Abstract
Background
Pulmonary hypertension (PH) is a serious complication in patients with systemic scleroderma (SSc), therefore it is important to identify the factors that could predict the presence and progression of PH. Skin biopsy is performed in patients with SSc to examine the type and severity of the disease. MicroRNAs (miRNAs) are potential biomarkers for various cardiovascular diseases including PH.
Methods and results
We determined the skin miRNA expression profile in 15 SSc patients with (n = 6) and without PH (n = 9). A mixture of equal amounts of miRNAs from PH and non-PH patients were prepared and used for miRNA PCR array analysis. The analysis identified 591 upregulated miRNAs and 57 downregulated miRNAs in the PH group. Of these, only miRNAs with a Ct value of less than 35 were subjected to further analysis. When a 1.5-fold difference was considered meaningful, 32 miRNAs were upregulated and 14 miRNAs were downregulated in the PH group. Interestingly, 5 out of 14 downregulated miRNAs belonged to the let-7 family. The results were validated by quantitative real-time PCR with specific primer for each miRNA, which showed significant downregulation of five let-7 family members (let-7a, -7d, -7e, -7f, -7g) in 6 PH compared with 9 non-PH skin samples. The expression levels of let-7d and 7b correlated negatively with pulmonary arterial pressure measured by echocardiography.
Conclusions
The results suggest that skin miRNA is a potentially useful marker for the presence and severity of PH in patients with SSc.
Keywords
Skin biopsy;MiRNA;Pulmonary hypertension;Systemic scleroderma
1. Introduction
Pulmonary hypertension (PH) is one of the most serious complications in patients with systemic scleroderma (SSc). Meta-analysis indicates that the prevalence of PH is 5 to 14% among SSc patients [1]. The presence of PH worsens prognosis in these patients [2], and thus it is important clinically and therapeutically to identify new factors and biomarkers that can predict the presence and progression of PH.
A number of clinical factors and biomarkers have been implicated in SSc-associated PH [3]; [4] ; [5]. Recently, much attention has been paid to microRNAs (miRNAs) as a potential biomarker for PH [6]. miRNA is a short non-cording RNA known to act as a negative regulator of various transcription factors [7]. miRNA was originally thought to function only intracellularly, but recent data suggest that it is also secreted and detected in the circulation [7]. Various miRNAs have been identified to be involved in the pathogenesis of PH [8] and circulating miRNA levels vary according to the severity of PH [9].
In patients with SSc, skin biopsy is performed to examine the type and severity of the disease. Although this procedure is not always required for diagnosis, skin biopsy samples are important to rule out other fibrosing diseases. We have previously shown that miRNA profiling in skin samples was useful to investigate the functional role of miRNAs in the development of SSc [10] ; [11]. However, the roles of miRNAs in the pathogenesis of SSc-associated PH have not been thoroughly investigated. In this study, we used skin biopsy samples to test the hypothesis that skin miRNAs expression profile varies among patients with SSc and depends on the presence or absence of PH.
2. Materials and methods
2.1. Study population
Data of 15 SSc patients who underwent skin tissue biopsy were available for analysis. They were divided into PH (n = 6) and non-PH groups (n = 9) according to the estimated pulmonary arterial pressure measured by ultrasound echocardiography (UCG). PH was defined as tricuspid valve regurgitation pressure gradient (TRPG) of more than 40 mm Hg. The echocardiographic findings of all study participants using Aplio XG (Toshiba, Tokyo, Japan) ultrasound systems were evaluated by two independent investigators who were blinded to the clinical data. The clinical and laboratory data reported in this study were obtained at the time of skin sampling. The study protocol was approved by the Human Ethics Review Committee of Kumamoto University and a signed consent form was obtained from each subject.
2.2. miRNA isolation, PCR array and real-time PCR
The miRNA was isolated from human skin tissue using the miRNeasy FFPE kit (Qiagen, Hilden, Germany). For PCR array, the miRNAs were reverse transcribed into first-strand cDNA using an RT2 miRNA First-Strand Kit (Qiagen). The cDNA was mixed with QuantiTast SYBR Green PCR Master Mix, and the mixture was added into 384-well RT2 miRNA PCR Array that included primer pairs for 1066 human miRNAs (Qiagen). For real-time PCR, the primers and templates were mixed with SYBR Advantage Quantitative PCR Premix (Clontech, Mountain View, CA). The DNA was amplified for 40 cycles of denaturation for 5 s at 95 °C and annealing for 20 s at 60 °C on C1000 Thermal Cycler (CFX384, Bio-Rad Laboratories, Hercules, CA). The transcript levels were normalized to those of U6 in the same sample.
