairy farm located in Mateur, Governorate of Bizerte (Northern Tunisia). Mateur (longitude 9.6 E; latitude 37.0 N, altitude 41 m asl) is a sub-humid region with an annual mean rainfall of 539 mm. The mean temperature in winter and summer are 6 and 33°C, respectively.
The farm consisted of approximately 1000 cattle heads lodging in 5 units of about 200 animals; the distance between the units is 60 to 350 metres. The cows are lodged in free-stalls while calves lived in individual pens.
The occurrence of fever, icterus, redwater and mortality in calves and cows led to the suspicion of leptospirosis in this herd. In addition, recurrent milk yield decrease and reproductive problems were reported in this farm, i.e. low fertility, abortions and stillbirths. A representative number (10%) of the cattle population was randomly sampled from 4 units. Sera samples were collected in sterile vacutainer tubes from 102 cows (aged between 2 and 8 years) and 17 calves (aged between 2 and 12 months).
Serology
The sera were tested for Leptospira spp. antibodies using the standard microscopic agglutination test (MAT) at 1/100 cut-off value (Faine et al. 1999). A titre equal or above 1/400 was considered to be high and correlated with acute clinical leptospirosis (André-Fontaine 2016). Briefly, panels of fourteen live reference strains of leptospires (Laboratory of Leptospires Diagnosis, National Veterinary School of Lyon, France) in exponential stage of growth were used as serogroups and serovars standards. Sera samples were serially diluted in phosphate-buffered saline (PBS) at 2-fold dilutions, from 1:50 to 1:6400 and mixed with the same volume of leptospires suspension. Two hours after incubation at 30°C, the plates were read under a dark-field microscope at 250× magnification. The MAT titre corresponds to the reciprocal of the highest dilution of the serum in which more than 50% of the leptospires agglutinated. Positive and negative standard sera and one antigen control were used in each run.
Nine tested serogroups consists of more than one serovars, only Pomona is single serovar in its serogroup.
Statistical analysis
Chi square test was performed with EpiInfo 2000 (CDC, Atlanta, USA) at 5% threshold. Principal components analysis (PCA) was performed with XLstat software for windows (Microsoft ©, WA, USA).
Results
Seroprevalences
The overall seroprevalence of anti-Leptospira spp. antibodies in cattle was 74.8 ± 7.8%. The seroprevalences were 81.4% (83/102) and 35% (6/17) in cows and calves, respectively (P < 0.0001) (Table 1).
| Cows | Calves | Overall | ||||
|---|---|---|---|---|---|---|
| Cattle unit | Total number | Positive/tested | Total number | Positive/tested | Total number | Positive/tested (%± S.E.) |
