Highlights
- We studied the development of tolerance to subchronic p.o. administration of DZN in rats, under both in vivo and in vitro conditions
- As a consequence of AChE inhibition, ACh neurotransmitters are being accumulated in over stimulated nicotinic and muscarinic receptors.
- With isolated diaphragm and ileum, we examined the down regulation of nicotinic and muscarinic receptor function through EFS technique.
- The results of our research could be useful in forensic diagnostics of organophosphate poisoning.
Abstract
Diazinon (DZN) is an organophosphate insecticide which exerts its effect through the inhibition of acetylcholinesterase enzyme (AChE). In this work, we studied the development of tolerance to subchronic p.o. administration of DZN in rats, under both in vivo and in vitro conditions. A group of 20 rats (2 groups, n = 10) was administered p.o. the 1/10 of established LD50 DZN (namely 55.87 mg/kg bw) for 28 days. On the 14th and 28th day of study with isolated diaphragm and ileum, we examined the downregulation of nicotinic and muscarinic receptor function through Electrical Field Stimulation (EFS). Maximum contractility of the diaphragm was recorded on the 14th day of the study (25% higher compared to the non-treated rats), while on the 28th day the contractions almost did not differ from the values found in non-treated rats. EFS of isolated ileum on the 14th day of study caused significantly higher contractions compared to the non-treated rats, but after 28 days, ileum contractions decreased approximately to the level of contractions in non-treated rats. On the 14th study day, we also recorded increased amplitude of spontaneous ileum contractions, compared to non-treated rats. The application of increasing ACh concentrations caused dose-dependent ileum contractions, without statistically significant differences of median effective concentration (EC50 ) values in non-treated and treated rats. Tolerance to subchronic DZN administration develops due to various adaptation mechanisms, including the most important one—downregulation of nicotinic and muscarinic receptor function.
Chemical compounds studied in this article
Diazinon (PubChem CID: 3017) ; Mecamylamine hydrochloride (PubChem CID: 13221) ; Pancuronium bromide (PubChem CID: 27350) ; Atropine sulfate monohydrate (PubChem CID: 64663)
Keywords
Diazinon ; Subchronic intoxication ; EFS ; Diaphragm ; Ileum ; Rats
1. Introduction
Diazinon (O,O -diethyl O -[4-methyl-6-(propan-2-yl) pyrimidin-2-yl] phosphorothioate) is an organophosphate insecticide with a broad spectrum of activity. Therefore, in the past it was used in agriculture and horticulture worldwide for controlling insects in crops, fruit and vegetables and as a pesticide in domestic, agricultural and public buildings. Within Veterinary Medicine, DZN has been used in ectoparasiticide formulations for sheep and cattle, and in collars and washes for external parasite control in companion animals [13] .
Poisoning of people and animals with DZN typically occurs through the ingestion of contaminated food or water. When diazinon gets into the body, through metabolic oxidative desulphurization and mediation of cytochrome P450 (CYP 450), the sulphur is being replaced with oxygen, thus creating phosphoryl compound—diazoxon, characterized by anticholinesterase activity [22] , [29] and [5] , leading to the accumulation of acetylcholine at nerve endings, resulting in overstimulation of the cholinergic system (nicotinic and muscarinic receptors) [4] .
Overstimulation of nicotinic receptors results in the paralysis of principal respiratory muscle-diaphragm, which is a life-threatening condition. Taking this into account, the phrenic nerve-diaphragm preparation [19] has been adopted as a common model in the study of toxic effects of organophosphates, as well as in the studies related to mice and rat diaphragm neuromuscular synapse [2] , [24] , [38] , [41] and [34] .
However, a significant limitation of this method is that phrenic nerve must be intact; and therefore it is not applicable to diaphragms of large animal species or to diaphragm biopsies. The alternative method relies on EFS, with diaphragm samples containing only intramuscular branches of phrenic nerve [42] .
The EFS reevaluation has been described by Seeger et al. [36] . EFS method may be used for two stimulation types of diaphragm samples: indirect stimulation – induced by the activation of postsynaptic nicotinic acetylcholine receptors (nAChR) through the release of ACh neurotransmitter, and direct stimulation – directly activating the contractile machinery of the muscle.