2.3. Statistical analysis
Data of two groups were compared for statistical significance by the Student t test. Correlations were assessed by Pearsons correlation coefficient. A p value of < 0.05 denoted the presence of a statistically significant difference.
3. Results
Table 1 shows the clinical characteristics of the study participants. All participants were female. There was no difference in age, body mass index, and blood pressure between the PH and non-PH groups. Auto-immune antibody, renal function, and high-sensitive CRP levels were not different between the two groups. Plasma BNP level was significantly higher in the PH group than in the non-PH group. Left ventricular function and dimension were not different between the two groups.
| Non-PH group (n = 9) | PH group (n = 6) | p value | |
|---|---|---|---|
| Age (years) | 73.0 ± 0.8 | 70.8 ± 4.7 | 0.59 |
| Females | 9 (100%) | 6 (100%) | |
| Body mass index (kg/m2) | 23.6 ± 1.9 | 24.0 ± 2.6 | 0.93 |
| Systolic blood pressure (mm Hg) | 147.3 ± 4.8 | 137.2 ± 9.1 | 0.50 |
| Diastolic blood pressure (mm Hg) | 81.7 ± 2.8 | 73.2 ± 4.8 | 0.30 |
| Rheumatic factor | 15.1 ± 6.3 | 21.2 ± 3.0 | 0.51 |
| Ribonucleoprotein (U/mL) | 1.09 ± 0.19 | 1.06 ± 0.24 | 0.93 |
| Scl-70 (U/mL) | 2.73 ± 0.82 | 3.54 ± 2.40 | 0.72 |
| Anti-centromere antibody (U/mL) | 97.4 ± 33.8 | 117.9 ± 40.1 | 0.71 |
| Brain natriuretic peptide (pg./mL) | 36.8 ± 8.6 | 315.9 ± 121.3 | 0.01 |
| hs-CRP (mg/dL) | 0.07 (0.01–0.24) | 0.2 (0.04–0.49) | 0.07 |
| Creatinine (mg/dL) | 0.55 ± 0.04 | 0.65 ± 0.06 | 0.19 |
| eGFR | 78.3 ± 4.0 | 69.2 ± 6.0 | 0.21 |
| HbA1c (%) | 5.8 ± 0.1 | 5.7 ± 0.1 | 0.69 |
| LVEF (%) | 68.2 ± 0.9 | 65.5 ± 2.80 | 0.29 |
| LVDd (mm) | 40.2 ± 1.4 | 38.1 ± 0.7 | 0.33 |
| Left atrium diameter (mm) | 31.6 ± 1.7 | 34.7 ± 2.6 | 0.33 |
| E/e′ | 11.0 ± 1.4 | 15.3 ± 4.3 | 0.28 |
| Pulmonary arterial pressure (mmHg) | 27.4 ± 0.87 | 66.7 ± 7.94 | < 0.01 |
Data are mean ± SEM, median (range) or (percentages).
Scl-70, anti-scleroderma antibody; hs-CRP, high-sensitivity C-reactive protein; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; LVDd, left ventricular diameter diastolic phase.
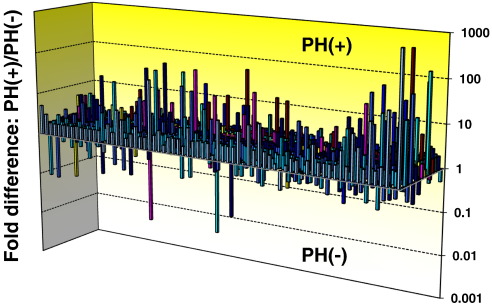
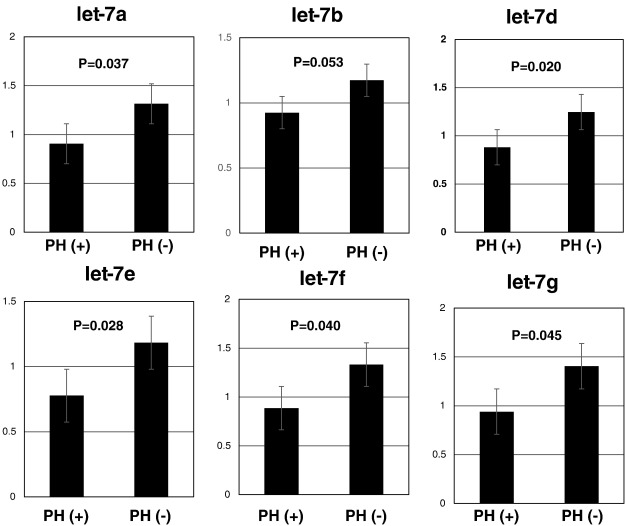
For comprehensive evaluation of the skin miRNA expression profile, we performed miRNA PCR array analysis using a mixture of equal amounts of miRNAs from 6 PH as well as 6 non-PH skin samples. As shown in Fig. 1, 591 miRNA were upregulated and 57 miRNAs were downregulated in the PH group. Of these, only miRNAs with Ct values less than 35 were considered for further analysis, and 1.5-folds up- or down-regulated miRNAs were selected. Table 2 lists the selected miRNAs. The expression of these miRNAs was validated by quantitative real-time PCR and the results showed significant downregulation of let-7a, 7d, 7e, 7f, 7g in the PH group (Fig. 2).