| 1 | 198 | 24/32 | 39 | 0/2 | 237 | 24/34 (70.6 ± 15.3) |
| 2 | 192 | 14/14 | 32 | 3/5 | 224 | 17/19 (89.5 ± 13.8) |
| 3 | 383 | 14/25 | 32 | 0/6 | 415 | 14/31 (45.2 ± 17.5) |
| 4 | 201 | 31/31 | 38 | 3/4 | 239 | 34/35 (97.1 ± 5.5) |
| Overall | 974 | 83/102 | 141 | 6/17 | 1115 | 89/119 (74.8 ± 7.8) |
| S.E.: Standard Error. | ||||||
Antibody titres
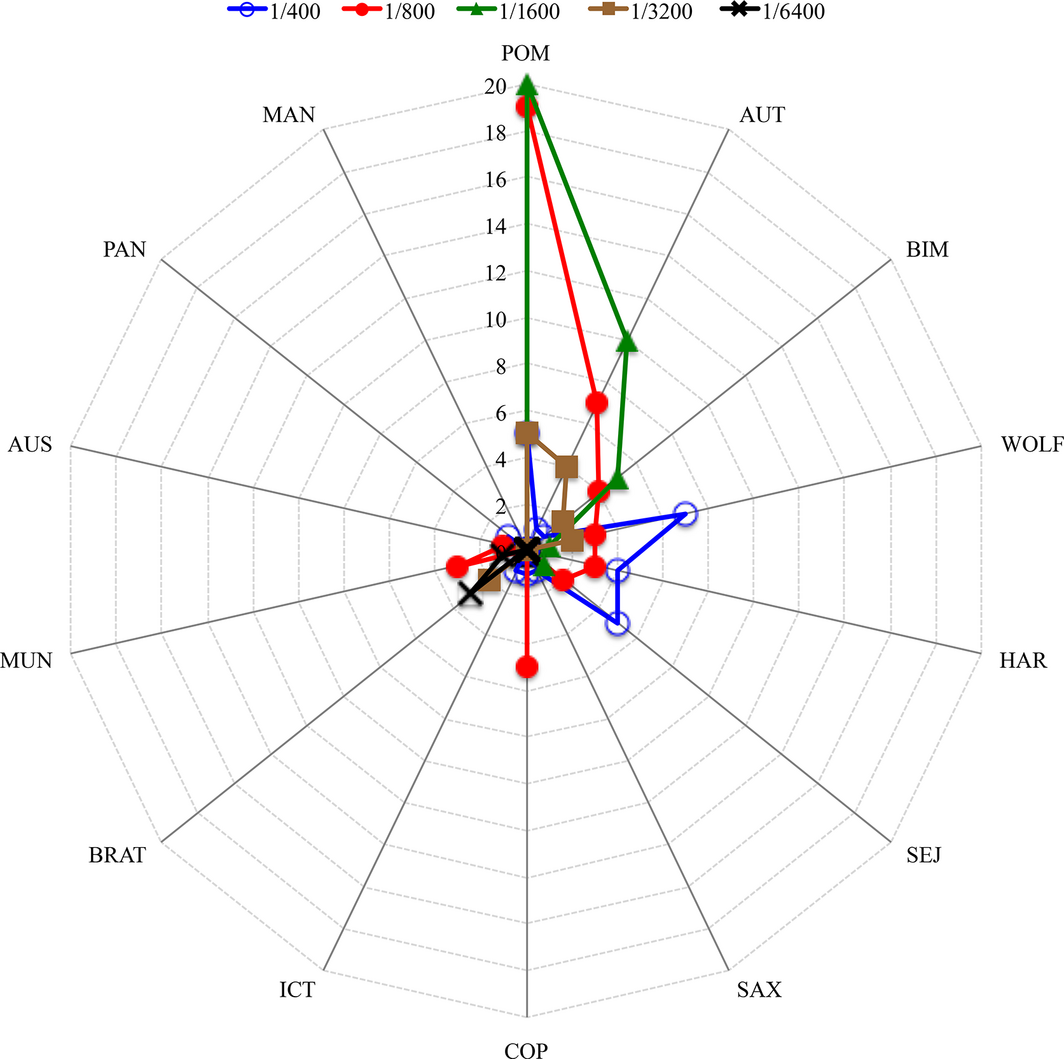
Positive sera titres ranged between 1/100 and 1/6400 with a median of 1/800 (Table 2; Fig. 1). More than half of the sera (68.7%; 332/483) reacted to at least one serovar at high titres (≥1/400). Three animals developed the highest titres (1/6400) for serovar Bratislava and one for serovar Munchen. High antibody titres to serovar Pomona were detected in 91% (61/67) of the tested animals (Table 2; Fig. 1). As sera of infected animals reacted to more than one serovar, the number of total seropositive animals (n = 89) was different from number of positive reactions to all tested serovars (n = 483).
| Antibodies titres | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Serogroup | Serovar | 1/100 | 1/200 | 1/400 | 1/800 | 1/1600 | 1/3200 | 1/6400 | Total (%) |
| Pomona (POM) | Pomona (POM) | 0 | 6 | 5 | 26 | 24 | 6 | 0 | 67 (13.9) |
| Autumnalis (AUT) | Autumnalis (AUT) | 4 | 7 | 6 | 16 | 16 | 4 | 0 | 53 (11) |
| Bim (BIM) | 3 | 7 | 14 | 14 | 9 | 5 | 0 | 52 (10.8) | |
| Icterohaemor-raghiae (ICT) | Copenhageni (COP) | 0 | 4 | 7 | 22 | 0 | 0 | 0 | 33 (6.8) |
| Icterohaemorraghiae (ICT) | 8 | 8 | 10 | 4 | 0 | 0 | 0 | 30 (6.2) | |
| Australis(AUS) | Australis (AUS) | 3 | 5 | 9 | 5 | 0 | 0 | 0 | 22 (4.6) |
| Munchen (MUN) | 1 | 17 | 12 | 18 | 0 | 0 | 1 | 49 (10.1) | |
| Bratislava (BRA) | 9 | 17 | 11 | 2 | 2 | 2 | 3 | 46 (9.5) | |
| Sejroe (SEJ) | Hardjo (HAR) | 6 | 13 | 17 | 4 | 2 | 0 | 0 | 42 (8.7) |
| Sejroe (SEJ) | 7 | 11 | 18 | 6 | 1 | 0 | 0 | 43 (8.9) | |
| Wolffii (WOLF) | 1 | 12 | 19 | 5 | 2 | 2 | 0 | 41 (8.5) | |
| Saxkoebing (SAX) | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 (0.2) | |
| Panama (PAN) | Panama (PAN) | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 2 (0.4) |
| Mangus (MAN) | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 2 (0.4) | |
| Total (%) | 42 (8.7) | 109 (22.6) | 130 (26.9) | 123 (25.5) | 56 (11.6) | 19 (3.9) | 4 (0.8) | 483 | |
| In bolded characters: The total number of positive reactions to all tested serovars | |||||||||
|
|
|
Figure 1. Number of animals reacting to different Leptospira serovars with the highest titres. |
The number of animals that developed the higher titre expressed against one or more serovars, is shown in the Figures 2 and 3. Out of 67 positive reactions to serovar(s) Pomona, 53 expressed the highest titre to this serovar alone or in addition to other serovars.
