Abstract
A toxicological evaluation of a umami flavour compound, 2-(((3-(2,3-dimethoxyphenyl)-1H -1,2,4-triazol-5-yl)thio)methyl)pyridine (S3643; CAS 902136-79-2), was completed for the purpose of assessing its safety for use in food and beverage applications. S3643 undergoes extensive oxidative metabolism in vitro with rat microsomes producing the S3643-sulfoxide and 4′-hydroxy-S3643 as the major metabolites. In incubations with human microsomes, the O -demethyl-S3643 and S3643-sulfoxide were produced as the major metabolites. In pharmacokinetic studies in rats, the S3643-sulfoxide represents the dominant biotransformation product. S3643 was not found to be mutagenic or clastogenic in vitro , and did not induce micronuclei in CHO-WBL cells. In subchronic oral toxicity studies in rats, the no-observed-adverse-effect-level (NOAEL) for S3643 was 100 mg/kg bw/day (highest dose tested) when administered in the diet for 90 consecutive days.
Abbreviations
amu , atomic mass unit ; AUC , area under the curve ; CL , plasma clearance ; Cmax , peak plasma concentration ; CYP , cytochrome P450 ; FDA , Food and Drug Administration ; FEMA , Flavour and Extract Manufacturers Association of the United States ; FL-no , FLAVIS number ; GLP , Good Laboratory Practices ; GMP , Good Manufacturing Practices ; GPCR , G-protein-coupled receptor ; GRAS , generally recognized as safe ; HPBL , human peripheral blood lymphocytes ; LC/MS , liquid chromatography with mass spectrometry ; MC , methylcellulose ; MSG , monosodium glutamate ; NOAEL , no-observed-adverse-effect-level ; NOEL , no-observed-effect-level ; OECD , Organization for Economic Cooperation and Development ; PK , pharmacokinetics ; RCG , Relative Cell Growth ; RMI , Relative Mitotic Index ; t1/2 , half-life ; Tmax , time to reach Cmax ; TK , toxicokinetics ; Vss , volume of distribution at steady-state
Keywords
Umami flavour ; S3643 ; FEMA GRAS ; Subchronic toxicological evaluation ; Genetic toxicological evaluation
1. Introduction
Umami, the savory taste of the amino acid l -glutamate, is one of the five basic taste qualities detected by humans. Monosodium glutamate (MSG) is the prototypical umami substance commonly added to many food and beverage compositions, often at concentrations of 0.1–0.8% (1000–8000 ppm) by weight, to improve their overall fullness and savory flavour. In addition, it is known that naturally occurring purine ribonucleotides such as inosine-5′-monophosphate and guanosine-5′-monophosphate which elicit no umami taste on their own, can synergistically potentiate the umami taste of glutamate, thereby requiring less MSG for a given flavouring application. While these purine ribonucleotides can be present along with glutamate in certain food ingredients such as autolyzed yeast extracts, they are expensive to either isolate from natural sources or to synthesize. Until recently, little progress has been made in identifying high potency artificial substitutes for MSG or potentiators of the effectiveness of naturally occurring glutamate already present in food products.
Umami substances are detected by a specific subset of taste receptor cells localized in the taste bud and characterized by the expression of members of the hTAS1R family of class C G-protein-coupled receptors (GPCRs), which are distantly related to calcium sensing receptor, V2R pheromone receptors, and metabotropic glutamate receptors [13] , [14] and [31] . Co-expression of both hTAS1R1 and hTAS1R3 in heterologous cells results in a functional, heteromeric receptor which is highly selective for umami stimuli, responding only to glutamate, aspartate, and l -2-amino-4-phosphonobutrate. Most importantly, the glutamate-induced activity of the hTAS1R1/hTAS1R3 heterodimer is also strongly potentiated by inosine-5′-monophosphate and guanosine-5′-monophosphate, thereby proving further support for its identity as the human receptor for umami taste. This functional assay for hTAS1R1/hTAS1R3 has been adapted for high-throughput screening of natural extract and synthetic libraries leading to the discovery of several classes of novel umami agonists including a series of highly potent oxalamide compounds [24] , [25] and [26] . Several of these oxalamide umami agonists have been evaluated for potential genotoxicity and in vivo toxicity in rodents and have received regulatory approval as flavouring agents including N1 -(2,4-dimethoxybenzyl)-N2 -(2-(pyridin-2-yl)ethyl)oxalamide (S336, CAS 745047-53-4, FEMA 4233, FL-no. 16.099, Savorymyx® UM33). S336 has worldwide regulatory approval for use as a flavour compound and has been used to reduce or replace MSG in a variety of products including sauces, frozen foods, cooking aids and snack foods.
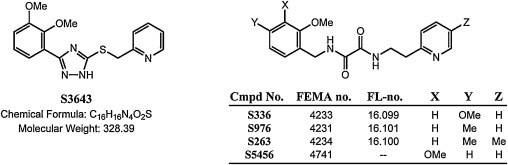
More recently, researchers at Senomyx, Inc. have reported a series of novel 3-aryl-5-alkylthio-1,2,4-triazoles, including 2-(((3-(2,3-dimethoxyphenyl)-1H -1,2,4-triazol-5-yl)thio)methyl)pyridine (S3643; CAS 902136-79-2), which are also potent agonists of the human umami receptor [27] and [28] . Like several of the aforementioned oxalamides, S3643 can provide an umami flavour effect in product applications equivalent to that of MSG at a 1000-fold lower concentration. The structure of S3643 along with representative analogs from the oxalamide series is shown in Fig. 1 .
|
|
|
Fig. 1. Structures of S3643 and oxalamide umami agonists. |
S3643 was reviewed by the Expert Panel of the Flavour and Extract Manufacturers Association of the United States (FEMA) and determined to be generally recognized as safe (GRAS) under conditions of intended use as a flavour ingredient [1] , [8] and [17] and therefore is available for use in human food in the United States as a “FEMA GRAS” flavour ingredient. S3643 was assigned FEMA GRAS Number 4798 in 2014 [1] .
The purpose of this publication is to summarize the results obtained from in vitro metabolism and in vivo pharmacokinetic (PK) studies, general toxicology studies in rodents, and genotoxicity studies conducted with S3643. Additional supporting data obtained in these studies with S3643 is included in a Supplementary Data section in the online publication.
2. Materials and methods
The batch of S3643 used for the in vitro /in vivo metabolism, in vivo pharmacokinetic, and 28-day range-finding toxicity studies (Lot no. BP110707, purity >99%, mp 114.7–115.5 °C), was synthesized at Ajinomoto Co., Inc., Kawasaki-Shi, Japan using the procedure described in US Patent No. 8,784,782 B2 and 8,968,708 B2 [27] and [28] . The batch of S3643 used for the in vitro genotoxicity and 90-day subchronic toxicity studies (Lot no. 60287-12-001-R, purity >98.5%, mp 114.7–115.3 °C) was synthesized at Ricerca Biosciences, LLC, Concord, OH using a slight modification of the same synthetic method but also prepared in conformance with Good Manufacturing Practices (GMPs) as described in the ICH GMP Guidelines for APIs [10] . Both batches of S3643 used for these studies gave identical 1 H NMR (400 MHz, d6 -DMSO), 13 C NMR (100 MHz, d6 -DMSO), FT-IR (KBr pellet), and mass spectral data.
All genetic toxicology studies were conducted in compliance with the United States Food and Drug Administration (FDA) Good Laboratory Practices (GLP) regulations 21 CFR Part 58 [6] and OECD guidelines [22] . The experimental design for these studies followed the OECD Guidelines for the Testing of Chemicals – 471, 473, and 487 [20] , [21] and [23] . The 28-day dose-range finding studies and 90-day toxicology studies in rats were conducted in compliance with FDA guidelines [7] Toxicological Principles for the Safety of Food Ingredients; the 90-day subchronic toxicology study was also conducted in compliance with GLP regulations, 21 CFR Part 58.
The receptor panel profiling and cytochrome P450 (CYP) inhibition assays on S3643 were conducted at MDS Pharma Services-Taiwan Ltd, Taipei, Taiwan. The in vitro microsomal metabolism studies on S3643 were carried out by PharmOptima, Portage, MI. The microsomal metabolism studies utilized male and female rat liver microsomes (Lot no. 1010122 and 0710104, respectively) and mixed gender human microsomes (Lot no. 087K1440) obtained from XenoTech, Lenexa, KS. The hERG channel inhibition assay on S3643 was carried out by Aviva Biosciences, San Diego, CA. Additional in vitro microsomal metabolism studies, as well as pharmacokinetic (PK) and in vivo metabolism studies on S3643 in rats were conducted at Senomyx, San Diego, CA. The analytical methods used for the in vitro metabolism, PK and in vivo metabolism studies can be found in the Supplementary Data section published online.