|
|
|
Fig. 1. The 3D-profile graphs of fold differences in the expression levels of each gene between non-PH and PH samples in the PCR Array. Columns pointing up (with z-axis values > 1) indicate up-regulation of gene expression, and columns pointing down (with z-axis values < 1) indicate down-regulation of gene expression in the PH samples, relative to the non-PH samples. |
| miRNA ID | Fold up-regulation | miRNA ID | Fold down-regulation |
|---|---|---|---|
| PH(+)/PH(−) | PH(+)/PH(−) | ||
| hsa-miR-1909* | 26.99 | hsa-miR-92a | − 1.57 |
| hsa-miR-221 | 8.60 | hsa-miR-767-5p | − 1.69 |
| hsa-miR-124 | 7.30 | hsa-miR-370 | − 1.81 |
| hsa-miR-3173-3p | 4.22 | hsa-let-7b | − 1.90 |
| hsa-miR-532-3p | 3.49 | hsa-miR-628-3p | − 1.92 |
| hsa-miR-1307 | 3.29 | hsa-let-7d | − 1.98 |
| hsa-miR-4286 | 2.60 | has-let-7f | − 2.05 |
| hsa-miR-205 | 2.55 | hsa-miR-433 | − 2.08 |
| hsa-miR-328 | 2.48 | hsa-miR-23a | − 2.13 |
| hsa-miR-200c | 2.39 | hsa-let-7a | − 2.19 |
| hsa-miR-3647-3p | 2.25 | hsa-miR-203 | − 2.39 |
| hsa-miR-200b | 2.23 | hsa-miR-199a-3p | − 2.49 |
| hsa-miR-23b | 2.08 | hsa-let-7 g | − 2.64 |
| hsa-miR-4301 | 2.08 | hsa-miR-30c | − 2.77 |
| hsa-miR-3607-5p | 2.06 | ||
| hsa-miR-421 | 2.04 | ||
| hsa-miR-24 | 1.99 | ||
| hsa-miR-125b | 1.91 | ||
| hsa-miR-125a-5p | 1.82 | ||
| hsa-miR-1247 | 1.81 | ||
| hsa-miR-27b | 1.79 | ||
| hsa-miR-490-3p | 1.75 | ||
| hsa-miR-1280 | 1.67 | ||
| hsa-miR-365 | 1.66 | ||
| hsa-miR-30b | 1.65 | ||
| hsa-miR-1260 | 1.60 | ||
| hsa-miR-7-2* | 1.60 | ||
| hsa-miR-720 | 1.60 | ||
| hsa-miR-3607-3p | 1.59 | ||
| hsa-let-7c | 1.58 | ||
| hsa-miR-27a | 1.51 | ||
| hsa-miR-126 | 1.50 |
|
|
|
Fig. 2. Let-7 family miRNA expression in non-PH (n = 9) and PH (n = 6) groups. Results are mean ± SEM. |
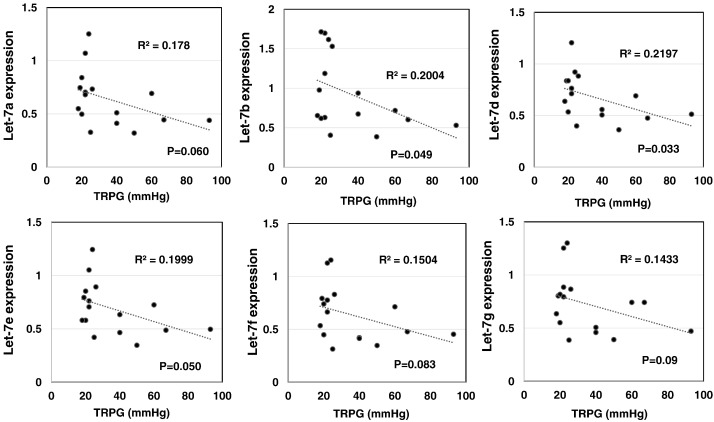
Next, the correlation between the expression levels of these miRNAs and the estimated pulmonary arterial pressure measured by UCG was assessed. The expression of let-7b and -7d correlated significantly and negatively with the estimated pulmonary arterial pressure (Fig. 3). The expression of let-7a, -7e, -7 g, -7f tended to correlate negatively with the estimated pulmonary arterial pressure, albeit insignificantly.