|
|
|
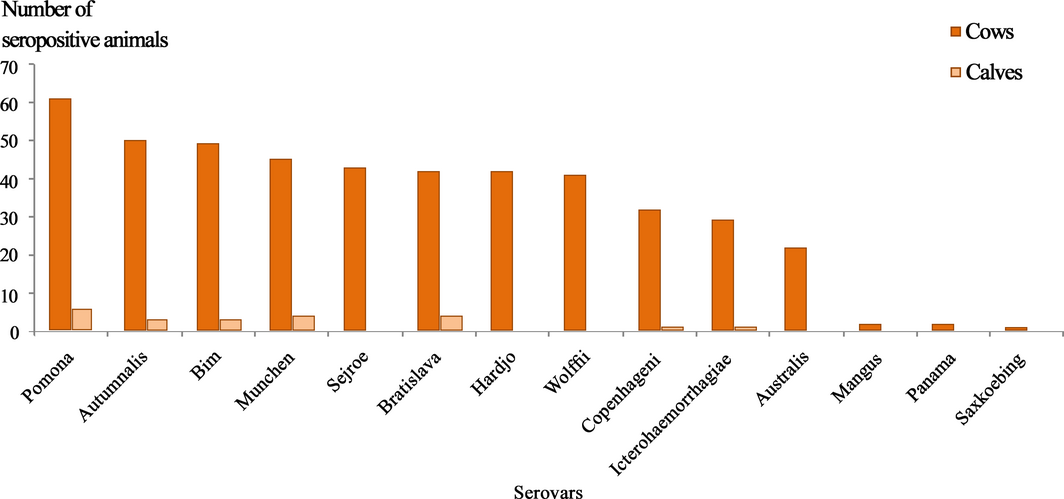
Figure 2. Number of seropositive animals reacting to against the 14 tested Leptospira spp. serovars. |
|
|
|
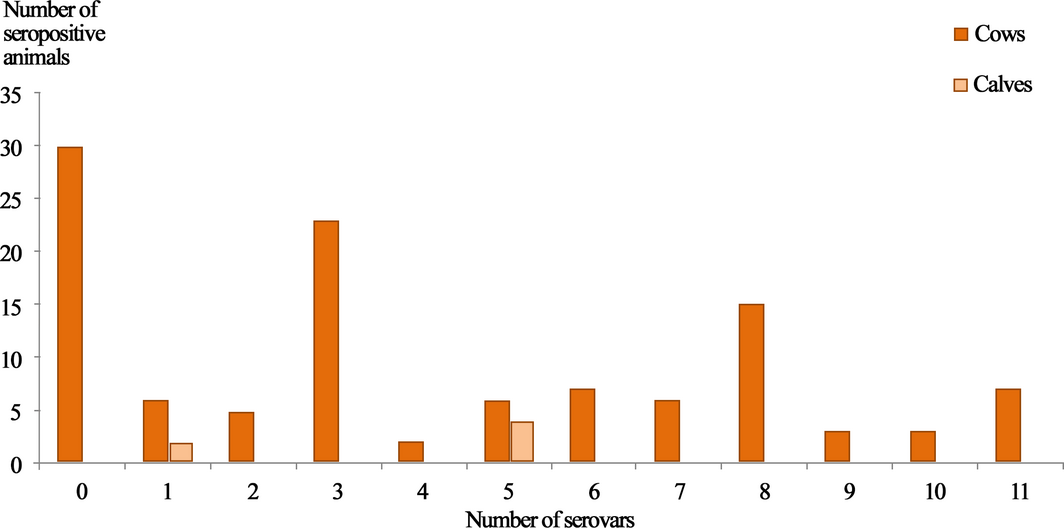
Figure 3. Number of animals showing seropositivity to different Leptospira serovars. |
Serovars
Animals reacted to all tested 14 serovars in the farm. Regardless of the highest titre cut-off, the most prevalent serovars were Pomona (75.3%), followed by Autumnalis (59.5%), Bim (58.4%) and Munchen (55%) (P < 0.05) (Fig. 2). The serovar Pomona was the most frequent in units 1, 2 and 4. In unit 3, out of the 14 seropositive sera tested, 13 reacted to one or more serovars from the serogroup Sejroe and one sera was positive to Pomona.