The in vitro genotoxicity studies for S3643 were conducted at Nucro-Technics, Scarborough, Ontario, Canada. The strains of S. typhimurium and E. coli , as well as rat liver S9 (9000 × g supernatant fraction of liver homogenate from Sprague-Dawley rats treated with Aroclor™ 1254) used in the reverse bacterial mutation assay were obtained from Molecular Toxicology Inc., Boone, NC. Cultures of human lymphocytes (Lot no. A2544) used for the chromosome aberration test were obtained from StemCell Technologies (Vancouver, BC, Canada) and were prepared from blood collected aseptically from a 20 year old, healthy, non-smoking donor. Chinese hamster ovary cell line WBL (CHO-WBL ) used for the in vitro micronucleus test was obtained from Dr. M.D. Baker, The Department of Pathobiology, University of Guelph (Guelph, ON, Canada). Rat liver S9 (9000 × g supernatant fraction of liver homogenate from Sprague-Dawley rats treated with phenobarbital and 5,6-benzoflavone) used in the chromosome aberration and micronucleus tests was obtained from Molecular Toxicology Inc., Boone, NC. The 28-day and 90-day subchronic toxicity studies for S3643 were conducted at MPI Research, Mattawan, WI. A description of the study designs is included in the individual study sections below. Detailed data tables for the genotoxicity, 28-day range-finder, and 90-day subchronic toxicity studies can be found in the Supplementary Data section published online.
3. Results and study designs
3.1. In vitro receptor and cytochrome P450 profiling of S3643
In vitro tests were conducted with S3643 to assess whether the compound interacts with any enzymes or receptors that might cause adverse or unexpected effects or affect drug metabolism. Preliminary in vitro screening for potential off-target activity of S3643 included tests for CYP inhibition, a receptor lead profiling panel (consisting of 68 receptor binding assays for GPCRs, ion channels, nuclear receptors, transporters), and a hERG inhibition assay. The preliminary tests for CYP inhibition were performed using recombinant human enzymes expressed in insect Sf9 cells using spectrofluorimetric substrates [2] and [18] . All assays were performed at a concentration of 10 μM of S3643. No significant responses (≥50% inhibition or stimulation) were found with S3643 in the lead profiling receptor screen. S3643 did not significantly inhibit the hERG ion channel current (<10%) in an in vitro hERG electrophysiology (patch clamp) assay [29] . However, S3643 did show significant inhibition of CYP1A2 and 3A4 (72% and 68% inhibition, respectively) at 10 μM in the spectrofluorimetric assay using recombinant human enzymes. As a follow up to the results obtained using spectrofluorimetic substrates, S3643 retested on the same panel of CYP enzymes utilizing human liver microsomes and CYP-specific substrates with detection of the CYP-specific metabolites by LC–MS/MS [15] and [30] . None of the CYP isoforms (including CYP1A2 and 3A4) were inhibited by >43% in the presence of 10 μM S3643 in this more definitive assay format (i.e. , all IC50 ’s >10 μM). The results from the CYP inhibition studies are summarized in Table 1 .
| CYP | Spectrofluorimetric assay, human recombinant enzymes, Sf9 cells | LC–MS/MS assay in human liver microsomes | ||
|---|---|---|---|---|
| Probe Substrate | % Inhibition (10 μM) | Probe Substrate | % Inhibition (10 μM) | |
| 1A2 | 3-cyano-7-ethoxycoumarin | 72% | phenacetin | 43% |
| 2C9 | 3-cyano-7-ethoxycoumarin | 5% | tolbutamide | 8% |
| 2C19 | 3-cyano-7-ethoxycoumarin | 42% | S-mephenytoin | 13% |
| 2D6 | 3-cyano-7-ethoxycoumarin | −3% | bufuralol | 22% |
| 3A4 | 7-benzyloxy-4-(trifluoromethyl)-coumarin | 68% | midazolam | 30% |
3.2. Absorption, distribution, metabolism, excretion
The in vitro metabolism of S3643 was studied using rat and human liver microsomes. A study of the PK and in vivo metabolism of S3643 was carried out in male and female Sprague-Dawley rats.
3.2.1. In vitro metabolism of S3643
The potential of S3643 to undergo oxidative metabolism was investigated using Sprague-Dawley rat and human liver microsomes in order to determine the similarity of the metabolic profile across these species. Reference standards were synthesized for the three potential oxidative metabolites that could be produced by mono-hydroxylation of the 2,3-dimethoxyphenyl moiety, as well as the S3643-sulfoxide, the two mono-O -demethylated S3643 analogs, and the analogous di-O -demethylated compound. S3643 (10 μM) was incubated with mixed gender, pooled rat and human liver microsomes (0.5 mg/mL) in the presence of NADPH (1.3 mM) at 37 °C for 60 min prior to quenching the samples with acetonitrile. Control samples included time zero and 60 min incubations without NADPH as well as testosterone incubation samples in order to verify microsome functionality.
Samples were centrifuged to separate the precipitated microsomes from the supernatant containing the parent compound and its metabolites. The supernatants from the S3643 incubations were analyzed by liquid chromatography-mass spectrometry (LC–MS) using a Phenomenex Kinetex C18 column (2.6 μm, 50 × 2.1 mm) with a 1.0 mM ammonium acetate and methanol gradient system, and a Thermo Electron Quantum Access mass spectrometer using heated electrospray ionization (HESI). The samples were scanned in the both positive and negative ionization modes for common Phase I transformations including addition of oxygen (+16), mono- and di-O -demethylation (−14 and −28, respectively), mono-O -demethylation + addition of oxygen (+2), addition of two oxygens (+32), and S -dealkylation (−91). Positive control incubations with testosterone with and without NADPH were analyzed in HESI positive ion mode. There was greater than 85% turnover of testosterone in parallel incubations with microsomes of both species in the presence of NADPH at the 60 min time point. Samples from a duplicate incubation with rat liver microsomes were analyzed by LC-QTOF/MS (Agilent iFunnel 6550A MS QTOF, positive mode) equipped with an Agilent 1290 Infinity Binary pump and an Agilent 1290 Infinity autosampler using a Zorbax Eclipse Plus C18 column (50 × 2.1 mm, 1.8 μm) with 2.0 mM ammonium acetate/0.1% formic acid/water and methanol gradient system to confirm the exact mass of the observed metabolites. Details of the analytical methods can be found in the Supplementary Data section.
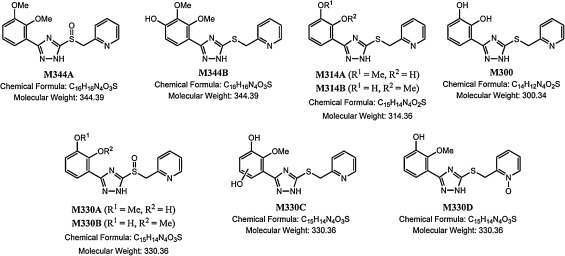
In the rat and human microsome incubations, roughly 18.4% and 54.9% of the parent was remaining at the end of the microsomal incubation period, respectively. At the 60 min time point, nine potential Phase I metabolites were observed at levels ranging from <1% to 18.6% of the initial S3643 peak area in both the rat and human incubations (see Fig. 2 and Table 2 ). The two major metabolites observed in the rat microsomal incubations were S3643-sulfoxide (M344A), and 4′-hydroxy-S3643 (M344B), representing roughly 18.6% and 10.5% of the initial S3643 peak area, respectively. The position of the hydroxyl group in metabolite M344B was confirmed by direct comparison to synthetic samples of the three possible hydroxy-2,3-dimethyoxyphenyl regioisomers (see Supplemental Material). The two major metabolites observed in the human microsomal incubations were an O -desmethyl-S3643 (M314B) and S3643-sulfoxide (M344A), representing roughly 13.8% and 6.48% of the initial S3643 peak area, respectively. The 4′-hydroxy-S3643 (M344B) seen as a major metabolite in the rat microsomal incubations was also present in the human microsomal incubations, but at significantly lower concentrations (1.82% of the initial S3643 peak area). Likewise, the O -desmethyl-S3643 (M314B) seen as a major metabolite in the human microsomal incubations was also seen as a minor metabolite in the rat incubations (2.28% of the initial S3643 peak area). Other metabolites observed in both the rat and human microsomal incubations include a second O -desmethyl-S3643 (M314A), a di-O -desmethyl-S3643 (M300), and two O -desmethyl-S3643 sulfoxides (M330A and M330B). A third metabolite (M330C) with molecular weight of 330 amu appears to be an O -desmethyl-S3643 which has been hydroxylated on the phenyl appendage. A fourth metabolite (M330D) with a molecular weight of 330 amu appears to be the result of addition of an oxygen atom to the pyridyl ring. Both M330C and M330D represent less than 1% of the initial S3643 initial peak area in the rat microsomal incubations. The structures of M344A, M344B, M314A, M314B, and M300 were confirmed by direct comparison to synthetic samples by LC–MS/MS. The structures of M330A, M330B, M330C, and M330D are based on their exact masses and MS fragmentation patterns, but have not been confirmed by comparison to synthetic samples (see Supplemental Material).
|
|
|
Fig. 2. Structures of S3643 major microsomal metabolites. |
| Metabolite | m /z (M+H) | Formula | % MS Peak Areaa | RT (min)b | |
|---|---|---|---|---|---|
| Rat | Human | ||||
| S3643 | 329.1067 | C16H17N4O2S+ | 18.4% | 54.9% | 5.79 |
| M344A | 345.1016 | C16H17N4O3S+ | 18.6% | 6.48% | 5.46 |
| M344B | 345.1016 | C16H17N4O3S+ | 10.5% | 1.82% | 4.42 |
| M314A | 315.0910 | C15H15N4O2S+ | 3.26% | 2.02% | 5.46 |
| M314B | 315.0910 | C15H15N4O2S+ | 2.28% | 13.8% | 4.26 |
| M330A | 331.0859 | C15H15N4O3S+ | 0.801% | 0.824% | 4.97 |
| M330B | 331.0859 | C15H15N4O3S+ | 3.39% | <1% | 4.05 |
| M330C | 331.0859 | C15H15N4O3S+ | <1% | <1% | 3.18 |
| M330D | 331.0859 | C15H15N4O3S+ | <1% | <1% | 4.31 |
| M300 | 301.0754 | C14H13N4O2S+ | <1% | <1% | 4.00 |
a. Percent integrated MS peak areas from HESI positive ion Q1 MS scan relative to integrated MS peak area of S3643 at time = 0.
b. Retention time using a Zorbax Eclipse Plus C18 column (50 × 2.1 mm, 1.8 μm) and 2 mM ammonium acetate/0.1% formic acid/water and methanol gradient system.