|
|
|
Fig. 3. Negative correlation between let-7 family miRNA expression and tricuspid valve regurgitation pressure gradient (TRPG). |
4. Discussion
In this study, we demonstrated that the expression level of let-7 family microRNAs in skin was lower in SSc patients with PH than those without PH. Furthermore, the expression of let-7 family miRNAs trended to correlate negatively with the estimated pulmonary arterial pressure. Especially, let-7b and -7d expression levels correlated significantly and negatively with the severity of PH in patients with SSc. These results indicate that skin miRNA levels could be a useful marker for the presence and severity of PH in patients with SSc.
Skin biopsy is performed even in patients with cardiovascular diseases to rule out the presence of secondary cardiomyopathies. Skin tissue biopsy is a less invasive procedure compared with endomyocardial biopsy, and is recognized as an informative screening technique for these diseases. We have previously shown that skin biopsy is a reliable and safe method for evaluation of amyloid deposition in patients with cardiac amyloidosis [12]. Similarly, skin miRNA is used as a biomarker of several skin diseases. For example, miRNA-205 was down-regulated in patients with dermatofibrosarcoma protuberans [13]. Another study showed that miR-7 level was significantly decreased in localized scleroderma [14]. The present study demonstrated the potential usefulness of skin miRNA as a marker for the presence and severity of PH in patients with SSc. Because skin biopsy is performed to examine the type and severity the disease in patients with collagen diseases, determination of skin miRNA expression level could be an additional tool to evaluate disease severity in these patients.
The Let-7 family miRNAs were the first discovered human miRNAs [15]. To our knowledge, there are only three publications on the relationship between let-7 family and PH [11]; [16] ; [17]. In these studies, let-7 miRNAs were significantly downregulated in lung tissue in an in vivo model of PH, in vitro smooth muscle cells, and in the sera of patients with chronic thromboembolic PH. Consistent with these findings, we observed low let-7 miRNA expression in skin samples of patients with SSs-associated PH in this study. The let-7 microRNA is known to repress cell proliferation [18], and knockdown of let-7d enhanced pulmonary fibrosis [19]. Therefore, low expression of skin let-7 family in the PH group could probably reflect systemically attenuated anti-fibrotic properties.
The present study has certain limitations. First, the study included only a small number of patients recruited at a single center. Further multicenter studies of larger populations are needed to confirm the present results. Second, we evaluated PH only by UCG. Right heart catheterization should be performed for accurate measurement of pulmonary artery pressure. Third, the present study could not evaluate the prognostic value of skin let-7 family expression levels due to the small number of patients enrolled in this study.
5. Conclusion
Skin miRNA is a potentially useful marker for evaluation of PH in patients with SSc. Skin biopsy can be considered in patients with collagen diseases to identify the presence and severity of PH.
Conflicts of interest
None.
Acknowledgments
The authors are grateful to Ms. Chiemi Shiotsu from the Department of Dermatology and Plastic Surgery, and Saeko Tokunaga, Megumi Nagahiro, and Ayuko Tateishi from the Department of Cardiology, Graduate School of Medical Sciences, Kumamoto University for the excellent technical assistance.