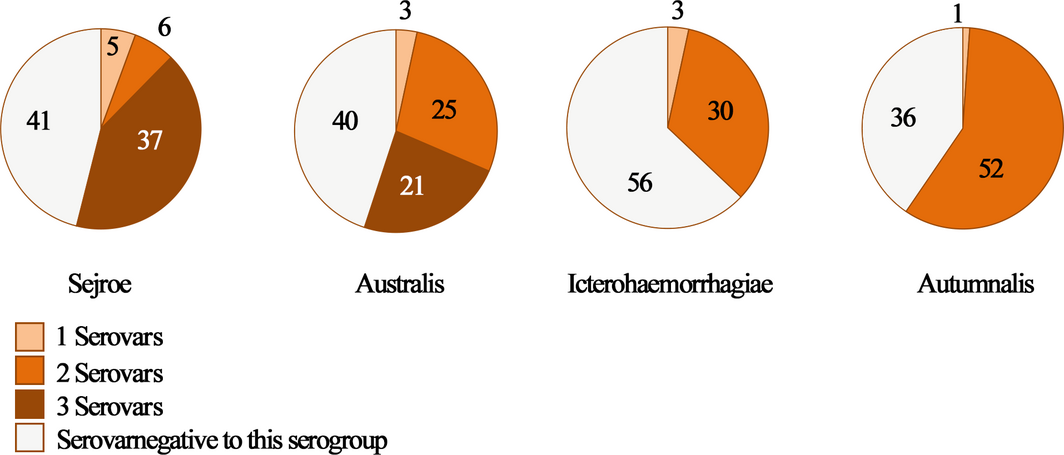
Only eight animals reacted to a single serovar (Pomona), cross-reactivity between at least two serovars were detected in 91% of positive animals (81/89), among them seven reacted to 11 serovars at the same time (Fig. 3). Among positive animals to serogroup Sejroe, 37/48 showed cross-reactivity to at least three serovars (Sejroe, Hardjo and Wolffii (n = 36); Sejroe, Wolffii and Saxkoebing (n = 1)). As expected, more than half of the animals (25/49; 51%) reacted to the serogroup Australis; they expressed cross-reactivity between two serovars (Munchen and Bratislava) and 21/49 (42.85%) to three serovars (Munchen, Bratislava and Australis). Most of the seropositive animals to serogroup Icterohaemorrhagiae (30/33; 90.9%) expressed cross-reactivity to the only two serovars (Icterohaemorrhagiae and Copenhageni). Roughly all animals positive to serogroup Autumnalis, reacted to both serovar Bim and serovar Autumnalis (52/53). The same observation was made for Panama and Mangus (2/2) (Fig. 4).