3.2.2. Pharmacokinetics of S3643 in rats
The PK parameters and oral bioavailability of S3643 in plasma was evaluated following either a single intravenous or oral administration in male and female Sprague-Dawley rats. For intravenous administration, 4 male and 4 female Sprague-Dawley rats were bolus injected with S3643 at 1 mg/kg bw in 10% ethanol in 20 mM potassium phosphate buffer (pH 7.4). Blood samples were collected from a jugular catheter at approximately 0, 2, 5, 10, 30 min, 1, 2, 4, and 8 h post-dose. For oral administration, 4 male and 4 female Sprague-Dawley rats per group were given a single dose of S3643 at either 10, 30, or 100 mg/kg bw in 1% methylcellulose (MC) by oral gavage. Blood samples were taken from a jugular catheter at approximately 0, 15, 30 min, 1, 2, 4, 8, and 24 h post-dose. Proteins from plasma samples (25 μL) were precipitated by addition of acetonitrile (75 μL) containing an internal standard [2-(((3-(2,4-dimethylphenyl)-1H -1,2,4-triazol-5-yl)thio)methyl)pyridine; S9767, 25 ng/mL], centrifuged, and 50 μL of the resulting supernatant was mixed with deionized water (100 μL). The resulting solutions were analyzed for S3643 by LC–MS/MS using a Waters SunFire C18 column (50 × 2.1 mm, 3.5 μm) with 0.1% formic acid/water and acetonitrile gradient system and a API 3200 Q-Trap mass spectrometer operated in positive ionization mode equipped with an Agilent 1100 binary pump with a CTC PAL injector. The parent compound and internal standard (IS) were detected using a source that was configured with turboionspray ionization in the positive mode using multiple-reaction monitoring (MRM) of mass transition pairs at m /z of 329.1/206.1 (S3643) and 297.1/174.1 (IS, S9767) amu. Details of the analytical methods can be found in the Supplementary Data section. Test article formulations prepared for this study were analyzed for concentration by HPLC-UV (265 nm). The PK parameters for S3643 are shown in Table 3 .
| Route | Dose (mg/kg bw) | Sex | Cmax (ng/mL) | Tmax (h) | t1/2 (h) | AUC0-last (ng·h/mL) | AUC0-last /dose (ng·h/mL/mg/kg) | % F |
|---|---|---|---|---|---|---|---|---|
| iv | 1.0 | M | 10870±1870 | 0.03 | 0.19 | 2790±187 | 2790 | – |
| F | 9840±1880 | 0.03 | 0.16 | 2110±353 | 2110 | – | ||
| oral gavage | 10 | M | 32510±7710 | 0.25 | 0.80 | 26650±7040 | 2665 | 95.5% |
| F | 14890±7870 | 0.25 | 0.89 | 14230±8860 | 1423 | 67.4% | ||
| 30 | M | 50880±10800 | 0.44 | 0.97 | 80330±29900 | 2678 | 96.0% | |
| F | 45380±5030 | 0.25 | 1.14 | 73510±33760 | 2450 | 116.1% | ||
| 100 | M | 78270±9520 | 0.31 | 1.66 | 183300±42930 | 1833 | 65.7% | |
| F | 98910±19350 | 0.38 | 1.66 | 239300±119400 | 2393 | 113.4% |
Male rat: CL = 5.97 mL/min/kg; Vss = 337 mL/kg Female rat: CL = 8.03 mL/min/kg; Vss = 325 mL/kg.
CL = clearance; Vss = steady-state volume of distribution; % F = bioavailability.
S3643 was rapidly eliminated after intravenous administration in both male and female rats with mean terminal half-lives (t1/2 ) of 0.19 and 0.16 h, respectively. Mean plasma clearance (CL) in rats averaged 5.97 mL/min/kg for males (10.8% of hepatic blood flow, [3] ) and 8.03 mL/min/kg for females (14.5% of hepatic blood flow), and the volume of distribution at steady-state (Vss ) averaged 337 and 325 mL/kg (50.4% and 48.6% of total body water volume) for males and females, respectively.
For oral administration, the mean values for the t1/2 of S3643 in plasma ranged from 0.80 to 1.66 h in male rats and 0.89–1.66 h in female rats. Both mean AUClast and mean Cmax increased with increasing oral dose, but the increase was not dose proportional in either male or female rats. For male rats, the mean Cmax for an oral dose of 10, 30, and 100 mg/kg bw was 32.51, 50.88, and 78.27 μg/mL, respectively; the mean AUClast for these doses was 26.65, 80.33, and 183.30 μg·h/mL, respectively. For female rats, the mean Cmax for an oral dose of 10, 30, and 100 mg/kg bw was 14.89, 45.38, and 98.91 μg/mL, respectively; the mean AUClast for these doses was 14.23, 73.51, and 239.30 μg·h/mL, respectively.
The ratio of female/male exposure to S3643 (Cmax and AUClast ) in plasma was used for comparison of gender differences. For intravenous administration, the female/male ratios of mean AUClast and Cmax were 0.76 and 0.91, respectively. For oral administration, the female/male ratios of mean AUClast ranged from 0.53 to 1.31 and the female/male ratio of mean Cmax ranged from 0.46 to 1.26. The absolute bioavailability (%F) ranged from 67.4% to 116.1% in female rats and 65.7% to 96.0% in male rats.
3.2.3. In vivo metabolism of S3643 in rats
The in vivo metabolism of S3643 was evaluated following a single oral administration in male and female Sprague-Dawley rats. A group of 4 male and 4 female Sprague-Dawley rats was administered 100 mg/kg bw of S3643 in 1% MC by oral gavage. Blood samples were collected from a jugular catheter at approximately 15, 30 min, 1, 2, 4, 8, and 24 h post-dose. Plasma samples (10 μL) from each animal was pooled together by each time point. Proteins from the combined plasma samples (80 μL) were precipitated by addition of acetonitrile (240 μL) containing an internal standard (S9767, 25 ng/mL), centrifuged, and 50 μL of the resulting supernatant was mixed with deionized water (100 μL). The resulting solutions were analyzed for the parent compound and metabolites by LC-QTOF/MS (Agilent iFunnel 6550A MS QTOF, positive mode) equipped with an Agilent 1290 Infinity Binary pump and an Agilent 1290 Infinity autosampler using a Zorbax Eclipse Plus C18 column (50 × 2.1 mm, 1.8 μm) with 2.0 mM ammonium acetate/0.1% formic acid/water and methanol gradient system. Details of the analytical methods can be found in the Supplementary Data section.
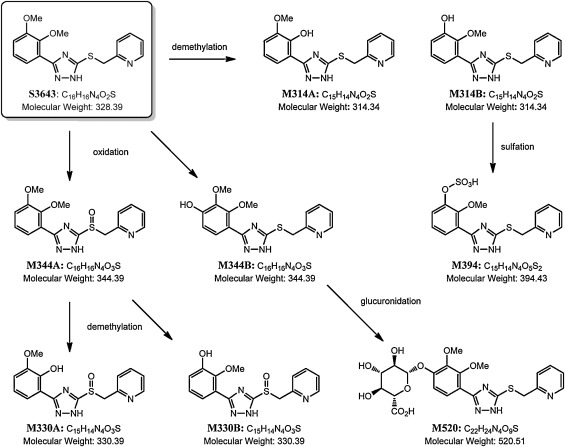
Seven Phase I metabolites and two Phase II metabolites of S3643 were observed in the rat plasma samples. The MS extracted ion chromatogram (EIC) peak areas for S3643 and its metabolites observed at the 1 h time point are shown in Table 4 . The Phase I metabolic biotransformation of S3643 involved oxidation of the thioether, demethylation, and hydroxylation of the phenyl appendage. The sulfoxide (M344A) of S3643 was the dominant biotransformation. Both mono-O -desmethyl-S3643 metabolites (M314A and M314B) and 4′-hydroxy-S3643 (M344B) were also observed in the rat plasma, but at much lower concentrations. The structures of M344A, M344B, M314A, and M314B were confirmed by direct comparison to synthetic samples by LC–MS/MS. In addition to traces of the other Phase I metabolites observed previously in the in vitro microsomal metabolism study (M330A, M330B, M300), a desmethyl, O -sulfate (M394) and glucuronide (M520) were also observed in the rat plasma samples. The metabolic pathway of S3643 in rats is shown in Fig. 3 .