References
- [1] J. Avouac, P. Airo, C. Meune, L. Beretta, P. Dieude, P. Caramaschi, et al.; Prevalence of pulmonary hypertension in systemic sclerosis in European Caucasians and metaanalysis of 5 studies; J. Rheumatol., 37 (2010), pp. 2290–2298
- [2] E.T. Koh, P. Lee, D.D. Gladman, M. Abu-Shakra; Pulmonary hypertension in systemic sclerosis: an analysis of 17 patients; Br. J. Rheumatol., 35 (1996), pp. 989–993
- [3] K.B. Highland; Recent advances in scleroderma-associated pulmonary hypertension; Curr. Opin. Rheumatol., 26 (2014), pp. 637–645
- [4] V. Thakkar, W.M. Stevens, D. Prior, O.A. Moore, J. Byron, D. Liew, et al.; N-terminal pro-brain natriuretic peptide in a novel screening algorithm for pulmonary arterial hypertension in systemic sclerosis: a case–control study; Arthritis Res. Ther., 14 (2012), p. R143
- [5] L. van Bon, A.J. Affandi, J. Broen, R.B. Christmann, R.J. Marijnissen, L. Stawski, et al.; Proteome-wide analysis and CXCL4 as a biomarker in systemic sclerosis; New Engl. J. Med., 370 (2014), pp. 433–443
- [6] G. Condorelli, M.V. Latronico, E. Cavarretta; MicroRNAs in cardiovascular diseases: current knowledge and the road ahead; J. Am. Coll. Cardiol., 63 (2014), pp. 2177–2187
- [7] E. van Rooij, E.N. Olson; MicroRNAs: powerful new regulators of heart disease and provocative therapeutic targets; J. Clin. Invest., 117 (2007), pp. 2369–2376
- [8] K. Yuan, M. Orcholski, X. Tian, X. Liao, V.A. de Jesus Perez; MicroRNAs: promising therapeutic targets for the treatment of pulmonary arterial hypertension; Expert Opin. Ther. Targets, 17 (2013), pp. 557–564
- [9] C. Wei, H. Henderson, C. Spradley, L. Li, I.K. Kim, S. Kumar, et al.; Circulating miRNAs as potential marker for pulmonary hypertension; PLoS One, 8 (2013), p. e64396
- [10] K. Makino, M. Jinnin, A. Hirano, K. Yamane, M. Eto, T. Kusano, et al.; The downregulation of microRNA let-7a contributes to the excessive expression of type I collagen in systemic and localized scleroderma; J. Immunol., 190 (2013), pp. 3905–3915
- [11] Z. Wang, M. Jinnin, H. Kudo, K. Inoue, W. Nakayama, N. Honda, et al.; Detection of hair-microRNAs as the novel potent biomarker: evaluation of the usefulness for the diagnosis of scleroderma; J. Dermatol. Sci., 72 (2013), pp. 134–141
- [12] S. Takashio, Y. Izumiya, M. Jinnin, M. Yamamuro, S. Kojima, H. Ihn, et al.; Diagnostic and prognostic value of subcutaneous tissue biopsy in patients with cardiac amyloidosis; Am. J. Cardiol., 110 (2012), pp. 1507–1511
- [13] I. Kajihara, M. Jinnin, M. Harada, K. Makino, N. Honda, T. Makino, et al.; miR-205 down-regulation promotes proliferation of dermatofibrosarcoma protuberans tumor cells by regulating LRP-1 and ERK phosphorylation; Arch. Dermatol. Res., 306 (2014), pp. 367–374
- [14] M. Etoh, M. Jinnin, K. Makino, K. Yamane, W. Nakayama, J. Aoi, et al.; microRNA-7 down-regulation mediates excessive collagen expression in localized scleroderma; Arch. Dermatol. Res., 305 (2013), pp. 9–15
- [15] S. Roush, F.J. Slack; The let-7 family of microRNAs; Trends Cell Biol., 18 (2008), pp. 505–516
- [16] L. Guo, Y. Yang, J. Liu, L. Wang, J. Li, Y. Wang, et al.; Differentially expressed plasma microRNAs and the potential regulatory function of let-7b in chronic thromboembolic pulmonary hypertension; PLoS One, 9 (2014), p. e101055
- [17] P. Caruso, M.R. MacLean, R. Khanin, J. McClure, E. Soon, M. Southgate, et al.; Dynamic changes in lung microRNA profiles during the development of pulmonary hypertension due to chronic hypoxia and monocrotaline; Arterioscler. Thromb. Vasc. Biol., 30 (2010), pp. 716–723
- [18] C.D. Johnson, A. Esquela-Kerscher, G. Stefani, M. Byrom, K. Kelnar, D. Ovcharenko, et al.; The let-7 microRNA represses cell proliferation pathways in human cells; Cancer Res., 67 (2007), pp. 7713–7722
- [19] K.V. Pandit, D. Corcoran, H. Yousef, M. Yarlagadda, A. Tzouvelekis, K.F. Gibson, et al.; Inhibition and role of let-7d in idiopathic pulmonary fibrosis; Am. J. Respir. Crit. Care Med., 182 (2010), pp. 220–229
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?