|
|
|
Figure 4. Frequency of serovars’ associations belonging to the same Leptospira serogroup. |
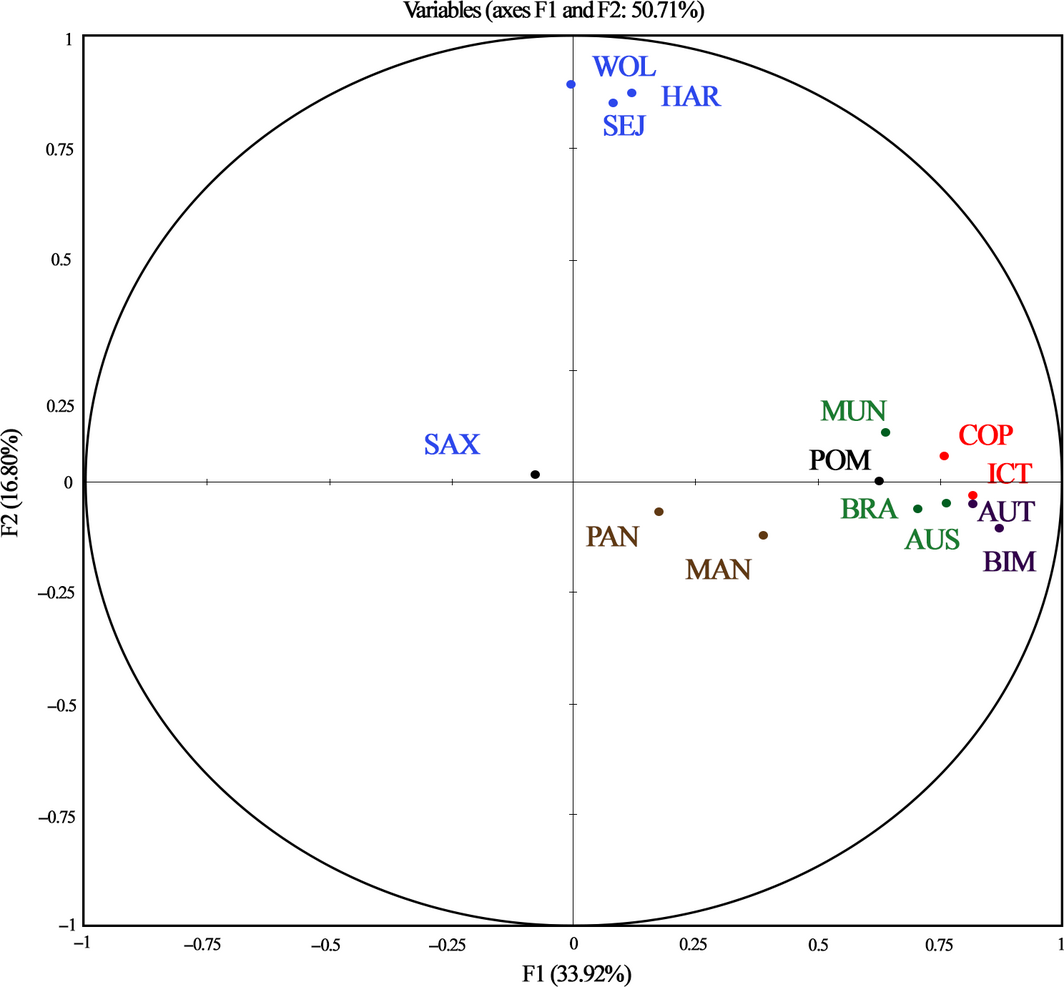
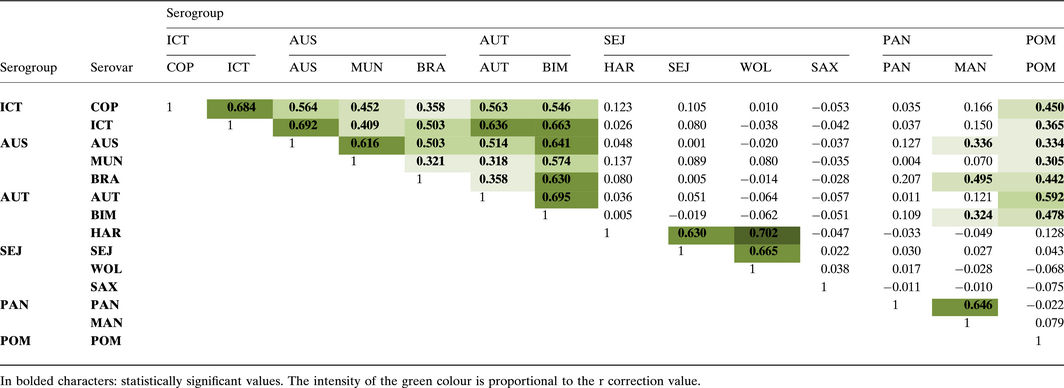
The Principal component analysis (PCA) showed high correlation values mainly between serovars belonging to the same serogroups (Fig. 5; Table 3). Curiously, animals developed cross-reactivity against Icterohaemorrhagiae, Australis and Autumnalis serogroups, and with less extent between Pomona and Icterohaemorrhagiae, Australis and Autumnalis serogroups. Sejroe did not cross-react with any of other serovar.