| Compound | m /z (positive) | Formula | Peak Area (×106 ) | % Peak Area | RT (min) |
|---|---|---|---|---|---|
| S3643 | 329.1069 | C16H17N4O2S+ | 69.9 | 55.52 | 5.80 |
| M344A | 345.1016 | C16H17N4O3S+ | 54.6 | 43.39 | 5.48 |
| M344B | 345.1016 | C16H17N4O3S+ | 0.119 | 0.09 | 4.43 |
| M314A | 315.0910 | C15H15N4O2S+ | 0.488 | 0.39 | 5.46 |
| M314B | 315.0910 | C15H15N4O2S+ | 0.0288 | 0.02 | 4.26 |
| M330A | 331.0859 | C15H15N4O3S+ | 0.0287 | 0.02 | 4.97 |
| M330B | 331.0859 | C15H15N4O3S+ | 0.0165 | 0.01 | 4.06 |
| M394 | 395.0478 | C15H15N4O5S2+ | 0.570 | 0.45 | 3.22 |
| M520 | 521.1337 | C22H25N4O9S+ | 0.121 | 0.10 | 2.74 |
| M300 | 301.0754 | C14H13N4O2S+ | (trace) | <0.01 | 4.00 |
|
|
|
Fig. 3. Metabolic pathway of S3643 in rats. |
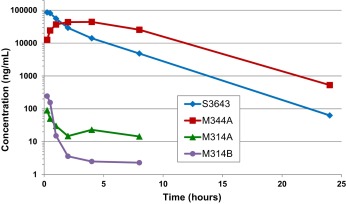
The mean combined AUClast of sulfoxide M344A was approximately 2.17-fold of the parent compound S3643 (see Table 5 and Fig. 4 ). The mean combined Cmax of sulfoxide M344A was 44.2 μg/mL (128.5 μM) versus 86.0 μg/mL (262 μM) for S3643. Both S3643 and its sulfoxide M344A have t1/2 in the range of 2.56-3.05 h. The Cmax of both O -desmethyl metabolites M314A and M314B was significantly lower (0.090 and 0.244 μg/mL, respectively). It is worth noting that metabolite M314B appears to have a significantly shorter t1/2 than its regioisomer M314A and their systemic exposures based on AUClast are similar despite the higher Cmax for M314B. This may be due in part to the rapid conversion of M314B to the corresponding O -sulfate M394. Although the actual concentrations of the desmethyl, O -sulfate (M394) and glucuronide (M520) could not be determined due to unavailability of synthetic samples, the MS EIC peak areas suggest that they are present in concentrations comparable to those of the O -desmethyl metabolites M314A and M314B. Overall, the results indicate that the sulfoxide (M344A) of S3643 represents the dominant biotransformation in rats.
| Cmpd | Time (h) | AUC (ng·h/mL) | Cmax (ng/mL) | Tmax (h) | t1/2 (h) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.25 | 0.50 | 1.0 | 2.0 | 4.0 | 8.0 | 24.0 | |||||
| Concentration (ng/mL) | |||||||||||
| S3643 | 86022.2 | 81446.8 | 55905.2 | 29188.1 | 14074.8 | 4832 | 63 | 228807.5 | 86022.2 | 0.25 | 2.56 |
| M344A | 12639.1 | 24340.5 | 36905.6 | 43577.9 | 44203.7 | 25558.1 | 529.5 | 497761.5 | 44203.7 | 4.00 | 3.05 |
| M314A | 89.6 | 50.1 | 30.1 | 14.8 | 23 | 14.4 | 0 | 183.9 | 89.6 | 0.25 | 3.67 |
| M314B | 243.9 | 156.1 | 15.1 | 3.6 | 2.5 | 2.3 | 0 | 148.3 | 243.9 | 0.25 | 0.69 |
|
|
|
Fig. 4. Mean plasma concentrations of S3643, M344A, M314A and M314B after oral administration of S3643 (100 mg/kg) to Sprague-Dawley rats (male and female combined, n = 8). |
3.3. Genotoxicity and mutagenicity studies
S3643 was evaluated for its genotoxic potential in vitro through standard (5-strain) Ames, chromosome aberration, and micronucleus tests (see Table 6 ). All genetic toxicology studies were conducted in compliance with the FDA GLP regulations 21 CFR Part 58 (2006) and OECD guidelines (1998). The data tables for the genotoxicity studies can be found in the Supplemental Material.
| End-Point | Test System | Concentration/Dose | Result |
|---|---|---|---|
| Reverse mutation (in vitro) | S. typhimurium strains TA98, TA100, TA1535, TA1537 and E. coli strain WP2 uvrA | S. typhimurium strains: | Negative |
| 130–2000μg/plate, plate incorporation, ±S9a | |||
| 130–2000μg/plate, preincubation, −S9 | |||
| 63–1000μg/plate, preincubation, +S9a | |||
| E. coli strain WP2 uvrA: | |||
| 250–5000μg/plate, plate incorporation and pre-incubation, ±S9a | |||
| Chromosome aberration (in vitro) | Primary human lymphocytes | 130–500μg/mL, 3h exposure, −S9 | Negative |
| 63–250μg/mL, 3h exposure, +S9b | |||
| 63–250μg/mL, 20h exposure, −S9 | |||
| Micronucleus formation (in vitro) | Chinese hamster ovary cells (CHO-WBL) | 100–300μg/mL, 3h exposure, −S9 | Negative |
| 50–200μg/mL, 3h exposure, +S9b | |||
| 16–63μg/mL, 19h exposure, −S9 |
a. S9 from rat liver homogenate from male Sprague-Dawley rats treated with Aroclor-1254.
b. S9 from rat liver homogenate from male Sprague-Dawley rats treated with phenobarbital/5,6-benzoflavone.
3.3.1. Bacterial reverse mutation test (5-Strain Ames)
S3643 was evaluated for the potential to induce point or frame shift mutations in S. typhimurium strains TA98, TA100, TA1535, TA1537 and E. coli strain WP2 uvrA in the presence and absence of metabolic activation with rat liver S9 from rats induced with Aroclor™ 1254. The assay was designed to meet the current OECD Guideline for Testing of Chemicals No. 471, Bacterial Reverse Mutation Test [20] .
In the plate incorporation assay, both in the presence and absence of S9, the maximum concentration of S3643 investigated for the S. typhimurium strains was 2000 μg/plate. In the preincubation experiment in the absence of S9, the maximum concentration of S3643 utilized for the S. typhimurium strains was also 2000 μg/plate. Due to increased toxicity in the presence of S9, the maximum concentration of S3643 investigated in the preincubation assay for the S. typhimurium strains was 1000 μg/plate. For the E. coli strain in both the plate incorporation and preincubation assays, the maximum concentration tested was 5000 μg/plate both in the presence and absence of S9. Once plated and at the end of the incubation period, slight and moderate precipitate was visible at 2000 and 5000 μg/plate, respectively. Therefore at these concentrations, the test article was evaluated at the limit of solubility in the test system. Slight toxicity was observed for S. typhimurium strains as evident by a reduction in colony counts at the higher concentrations when compared to the concurrent negative controls. However, a normal lawn was observed for all conditions and toxicity was not observed for the E. coli strain in all conditions. For all strains and conditions, a minimum of 5 concentrations were analyzable for mutagenicity.
For the plate incorporation test, with or without metabolic activation, S3643 did not produce any statistically significant increases in revertants over the concurrent negative controls. The preincubation test confirmed the negative results of the plate incorporation test as S3643 did not produce any increases in revertants over the concurrent negative controls. The negative controls for each tester strain were within the historical negative control data. All concurrent positive controls induced significant increase (p < 0.01, t -test) in colony counts (at least 3.4-fold) when compared to the corresponding negative controls and were at levels similar to the historical positive control data. Thus, it was concluded that S3643 was not mutagenic to S. typhimurium strains TA98, TA100, TA1535, TA1537 and E. coli strain, WP2 uvrA , in the absence and presence of metabolic activation.
3.3.2. In vitro chromosome aberration test
S3643 was investigated for its potential to induce structural and numerical chromosome aberrations in mammalian cells, both in the presence and absence of a supplemental liver fraction (S9) from rats treated with phenobarbital and 5,6-benzoflavone. The experimental design followed the OECD Guideline for the Testing of Chemicals No. 473, In Vitro Mammalian Chromosome Aberration Test [21] . A preliminary toxicity test was performed to establish the dose range for testing in the cytogenetic test. In the chromosome aberration assay, human peripheral blood lymphocytes (HPBL) were treated for 3 and 20 h in the non-activated test system and for 3 h in the S9-activated test system. Solvent and positive control (mitomycin C, −S9; cyclophosphamide, +S9) cultures were also included. For all conditions, cultures were harvested approximately 20–21 h after initiation of treatment. Approximately 3 h prior to harvesting, colchicine was added to the cultures at 0.1 μg/mL to arrest cells in metaphase.
Test article precipitate was only observed in the test system for 20-h exposure at the highest concentration of 500 μg/mL. For the 3-h exposure experiment without S9 the concentrations 63, 130, 250 and 500 μg/mL yielded Relative Cell Growth (RCG) of 100, 93, 99 and 83% and Relative Mitotic Index (RMI) of 89, 94, 64 and 42%, respectively. For 20-h exposure without S9 the concentrations 63, 130, 250 and 500 μg/mL, yielded RCGs of 101, 98, 86 and 51%. The lowest three concentrations yielded RMIs of 87, 62 and 27%, respectively. The highest concentration was not analyzable for RMI due to toxicity. For the 3-h exposure experiment with S9 the concentrations 31, 63, 130, and 250 μg/mL yielded RCGs of 94, 85, 73 and 78% and RMIs of 113, 93, 76 and 60%, respectively. All conditions were tested at the limit of test article toxicity evaluated by RCG and RMI levels.