|
|
|
Figure 5. Principal component analysis of Leptospira spp. serovars associations. Bleu, serovars of Sejroe serogroup; Green, serovars of Australis serogroup; Red, serovars of Icterohaemorrhagiae serogroup; Purple, serovars of Autumnalis serogroup; Brown, serovars of Panama serogroup . |
Table 3. Correlation matrix of the Principal components analysis for different serovars
Discussion
This study showed a high seroprevalence of Leptospira spp. antibodies (74.8 ± 7.8%) and confirmed a leptospirosis outbreak in the studied farm. High antibody titres (≥1/400) were reported in 68.7% of seropositive animals indicating an active infection. In North Africa, few data are available about leptospirosis in both humans and animals. In Centre of Tunisia, we showed lower seroprevalence (25%) in sheep with no leptospirosis clinical cases (Khamassi Khbou et al. 2010).
In Egypt, the seroprevalence with MAT varied between 37.6 and 44.4% (Felt et al. 2011; Samir et al. 2015). The seroprevalences with MAT in Northern Morocco and Middle Atlas were 5 (5/99) and 1% (1/98) in goats and sheep with abortion history respectively (Benkirane et al. 2015).
According to the highest titre expressed by sera, Pomona was the predominant serovar (79.1%; 53/67). This serovar(s), was reported in other countries in cattle, by MAT in Iran (13/29) (Khalili et al. 2014); or by culture and PCR in Egypt (5/7) (Hatem et al. 2014). Serovar Pomona causes leptospirosis in cattle (Bharti et al. 2003) and is involved in foetal losses and abortion among non-vaccinated cows (Sanhueza et al. 2013). As in Tunisia there are no pigs, the main reservoir of serovar Pomona (Bharti et al. 2003), rodents may play this role but further studies are needed to confirm this role.
In contrast, Hardjo (Sejroe serogroup) is considered as the most prevalent serovar in cattle in England, Columbia, Chile and Brazil (Ellis et al. 1981; Hernández-Rodríguez et al. 2011; Lilenbaum & Martin 2014; Salgado et al. 2014). In France between 1988 and 2007, the most frequent serovar in cattle was Serjoe (34%) (André-Fontaine 2016). Similar trend was observed in the unit 3 of the present survey (13/14). As in this unit, the cross-reactivity with other serovars was low (Pomona, Panama and Man, one of each) and high titres (>3200) were expressed against serovar Wolffii, we can argue that in this unit, the animals were infected by a serovar belonging to the Sejroe serogroup.
Roughly all the tested animals (81/89) were seropositive to more than one serovar. Seropositivity to three serovars was the most frequent (23/81), and surprisingly, seven animals were positive to 11 serovars. Allan et al. (2015) reported that cattle are carriers of the widest range of Leptospira serogroups, but in our study, we cannot argue that positive animals that reacted to multiple serovars were co-infected, as co-infection is only confirmed by Leptospira culture or PCR. The seropositivity to several serovars is likely to be due to cross-reactivity, which is more intense between serovars belonging to the same serogroup (Faine et al. 1999). Based on highest titres, Pomona remains the predominant serovar, even in animals that showed high titres against multiple serovars. The combination of Pomona and Autumnalis was the most frequent among animals that strongly reacted to several serovars.
Antibodies are produced against species serovar-specific and serogroup-specific antigens (World Health Organization (WHO) 2003; Seenichamy et al. 2014). Although, cross-reactivity to multiple serovars is considered as a limit of MAT (Suepaul et al., 2011), when it is associated to high titres, it could indicate the pattern of the infected serogroups, as animals develop an intense immunological response in MAT to the first causative Leptospira serovar with higher titres than the others (Chappel et al. 2004; Ayral et al. 2014; André-Fontaine 2016; Blanco et al. 2016). If just the highest titre values are considered, it can be argued that Pomona was the predominant infecting serovar in three cattle units (1; 2 and 4), but in the cattle unit 3, serovar Sejroe was the most prevalent (13/14).
This study showed the presence of high seroprevalence with high titres against Leptospira serovar Pomona. The prevalence reported herein might be underestimated because we did not use local Leptospira serovars from Tunisia for MAT, and this could induces a decrease in MAT sensitivity (Office International des Epizooties (OIE) 2014; Mgode et al. 2015).
Nationwide surveys targeting on one hand different animal species in different Tunisian geographical regions should be carried out. At the same time, a seroepidemiological survey should be carried out in human Tunisian population to establish a list of the serovars that are mostly involved in human cases. These surveys will provide both human and animal health decision makers and practitioners with reliable and comprehensive epidemiological data to control this neglected disease.
Acknowledgements
The authors thank Prof. Geneviève André-Fontaine for her corrections of the manuscript and Prof. Angeli Kodjo for his advice.
Source of funding
This work was in part financially supported by the “Programme de recherche multidisciplinaire et inter-institutionnelle: Etude des contraintes relatives á la filière lait bovin dans la région de Bizerte: vers un nouveau bassin laitier ?”; IRESA, Tunisia. The study was supported financially by Laboratoire d’épidémiologie d'infections enzootiques des herbivores en Tunisie (Ministére de I'Enseignement Supérieur, de la Recherche Scientifique et des Technologies de l'Information et de la Communication).
Conflict of interest
There are no potential conflicts of interest.
Contribution
M. Khamassi Khbou: participated in sample collection, data analyses and wrote the paper. K. Haouala: participated in sample collection and coordinated the whole survey in field. M. Benzarti: designed the experiment.
References
- Allan K.J., Biggs H.M., Halliday J.E.B., Kazwala R.R., Maro V.P., Cleaveland S., Crump J.A. (2015). Epidemiology of leptospirosis in Africa: a systematic review of a neglected zoonosis and a paradigm for ‘one health’ in Africa. PLoS Neglected Tropical Diseases9, 1–25.
- André-Fontaine G. (2016). Leptospirosis in domestic animals in France: serological results from 1988 to 2007. Revue Scientifique et Technique (International Office of Epizootics)35. In press.
- Ayral F.C.D.J., Bicout H., Pereira M. Artois & Kodjo A. (2014) Distribution of Leptospira serogroups in cattle herds and dogs in France. American Journal of Tropical Medicine and Hygiene91(4), 756–759.
- Benkirane A., Noury S., Hartskeerl R.A., Goris M.G., Ahmed A., Nally J.E. (2014). Preliminary investigations on the distribution of Leptospira serovars in domestic animals in North-West Morocco. Transboundary and Emerging Diseases63(2), 178–184. doi: 10.1111/tbed.12252.
- Benkirane A., Essamkaoui S., El Idrissi A., Lucchese L. & Natale A. (2015) A sero-survey of major infectious causes of abortion in small ruminants in Morocco. Veterinaria Italiana51(1), 25–30.
- Bharti A.R., Nally J.E., Ricaldi J.N., Matthias M.A., Diaz M.M., Lovett M.A.et al. (2003) Leptospirosis: a zoonotic disease of global importance. The Lancet Infectious Diseases3(12), 757–771.
- Blanco R.M., do Santos L.F., Galloway R.L., Romero E.C. (2016). Is the microagglutination test (MAT) good for predicting the infecting serogroup for leptospirosis in Brazil?Comparative Immunology, Microbiology and Infectious Diseases44, 34–36.
- Chappel R.J., Goris M., Palmer M.F. & Hartskeerl R.A. (2004) Impact of proficiency testing on results of the microscopic agglutination test for diagnosis of leptospirosis. Journal of Clinical Microbiology42, 5484–5488.
- Desvars A., Cardinale E. & Michault A. (2011) Animal leptospirosis in small tropical areas. Epidemiology and Infection39, 167–188.
- Durand M. & Loquerie R. (1960) La leptospirose bovine en Tunisie: premier foyer décelé dans le Gouvernorat du Kef. Archives de L'Institut Pasteur de Tunis37, 67–72.
- Ellis W.A., O'brien J.J., Cassels J. (1981). Role of cattle in the maintenance of Leptospira interrogans serotype Hardjo infection in Northern Ireland. The Veterinary Record108, 555–557.
- Faine S., Adler B., Bolin C. & Perolat P. (1999) Leptospira and Leptospirosis. Medsci,Melbourne, Australia.
- Felt S.A., Wasfy M.O., El-Tras W.F., Samir A., Adel Rahman B., Boshra M.et al. (2011) Cross-species surveillance of Leptospira in domestic and peridomestic animals in Mahalla City, Gharbeya Governorate, Egypt. The American Journal of Tropical Medicine and Hygiene84(3), 420–425.
- Grooms D.L. (2006) Reproductive losses caused by bovine viral diarrhea virus and Leptospirosis. Theriogenology66(3), 624–628.
- Hatem M.E., Ata N.S., Abdou A.M., Ibrahim E.S., Bakry M.A. & Samir A. (2014) Surveillance of bovine leptospirosis: isolation and serodiagnosis. Global Veterinaria13(1), 127–132.