Under these test conditions, no structural or numerical chromosome aberrations were observed in the S3643-treated cultures beyond those seen in the concurrent solvent controls. All concurrent positive controls induced significant numbers (p < 0.001) of cells with chromosome aberrations. Based on the findings of this study, S3643 was concluded to be negative for the induction of structural and numerical chromosome aberrations in both non-activated and S9-activated test systems in the in vitro mammalian chromosome aberration test using HPBL.
3.3.3. In vitro micronucleus test
The purpose of this study was to evaluate the genotoxic (clastogenic/aneugenic) potential of S3643 as measured by its ability to induce micronuclei in Chinese hamster ovary cells (CHO-WBL ) in both the absence and presence of liver preparations (S9 mix) from rats treated with phenobarbital and 5,6-benzoflavone. The experimental design followed the OECD Guideline for the Testing of Chemicals – 487, In Vitro Mammalian Cell Micronucleus Test [23] .
Cultures of CHO-WBL cells were exposed to varying concentrations of S3643 under three different conditions: (1) a 3-h exposure in the absence of S9 metabolic activation; (2) a 3-h exposure in the presence of S9; and (3) a 19-h exposure in the absence of S9 activation. Solvent and positive control (colchicine, −S9; cyclophosphamide, +S9) cultures were also included. Cytochalasin B at 3 μg/mL was present in the medium after the short-term exposure to S3643 and during the extended exposure in its entirety. Cells were harvested approximately 19–21 h (i.e. , approximately 1.5 × the normal cell cycle length) and two thousand binucleated cells per concentration (one thousand binucleated cells per culture; two cultures per concentration) were examined. All conditions were tested near the limit of test article toxicity evaluated by Cytokinesis-Block Proliferation Index (CBPI) levels.
Test article precipitate was observed in the test system only at the highest exposure concentration of 600 μg/mL. Since this concentration was toxic to the cells and not harvested, all analyzable concentrations of S3643 were entirely soluble in the test system. For the 3-h exposure experiment without S9, the concentrations 100, 200, 300, and 450 μg/mL yielded 1, 0, 25 and 93% of cells in cytostasis, respectively; concentrations of 0, 100, 200 and 300 μg/mL were analyzed for micronuclei. For the 19-h exposure without S9, the concentrations 16, 31, 63, 80 and 130 μg/mL yielded 10, 22, 50, 87 and 93% cells in cytostasis, respectively; concentrations of 0, 16, 31 and 63 μg/mL were analyzed for micronuclei. For the 3-h exposure experiment with S9, the concentrations 50, 100, 200 and 300 μg/mL yielded −1, −2, 29 and 91% cells in cytostasis, respectively; concentrations of 0, 50, 100 and 200 μg/mL were analyzed for micronuclei.
For all conditions, there was no significant increase in micronuclei (p > 0.01) observed in the S3643-treated cultures beyond those seen in the concurrent solvent controls. All positive controls induced significant numbers (p < 0.0001) of cells with micronuclei over the concurrent solvent controls. It was concluded that exposure to S3643 did not induce micronuclei in cultured Chinese hamster ovary cells under the conditions of the test.
3.4. In vivo toxicological studies
S3643 was evaluated in 28-day dose-range finding and 90-day subchronic toxicology studies in rats in compliance with the FDA guidelines [7] Toxicological Principles for the Safety of Food Ingredients (see Table 7 ). Summary data tables for the 28 and 90-day toxicology studies for S3643 can be found in the Supplemental Material.
| Study | Species/Gender (N value) | Dose | Findings |
|---|---|---|---|
| 28-day Dose Range Finding Toxicity Study | Male & Female Sprague-Dawley Rats | 10, 30, 100mg/kg bw/day (food ad-mix) | No test-article related findings; NOEL=100mg/kg bw/day |
| –5 animals/sex/group | |||
| 90-day Sub-Chronic Toxicity Study | Male & Female Sprague-Dawley Rats | 10, 30, 100mg/kg bw/day (food ad-mix) | Lower body weight gain in females at 100mg/kg/day |
| –20 animals/sex/group | No adverse test-article related findings; NOAEL=100mg/kg bw/day |
3.4.1. 28-day dose-range finding toxicity study
The purpose of this study was to evaluate the potential systemic toxicity of S3643 in rats after dietary administration for 28 days in order to select doses for the 90-day subchronic toxicity study in rats. Three treatment groups of five male and five female CD® [Crl:CD® (SD)] rats (Charles River Laboratories, Portage, MI), were administered the test article at dose levels of 10, 30, and 100 mg/kg bw/day. One additional group of five animals/sex served as the control and received untreated (vehicle) diet. The test substance was administered continuously via the diet throughout the 28 day treatment period. Dietary concentrations (ppm) of S3643 for each group were adjusted each week based on bodyweight and food consumption data, in order to achieve constant doses in terms of mg/kg bw/day. Survival, clinical observations, body weight, food consumption, clinical chemistry, ophthalmic examinations, organ weights, and macroscopic evaluations of all animals were used to assess potential toxicity. Ophthalmoscopic examinations were conducted pretest and prior to the terminal necropsy. Blood and urine samples for clinical pathology evaluations were collected from all animals prior to the terminal necropsy. At study termination, animals were sacrificed by carbon dioxide asphyxiation before subsequent exsanguination. Necropsy examinations were performed, organ weights were recorded, and only the liver was microscopically examined for animals treated at 0 and 100 mg/kg bw/day.
Once daily oral administration of S3643 for 28 days was well tolerated in rats at dose levels up to 100 mg/kg bw/day, the highest dose tested. There was no test article-related mortality observed and all animals survived until scheduled euthanasia. There were no test article-related clinical signs, or effects seen on ophthalmic examinations, hematology parameters, coagulation parameters, red blood cell morphology, clinical chemistry parameters, or urine chemistry parameters during this study. There appeared to be a slight (non-statistically significant) decrease in body weight gain in the female animals receiving the 30 and 100 mg/kg bw/day doses relative to the control group, which correlated with decreased food consumption. This was not observed in any of the male animal dose groups. There were no test article-related gross observations, changes in absolute or relative organ weights, or microscopic findings observed in the liver at study termination. Based on these results, the no-observed-effect level (NOEL) was considered to be 100 mg/kg bw/day for both male and female rats.
3.4.2. 90-day subchronic toxicity study
The purpose of this study was to evaluate the potential subchronic toxicity and toxicokinetic (TK) profile of the test article, S3643, in rats after dietary administration for 90 days. Compound was administered in the diet to four groups of twenty male and twenty female CD® [Crl:CD® (SD)] rats (Charles River Laboratories, Portage, MI) at dose levels of 0 (control), 10, 30, and 100 mg/kg bw/day. Additionally, one control group of three animals/sex and three treated groups of six animals/sex/group served as toxicokinetic (TK) animals and received the vehicle or test article diet in the same manner as the main study groups at respective dose levels of 0, 10, 30, and 100 mg/kg bw/day. The test substance was administered continuously via the diet throughout the treatment period. Dietary concentrations (ppm) of S3643 for each group were adjusted each week based on bodyweight and food consumption data, in order to achieve constant doses in terms of mg/kg bw/day. At the conclusion of the study, animals were sacrificed by carbon dioxide asphyxiation before subsequent exsanguination.
Survival, clinical observations, body weight gain, food consumption, hematology, clinical chemistry, urinalysis, organ weights, macroscopic examination, and histopathologic evaluation of at least 54 tissues were performed to assess potential toxicity (control and high dose animals only; see Supplementary Data for list of tissues examined histopathologically). A functional observational battery (including, but not limited to, evaluation of activity, arousal, autonomic and physical function, neuromuscular function, salvation, and respiration), motor activity assessment, and opthalmoscopic examinations were conducted pretest and again during 13th week of test article administration for all main study animals. Samples for hematology and clinical chemistry evaluations were collected from all the main study animals on Days 14 and 45, and again prior to termination. Urinalysis and samples for coagulation evaluations were collected prior to termination only. Blood for TK analysis was collected from one cohort of 3 animals/sex (control animals) at 1 h post the start of the dark cycle on Days 7 and 90. Samples were collected at alternating time points from 2 cohorts of 3 animals/sex (treated animals) at 1, 3, 6, 12, and 24 h post the start of the dark cycle on Days 7 and 90. At study termination, necropsy examinations were performed and organ weights were recorded for all main study animals and appropriate organ weight ratios were calculated (relative to body and brain weights). Microscopic examination of fixed hematoxylin and eosin-stained paraffin sections were performed on sections of tissues from the control and high-dose (100 mg/kg bw/day) groups. Livers were examined microscopically in all main study males and females at all dose levels.
Average daily compound consumption for animals given 10 mg/kg bw/day was 10.3 and 10.7 mg/kg bw/day; for 30 mg/kg bw/day, 30.8 and 30.9 mg/kg bw/day; and for 100 mg/kg bw/day, 104.4 and 102.3 mg/kg bw/day, for males and females, respectively. Formulation analysis demonstrated that the formulation preparation method produced homogeneous preparations (RSD < 20%).
Systemic exposure to S3643 was highly variable and appeared to be independent of sex following dietary administration of S3643 to male and female rats (see Table 8 ). Individual and mean plasma concentration data and female to male dose normalized AUC ratios were not indicative of any consistent gender difference (female to male exposure ratio ranged from 0.445 to 2.22). Therefore, the TK parameters were calculated from the combined mean plasma concentrations from both the male and female animals. Peak S3643 mean plasma concentrations were achieved by 6 h post the start of the dark cycle for the low and mid dose groups and 3 or 12 h post the start of the dark cycle for the high dose group on Days 7 and 90. Consistent with results from PK studies in rats, systemic exposure to S3643 was relatively high. For example, at 100 mg/kg bw/day, the combined mean Cmax on Day 90 was 10.40 μg/mL (31.7 μM), and the combined mean AUC0-24hr was 184.0 μg·h/mL. Systemic exposure (AUC0–24 h ) and Cmax values of S3643 generally increased with increasing dose in a greater than dose proportional manner on Day 7 and on Day 90. For example, a 1:2.95:9.55-fold increase in achieved daily dose resulted in an approximate 1:5.8:21.1-fold increase in AUC0–24 h and an approximate 1:7.5:22.5-fold increase in Cmax values on Day 7. Dose normalized systemic exposure (AUC0–24 h /Dose) appeared to be greater on Day 90 than on Day 7. Dose normalized accumulation ratios ranged from 1.35 to 1.86.
| Time Point | Nominal Dose (mg/kg bw) | Actual Dosea (mg/kg bw) | Cmax (ng/mL) | Tmax (hrs) | AUC0-24hr (ng·h/mL) | Cmax /dose (ng/mL/mg/kg) | AUC0–24 h /dose (ng·h/mL/mg/kg) | ACUM Ratiob |
|---|---|---|---|---|---|---|---|---|
| Day 7 | 10 | 8.63 | 276 | 6 | 4960 | 31.9 | 574 | – |
| 30 | 25.5 | 2060 | 6 | 29000 | 80.6 | 1140 | – | |
| 100 | 82.4 | 6230 | 12 | 105000 | 75.7 | 1280 | – | |
| Day 90 | 10 | 10.8 | 846 | 6 | 11500 | 78.3 | 1070 | 1.86 |
| 30 | 32.3 | 3160 | 6 | 50300 | 98.0 | 1560 | 1.37 | |
| 100 | 107 | 10400 | 3 | 184000 | 97.5 | 1730 | 1.35 |
a. Actual dose levels were determined using the weekly food consumption values and test article concentration in the diet. Individual values were averaged and the average daily dose achieved for Weeks 1 and 13 (Days 7 and 90) were used in toxicokinetic analysis.
b. ACUM Ratio = AUC0-24hr /dose at Day 90/AUC0-24hr /dose at Day 7.
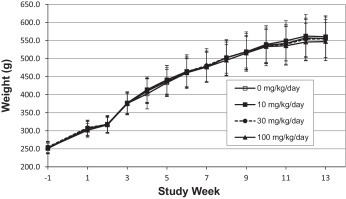
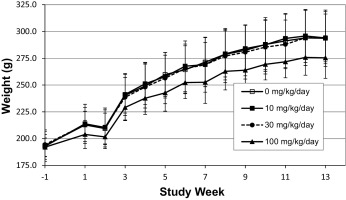
Mean body weight among main study females at 100 mg/kg bw/day was statistically significantly lower beginning at Week 5 (7% lower, p < 0.05) when compared to controls. The lower means coincided with decreased mean food consumption (6–14% lower, p < 0.05) and generally persisted during the dosing period. There were no corresponding changes in mean body weight or food consumption in the main study males at any dose level (see Fig. 5 and Fig. 6 ). These effects were considered test article-related but not adverse at these magnitudes.
|
|
|
Fig. 5. Mean body weights of male Sprague-Dawley rats (n = 20) receiving S3643 for 13 weeks. |
|
|
|
Fig. 6. Mean body weights of female Sprague-Dawley rats (n = 20) receiving S3643 for 13 weeks. |
There were no test article-related effects among hematology parameters, coagulation times, or clinical chemistry analytes in either sex at any dose level. All mean and individual values were considered within an acceptable range for biologic and/or procedure-related variation despite occasional mean values that reached statistical significance. There were no test article-related alterations observed among urinalysis parameters in either sex at any dose level. There were occasional differences found in urine volume and specific gravity that were not considered toxicologically meaningful due to their sporadic nature and the inherent variability of these endpoints.
There were no test article-related macroscopic changes or toxicologically significant test article-related organ weight changes noted at any dose level. Test article-related organ weight changes consisted of increased liver weights in males at 100 mg/kg bw/day relative to control animals (absolute liver weight: 12.5% increase, p > 0.05; liver/body weight: 14.1% increase, p < 0.01; liver/brain weight: 13.5% increase, p < 0.05). All other statistically significant organ weight changes were considered incidental/spontaneous as they were non dose-related, were related to body weight changes, and/or lacked microscopic correlates.
Test article‐related microscopic findings were limited to a minimal degree of liver centrilobular hepatocyte hypertrophy in three males at 100 mg/kg bw/day. This change consisted of a very slight increase in the size of the hepatocytes around the central vein of the liver which correlated to increased liver weights observed in males at this dose level as noted above. A malignant kidney tumor (amphophilic-vacuolar renal tubule carcinoma) was present in the kidneys of one female animal at 10 mg/kg/day. This tumor was considered incidental, as it occurred in the low dose group, there were no other renal tubule tumors in other animals, there were no pre-neoplastic renal lesions such as tubular hyperplasia in other animals, and thus was viewed to likely to be of spontaneous, possibly familial, origin. A benign uterus (cervix) smooth muscle tumor (leiomyoma) was also present in the same animal. This tumor was also considered incidental, as it occurred in the low dose group, there were no other uterus tumors in other animals, and there were no pre-neoplastic uterus lesions in other animals. All other microscopic observations were considered incidental/spontaneous as they were typical of rats of this age, similar in incidence across groups, and/or of low/isolated frequency.
Overall, there were no adverse test article-related effects noted for any parameter examined. As a result, the no-observed-adverse-effect-level (NOAEL) following 90 days of dietary administration was 100 mg/kg bw/day, the highest dose level tested, in male and female rats.
4. Discussion and conclusions
S3643 is a member of a novel series of substituted 1,2,4-triazole agonists of the human umami receptor hTAS1R1/hTAS1R3, which differs from previously reported oxalamide-based umami agonists mainly by the replacement of the oxalamide moiety by a 1,2,4-triazole ring. In CYP inhibition assays using spectrofluorimetric substrates with recombinant enzymes, S3643 exhibited significant inhibition of CYP1A2 and CYP3A4 (72% and 68% inhibition, respectively) at 10 μM. A structurally related oxalamide analog, N1 -(2,3-dimethoxybenzyl)-N2 -(2-(pyridin-2-yl)ethyl)oxalamide (S5456), was also found to inhibit CYP3A4 by 51% at 10 μM using the same assay format (Senomyx, unpublished results). However, neither compound significantly inhibited any of the CYP-isozymes (i.e. , <50% inhibition at 10 μM) when retested on the same panel of CYP enzymes in the more definitive assay format utilizing human liver microsomes and CYP-specific substrates. Given the low anticipated use levels of S3643 [1] , this level of CYP inhibition is not expected to interfere with normal metabolic processes. No other significant off-target activities were seen in any of the receptor profiling assays with 10 μM of either S3643 or S5456.
The PK profile of S3643 in male and female Sprague-Dawley rats is very similar to that of the previous reported oxalamide umami agonists S5456 and S336 (see Table 3 and Table 9 ). S3643 was rapidly eliminated after intravenous administration in both male and female rats with mean terminal t1/2 of 0.19 and 0.16 h, respectively. S5456 was also rapidly eliminated after intravenous administration of a 1.0 mg/kg bw dose with a mean terminal t1/2 of 0.32 h in both male and female rats (Chi, unpublished results). Likewise, S336 at an intravenous dose of 0.1 mg/kg bw, was also rapidly cleared with a mean terminal t1/2 of 0.42 h in male rats [11] .
| Cmpd | Route | Dose (mg/kg bw) | Sex | Cmax (ng/mL) | Tmax (h) | t1/2 (h) | AUC0-last (ng·h/mL) | % F |
|---|---|---|---|---|---|---|---|---|
| S5456a | iv | 1.0 | M | 2633±363 | 0.03 | 0.32 | 757±112 | – |
| F | 2398±391 | 0.03 | 0.32 | 817±186 | – | |||
| oral gavage | 10 | M | 3120±787 | 0.25 | 0.95 | 3001±498 | 39.6% | |
| F | 3258±1087 | 0.25 | 1.20 | 4201±1013 | 51.4% | |||
| 30 | M | 7643±2404 | 0.31 | 1.09 | 9535±4190 | 42.0% | ||
| F | 10980±1500 | 0.33 | 1.59 | 26610±7306 | 108.6% | |||
| 100 | M | 33070±14550 | 0.25 | 1.35 | 55950±19820 | 73.9% | ||
| F | 34670±4196 | 0.92 | 2.92 | 204400±44540 | 250.2% | |||
| S336b | iv | 0.1 | M | 207±25.8 | 0.03 | 0.42 | 65.0±1.3 | – |
| oral gavage | 10 | M | 7027±925 | 0.25 | 0.69 | 5809±444 | 89.4% | |
| 30 | M | 23430±1436 | 0.19 | 0.71 | 22260±727 | 114.2% | ||
| 100 | M | 36270±6004 | 0.25 | 0.98 | 87490±13350 | 134.6% |
a. PK study of S5456 used 4 animals/sex for intravenous dosing, and 3 animals/sex/group for oral dosing.
b. PK study of S336 used 4 male rats for intravenous dosing, and 3 male rats/group for oral dosing.