- Hernández-Rodríguez P., Díaz C.A., Dalmau E.A. & Quintero G.M. (2011) A comparison between polymerase chain reaction (PCR) and traditional techniques for the diagnosis of leptospirosis in bovines. Journal of Microbiological Methods84, 1–7.
- Horton K.C., Wasfy M., Samaha H., Abdel-Rahman B., Safwat S., Abdel Fadeel M., Dueger E. (2014). Serosurvey for zoonotic viral and bacterial pathogens among slaughtered livestock in Egypt. Vector-Borne and Zoonotic Diseases14, 633–639.
- Khalili M.S., Ehsanollah A.Mohammad, Reza A., Gholamreza S.T., Saeed M.D.Elham & Sajad H. (2014) Seroprevalence of bovine leptospiral antibodies by microscopic agglutination test in Southeast of Iran. Asian Pacific Journal of Tropical Biomedicine4(5), 354–357.
- Khamassi Khbou M., Hammami S. & Kodjo A. (2010) Séroprévalence des anticorps anti-leptospires chez les ovins dans la région d'El Fahs, Tunisie. Revue de Médecine Vétérinaire161(4), 185–192.
- Koizumi N., Muto M., Yamamoto S., Baba Y., Kudo M., Tamae Y.et al. (2008) Investigation of reservoir animals of Leptospira in the Northern part of Miyazaki prefecture. Japanese Journal of Infectious Diseases61(6), 465–468.
- Levett P.N. (2003) Usefulness of serologic analysis as a predictor of the infecting serovar in patients with severe leptospirosis. Clinical Infectious Diseases36, 447–452.
- Lilenbaum W. & Martin G. (2014) Leptospirosis in cattle: a challenging scenario for the understanding of the epidemiology. Transboundary and Emerging Diseases61, 63–68.
- Mgode G.F., Machang'u R.S., Mhamphi G.G., Katakweba A., Mulungu L.S., Durnez L.et al. (2015) Leptospira serovars for diagnosis of leptospirosis in humans and animals in Africa: common Leptospira isolates and reservoir hosts. PLoS Neglected Tropical Diseases9, e0004251. doi: 10.1371/journal.pntd.0004251. eCollection.
- Office International des Epizooties (OIE), 2014. Leptospirosis. Chapter 2.1.9. Manual of diagnosis tests and vaccines (Terrestrial Manual) . Available at: http://www.oie.int/fileadmin/Home/fr/Health_standards/tahm/2.01.09_LEPTO.pdf
- Picardeau M. (2013) Diagnosis and epidemiology of leptospirosis. Médecine et Maladies Infectieuses387(43), 1–9.
- Salgado M., Otto B., Sandoval E., Reinhardt G., Boqvist S.A. (2014) Cross sectional observational study to estimate herd level risk factors for Leptospira spp. serovars in small holder dairy cattle farms in southern Chile. BMC Veterinary Research10, 126. doi: 10.1186/1746-6148-10-126
- Samir A., Soliman R., El-Hariri M., Abdel-Moein K. & Hatem M.E. (2015) Leptospirosis in animals and human contacts in Egypt: broad range surveillance. Revista da Sociedade Brasileira de Medicina Tropical48(3), 272–277.
- Sanhueza J.M., Heuer C. & West D. (2013) Contribution of Leptospira, Neospora caninum and bovine viral diarrhea virus to fetal loss of beef cattle in New Zealand. Preventive Veterinary Medicine112, 90–98.
- Seenichamy A., Bahaman A.R., Mutalib A.R., Khairani-Bejo S. (2014) Production and characterization of a polyclonal antibody of anti-rLipL21-IgG against Leptospira for early detection of acute leptospirosis. BioMed Research International2014, Article ID 592858, 8 pages. doi:10.1155/2014/592858
- Suepaul S.M., Carrington C.V., Campbel M., Borde G. & Adesiyun A.A. (2011) Serovars of Leptospira isolated from dogs and rodents. Epidemiology and Infection138(7), 1059–1070.
- Thiermann A.B. (1984) Leptospirosis currents, developments and trends. Journal of the American Veterinary Medical Association184(6), 722–725.
- Vinetz J.M., Glass G.E., Flexner C.E., Mueller P. & Kaslow D.C. (1996) Sporadic urban leptospirosis. Annals of Internal Medicine125, 794–798.
- World Health Organization (WHO). (2003). Human leptospirosis: guidance for diagnosis, surveillance and control. Available at: http://www.who.int/csr/don/en/WHO_CDS_CSR_EPH_2002.23.pdf
Document information
Published on 09/06/17
Submitted on 09/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?