The PK of S3643, S5456, and S336 has been studied at oral doses of 10, 30, and 100 mg/kg bw. The plasma t1/2 values for all three compounds tended to increase with increasing oral dose. For S3643, t1/2 ranged from 0.89 to 1.66 h in female rats and 0.80 to 1.66 h in male rats. The t1/2 values for S5456 ranged from 1.20 to 2.92 h in female rats and 0.95 to 1.35 h in male rats. Similar results were seen with S336 with t1/2 ranging from 0.69–0.98 h in male rats [11] . The Cmax of S3643 at the 100 mg/kg bw dose was 78.3 μg/mL (238 μM) in male rats and 98.9 μg/mL (310 μM) in females. The Cmax at the 100 mg/kg oral dose of S5456 was similar in both male and female rats ranging from 33.1–34.7 μg/mL (96.3–101 μM); the Cmax for S336 at this dose in male rats was 36.3 μg/mL (106 μM).
For all three compounds, AUClast increased in a greater than dose proportional manner. This was particularly true for S5456 where a 10-fold increase in oral dose led to a 48.7-fold increase in AUClast in female animals and an 18.6-fold increase in males. For S336, which was only studied in male rats, a 10-fold increase in oral dose led to 15.1-fold increase in AUClast . For S3643, a greater than dose proportional increase was only seen in female animals where a 10-fold increase in oral dose led to a 16.8-fold increase in AUClast ; for males, a less than dose proportional increase (6.88-fold) was observed. With both S3643 and S5456, a greater than dose proportional increase in mean AUC0–24 h was also seen in the TK studies associated with the 90-day subchronic toxicity study with S3643 and a 28-day subacute study conducted on S5456. As was seen in the single dose PK studies, the exposure of S5456 was significantly higher (2–3 fold) in female rats than in males on both Day 1 and Day 28 (Senomyx, unpublished results).
The non-linear kinetics seen with oral doses of both S3643 and the oxalamides is largely driven by a significant decrease in plasma clearance at higher doses which is indicative of saturation of an elimination mechanism. It is not known whether this dose-dependent decrease in intrinsic clearance is due to a saturation of first pass hepatic metabolism or saturation of some other elimination mechanism such as active renal transport. However, the larger increases in AUC’s seen with S5456 and S3643 in female rats relative to males, suggests that the non-linear kinetics may be due to saturation of hepatic metabolism. Gender-dependent metabolism of xenobiotics is a well-known phenomenon in rats that has been attributed to differences in the profile of cytochrome P450 isozymes found in male and female rat liver [12] and [19] . It is possible one or more of the CYP enzymes involved in the metabolism of these compounds is less abundant in female rats, and becomes saturated at higher compound concentrations. If this metabolic pathway is rate-limiting for the elimination of the compound, this could result in significantly lower intrinsic clearance and higher AUCs in female rats versus males.
S3643 was evaluated for its genotoxic potential through a standard battery of in vitro genotoxicity assays which included a bacterial reverse mutation assay (S. typhimurium strains TA98, TA100, TA1535, TA1537 and E. coli strain WP2 uvrA ), a chromosome aberration test in HPBL, and a micronucleus test in CHO-WBL cells. S3643 was found to be neither mutagenic, clastogenic, nor aneugenic in these in vitro genotoxicity assays. Oxalamide S5456 has also been shown to be non-mutagenic in a bacterial reverse mutation assay using the same tester strains at concentrations up to 5000 μg/plate with and without metabolic activation (Senomyx, unpublished results). In a plate incorporation assay, oxalamide S336 was found to be weakly positive to TA1535 in the absence of metabolic activation, and weakly positive to TA100 in a pre-incubation assay in the presence of metabolic activation. However, in neither case was a dose response observed and S336 consistently tested negative in the other tester strains under all conditions at concentrations of up to 5000 μg/plate. Given the lack of a dose-dependent response and the fact that the mean number of revertants was below historical spontaneous reversion or negative control values, it was concluded that S336 was non-mutagenic [5] .
S336 was also found to be non-clastogenic in a chromosomal aberration test in CHO-WBL cells in the presence and absence of metabolic activation at concentrations up to 5000 μg/mL [5] . S336 was also evaluated for clastogenic activity and/or interference with the spindle apparatus in an in vivo mouse micronucleus assay. S336 was administered by intraperitoneal (ip) injection to male Swiss albino (CD-1) mice (21 animals/dose group) at doses up to 800 mg/kg bw (maximum tolerated dose). At 24, 36, and 48 h following dose administration, 7 animals from each group were sacrificed, and their femoral bone marrow was harvested, fixed and stained. There were no statistically significant differences observed in the number of polychromatic erythrocytes with micronuclei or reductions in the ratio of polychromatic erythrocytes to total erythrocytes between the S336 treated groups and the negative control [5] . Overall, the results of the genotoxicity studies conducted on S3643 and the oxalamide umami agonists S5456 and S336 indicate no safety concern for these substances with respect to genotoxicity.
The doses of S3643 selected for the 28- and 90-day toxicology studies were designed to provide a high margin of safety rather than define a maximum tolerated dose (MTD) in rats. Applying a 1000-fold margin of exposure in extrapolating animal data to humans to account for species differences in susceptibility, numerical differences in population ranges between the test animals and the human population, the greater variety of complicating disease processes in the human population, and the possibility of synergistic action among food additives, is believed to be an adequate margin of safety for most substances proposed for use in food [4] and [16] . Based on the anticipated annual volume of use (2000 kg), the per capita intake (“eaters only”) of S3643 for use as a flavour ingredient was calculated to be 295 μg/person/day (5 μg/kg bw/day) [1] . Therefore, based on the low anticipated use level of S3643, a NOAEL of 100 mg/kg bw/day in a sub-chronic toxicology study would provide over a 20,000-fold margin of safety.
In the 90-day subchronic toxicology study with S3643, the mean body weight of the female rats at 100 mg/kg bw/day was statistically significantly lower beginning at Week 5 (7% lower, p < 0.05) when compared to controls which correlated with decreased mean food consumption (6–14% lower, p < 0.05) throughout the dosing period. There were no corresponding changes in mean body weight or food consumption in the main study males at any dose level. These effects were considered test article-related but not adverse at these magnitudes. There were no test article-related effects among clinical signs, ophthalmic examinations, hematology parameters, coagulation times, clinical chemistry parameters, or urinalysis parameters, or in the functional observation battery in either sex at any dose level. Test article-related organ weight changes consisted of increased liver weights in males at 100 mg/kg bw/day relative to control animals which correlated to a minimal degree of centrilobular hepatocyte hypertrophy. Centrilobular hepatocyte hypertrophy is usually representative of enzyme induction, which is considered an adaptive response [9] and therefore not considered to be adverse. The 90-day subchronic toxicity study established a NOAEL for S3643 of 100 mg/kg bw/day (the highest dose evaluated), for both male and female Sprague-Dawley rats.
The potential for in vivo toxicity of two of the structurally related oxalamide umami agonists has also been evaluated in rats. The toxicity of S5456 was studied in male and female Crl:CD (SD) rats (10 animals/sex/group) at doses up to 140 mg/kg bw/day administered as a suspension in 1% MC by oral gavage for 28 days. Similarly, the toxicity of S336 was evaluated in male and female Crl:CD (SD) IGS BR rats (20 animals/sex/group) via feeding in the diet at nominal doses up to 100 mg/kg bw/day for 92 or 93 days. With both S5456 and S336, there were no test article-related effects among clinical signs, body weights, ophthalmic examinations, hematology parameters, coagulation times, clinical chemistry parameters, or urinalysis parameters, or in the functional observation battery in either sex at any dose level. There were no test article-related organ weight, macroscopic or microscopic changes in the tissues examined noted at any dose level. The 28-day short term toxicity study established a NOEL for S5456 of 140 mg/kg bw/day (the highest dose evaluated), for both male and female Sprague-Dawley rats (Senomyx, unpublished results). Likewise, the NOEL in rats administered S336 in diet for 92 or 93 days was at least 100 mg/kg bw/day [5] .
According to the Maximized Survey-derived Daily Intake (MSDI) approach, the margin of safety for S336 based on the estimated daily per capita intake of this substance in both Europe and the USA was viewed to be adequate and it was not anticipated to pose a safety concern when used as a flavouring substance at the estimated levels of intake [5] . Based on a daily per capita intake estimate of 295 μg/person/day [1] and its NOAEL in the 90-day subchronic toxicity study in rats, S3643 is also believed not to pose a safety hazard when used as a flavouring substance.
In conclusion, S3643 demonstrated a lack of genotoxicity with or without metabolic activation in vitro at concentrations that greatly exceed those observed in rat plasma following oral administration of S3643 at doses up 100 mg/kg bw. The results of a 90-day subchronic toxicity study established NOAEL for S3643 of 100 mg/kg bw/day (the highest dose evaluated), for male and female rats. Assuming that the systemic exposure of S3643 after oral administration to humans is comparable to that observed at an equivalent dose in the rat, this NOAEL is several orders of magnitude higher than the anticipated human exposure for S3643 under the conditions of intended use [1] .
Conflict of interest
All the studies described herein were funded by Senomyx, Inc.
Transparency document
Transparency Document.
Acknowledgements
The authors wish to thank Ginger Toschiaddi of Senomyx, Inc., for carrying out the in-life phase of the pharmacokinetic studies on S3643. The authors would also like to thank Dr. Joseph Fotsing and Qing Chen for preparing synthetic samples of the potential metabolites of S3643.
Appendix A. Supplementary data
The following are Supplementary data to this article:
References
- [1] S.M. Sohen, S. Fukushima, N.J. Gooderham, S.S. Hecht, L.J. Marnett, I.M.C.M. Rietjens, R.L. Smith, GRAS Flavouring Substances 27: The 27th publication by the Expert Panel of the Flavour and Extract Manufacturers Association provides an update on recent progress in the consideration of flavouring ingredients generally recognized as safe under the Food Additive Amendment. Food Technology, 69(8) 40–59 (Key findings of the FEMA Expert Panel GRAS determinations for each substance on the GRAS 27 list are available online: www.femaflavor.org ) (2015).
- [2] C.C. Crespi, C.L. Miller, B.W. Penman; Mitrotiter plate assays for inhibition of human: drug-metabolizing cytochromes P450; Anal. Biochem., 248 (1997), pp. 188–190
- [3] B. Davies, T. Morris; Physiological parameters in animals and humans; Pharm. Res., 10 (1993), pp. 1093–1095
- [4] EFSA; EFSA panel on food contact materials, enzymes, flavourings and processing aids. 2010. Draft guidance on the data required for the risk assessment of flavourings; EFSA J., 8 (6) (2010), p. 1623 Available online www.efsa.europa.eu
- [5] EFSA CEF Panel, Scientific Opinion on Flavouring Group Evaluation 94, Revision 2 (FGE.94Rev2): Consideration of aliphatic amines and amides evaluated in an addendum to the group of aliphatic and aromatic amines and amides evaluated by the JECFA (68th meeting), EFSA Journal 12(4) (2014), 3622.
- [6] FDA, Good Laboratory Practice for Nonclinical Laboratory Studies, 21 CFR Part 58, National Archives and Records Administration (2006).
- [7] FDA, Guidance for Industry and Other Stakeholders. Toxicological Principles for the Safety Assessment of Food Ingredients. Redbook 2000 (2010).
- [8] J.B. Hallagan, R.L. Hall; Under the conditions of intended use—new developments in the FEMA GRAS program and the safety assessment of flavour ingredients; Food Chem. Toxicol., 47 (2009), pp. 267–278
- [9] A.P. Hall, C.R. Elcombe, J.R. Foster, T. Harada, W. Kaufmann, A. Knippel, K.D. Küttler, E. Malarkey, R.R. Maronpot, A. Nishikawa, T. Nolte, A. Schulte, V. Strauss, M.J. York; Liver hypertrophy: a review of adaptive (adverse and non-adverse) changes conclusions from the 3rd international ESTP expert workshop; Toxicol. Pathol., 40 (2012), pp. 971–994
- [10] ICH, ICH Harmonised Tripartite Guideline: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients-Q7 (2000).
- [11] JECFA; (Joint FAO/WHO Expert Committee on Food Additives) (2011). Safety Evaluation of Certain Food Additives and Contaminants. Seventy-third Meeting of the Joint FAO/WHO Expert Committee on Food Additives, WHO Food Additives Series: 64. WHO, Geneva 2011; http://www.inchem.org/documents/jecfa/jecmono/v64je01.pdf
- [12] R. Kato, Y. Yamazoe; Sex-specific cytochrome P450 as a cause of sex- and species-related differences in drug toxicity; Toxicol. Lett., 64/65 (1992), pp. 661–667
- [13] X. Li, L. Staszewski, H. Xu, K. Durick, M. Zoller, E.A. Alder; Human receptors for sweet and umami taste; PNAS, 99 (7) (2002), pp. 4692–4696
- [14] X. Li; T1R receptors mediate sweet and umami taste; Am. J. Clin. Nutr., 90 (suppl) (2009), pp. 733S–737S
- [15] T. Lin, K. Pan, J. Mordenti, L. Pan; In vitro assessment of cytochrome P450 inhibition: strategies for increasing LC/MS-based assay throughput using one-point IC50 method and multiplexing high-performance liquid chromatography ; J. Pharm. Sci., 96 (9) (2007), pp. 2485–2493
- [16] B. Magnuson, I. Munro, P. Abbot, N. Baldwin, R. Lopez-Garcia, K. Ly, L. McGirr, A. Roberts, S. Socolovsky; Review of the regulation and safety assessment of food substances in various countries and jurisdictions; Food Addit. Contam. Part A, 30 (7) (2013), pp. 1147–1220
- [17] L.J. Marnett, S.M. Cohen, S. Fukushima, N.J. Gooderham, S.S. Hecht, I.M.C.M. Rietjens, R.L. Smith, T.B. Adams, J.B. Hallagan, C. Harman, M.M. McGowen, S.V. Taylor; GRAS Flavouring Substances 26: the 26th publication by the Expert Panel of the Flavour and Extract Manufacturers Association provides an update on recent progress in the consideration of flavouring ingredients generally recognized as safe under the Food Additive Amendment; Food Technol., 67 (8) (2013), pp. 38–56
- [18] V.P. Miller, D.M. Stresser, A.P. Blanchard, S. Turner, C.L. Crespi; Fluorometric high-throughput screening for inhibitors of cytochrome P450; Ann. N. Y . Acad. Sci., 919 (2000), pp. 26–32
- [19] C.A. Mugford, G.L. Kedderis; Sex-dependent metabolism of xenobiotics; Drug Metab. Rev., 30 (3) (1998), pp. 441–498
- [20] OECD; Guideline Test No. 471: Bacterial Reverse Mutation Test. In: OECD Guidelines for the Testing of Chemicals; Organisation for Economic Co-operation and Development, Paris (1997)
- [21] OECD; Guideline Test No. 473: In Vitro Mammalian Chromosome Aberration Test. In: OECD Guidelines for the Testing of Chemicals; Organisation for Economic Co-operation and Development, Paris (1997)
- [22] OECD; Principles on good laboratory practice (as revised in 1997); OECD Series on Principles of Good Laboratory Practice and Compliance Monitoring, Number 1, OECD Environmental Health and Safety Publications, Environment Directorate, Organisation for Economic Co-operation and Development, Paris (1998)
- [23] OECD; Guideline test no. 487: In vitro mammalian cell micronucleus test ; OECD Guidelines for the Testing of Chemicals, Organisation for Economic Co-operation and Development, Paris (2010)
- [24] C. Tachdjian, X. Li, M. Qi, M. Rinnova, G. Servant, M. Zoller, Flavors, Flavor Modifiers, Tastants, Taste Enhancers, Umami or Sweet Tastants, and/or Enhancers and Use Thereof. US Patent No. 7, 476, 399 B2, issued January 13, 2009 (2009).
- [25] C. Tachdjian, A.P. Patron, S.L. Adamski-Werner, F. Bakir, Q. Chen, Darmohusodo Hobsen, S.T. Li, X. Qi, M. Rogers, D.H. Rinnova, M. Servant, G. Tang, X.-Q. Zoller, M. Wallace, D. Xing, A. Gubernator, Flavors, Flavor Modifiers, Tastants, Taste Enhancers, Umami or Sweet Tastants, and/or Enhancers and Use Thereof. US Patent No. 8, 124, 121 B2, issued February 28, 2012 (2012).
- [26] C. Tachdjian, A.P. Patron, S.L. Adamski-Werner, F. Bakir, Q. Chen, Darmohusodo Hobsen, S.T. Li, X. Qi, M. Rogers, D.H. Rinnova, M. Servant, G. Tang, X.-Q. Zoller, M. Wallace, D. Xing, A. Gubernator, Flavors, Flavor Modifiers, Tastants, Taste Enhancers, Umami or Sweet Tastants, and/or Enhancers and Use Thereof. US Patent No. 8, 895, 050 B2, issued November 25, 2014 (2014a).
- [27] C. Tachdjian, M. Lebl-Rinnova, D. Wallace, Compounds Comprising Linked Heteroaryl Moieties and Their Use as Novel Umami Flavor Modifiers, Tastants and Taste Enhancers for Comestible Compositions. US Patent No. 8, 784, 782 B2, issued July 22, 2014 (2014b).
- [28] C. Tachdjian, M. Lebl-Rinnova, D. Wallace, Compounds Comprising Linked Heteroaryl Moieties and Their Use as Novel Umami Flavor Modifiers, Tastants and Taste Enhancers for Comestible Compositions. US Patent No. 8, 968, 708 B2, issued March 3, 2015 (2015).
- [29] H. Tao, D. Santa Ana, A. Guia, M. Huang, J. Ligutti, G. Walker, K. Sithiphong, F. Chan, T. Guoliang, Z. Zozulya, S. Saya, R. Phimmachack, C. Sie, J. Yuan, L. Wu, J. Xu, A. Ghetti; Automated tight seal electrophysiology for assessing the potential hERG liability of pharmaceutical compounds; Assay Drug Dev. Technol., 2 (5) (2004), pp. 497–506
- [30] S. Zambon, S. Fontana, M. Kajbaf; Evaluation of cytochrome P450 inhibition assays using human liver microsomes by a cassette analysis/LC-MS/MS; Drug Metab. Lett., 4 (3) (2010), pp. 120–128
- [31] F. Zhang, B. Klebansky, R.M. Fine, H. Xu, A. Pronin, H. Liu, C. Tachdjian, X. Li; Molecular mechanism for umami taste synergism; PNAS, 106 (52) (2008), pp. 20930–20934
Document information
Published on 02/05/17
Accepted on 02/05/17
Submitted on 02/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?