The overstimulation of muscarinic receptors (M3 subtype) causes bronchoconstriction, airway hyper-reactivity and bronchial mucus overproduction [11] ), resulting in severe clinical picture of poisoning with organophosphate compounds. It is well known that M3 subtype of muscarinic receptors is also found in smooth airway and ileum muscles [7] and a primary role of the receptors is to contract smooth muscles [26] , as well as to regulate the activity of many glands, both endocrine and exocrine. Therefore, an isolated rat ileum may be used as a muscarinic receptor model for the study on toxic effects of organophosphates.
Having in mind relatively short DZN half-time and a risk of misinterpretation of laboratory data related to AChE activity (still a “gold” standard in diagnosing organophosphate poisoning), as well as nonspecific clinical manifestations of DZN poisoning, the aim of this study was to examine the EFS method as a potential tool for the diagnostic of DZN poisoning.
2. Material and methods
2.1. Animals
All animal procedures were conducted in accordance with the Directive 2010/63/EU on the protection of animals used for study and other scientific purposes and approved by the Ethical Committee of the Faculty of Veterinary Medicine of Belgrade University.
In vivo tests (determination of LD50 values) and in vitro tests (on isolated diaphragm and ileum) were conducted on a total of 110 white male Wistar rats, weighing 200 ± 20 g. The rats were housed under standard conditions for laboratory animals, on a 12 h light/dark cycle, at room temperature 21–24 °C, and ad libitum access to standard diet and water.
2.2. Substances and methods of administration
For in vivo tests, we used technical DZN (Makhteshim Chemical Works Ltd., Israel) minimum purity 95%, and corn oil as a solvent of DZN (cold-pressed oil from corn germs) (Uvita, Serbia). During in vivo tests, DZN was orally administered to rats, using a stiff gastric sonde (75 mm length) (Hauptner, Switzerland). Maximum volume administered p.o. did not exceed 0.1 ml/100 g of rat bw.
For in vitro tests on isolated rat diaphragm and ileum we used: mecamylamine hydrochloride (Sigma-Aldrich, St. Louis, MO, USA), pancuronium bromide (Sigma-Aldrich, Germany), atropine sulphate (Sigma-Aldrich, St. Louis, MO, USA), acetylcholine (Sigma-Aldrich, St. Louis, MO, USA) and distilled water as a solvent. The organ bath was filled with aerated Tyrode’s solution (125 mM NaCl, 24 mM NaHCO3 , 0.4 mM NaH2 PO4 , 5.4 mM KCl, 1 mM MgCl2 , 1.8 mM CaCl2 , 10 mM Glucose, 95% O2 , 5% CO2 , pH 7.4), and temperature was maintained at 37 °C. The substances were applied to isolated organ bath through 1 ml syringe marked in hundredths.
2.3. Procedures
2.3.1. Determination of acute oral toxicity (LD50 ) of DZN in rats
The testing was conducted on 30 rats, divided into 4 equal experimental groups of 6 rats each, and the control one. DZN was administered p.o., in single doses ranging from 400 to 700 mg/kg of rat bw. Control rats were dosed once with 0.1 ml/100 g of bw DZN solvent (corn oil). The mortality of treated rats was monitored on daily basis for 7 days after the administration.
2.3.2. Determination of acute oral toxicity (LD50 ) of DZN in rats treated for 28 days with 1/10 LD50
The study included 50 rats, divided into 4 equal experimental groups (of 10 rats each) and the control one. DZN was administered p.o. for 28 consecutive days at 1/10 LD50 . Control rats were orally administered only DZN solvent (corn oil) at 0.1 ml/100 g bw. After 28 days of treatment, survived rats were randomized into four new experimental groups of 6 rats each, in order to determine the value of LD50 . DZN was then administered in doses ranging from 500 to 800 mg/kg bw. The mortality of all treated rats was monitored on daily basis for 7 days after the administration.
Two determined LD50 DZN values (LD50 in non-treated rats and LD50 in rats treated for 28 days with 1/10 LD50 ) were analysed and compared in the Results and the Discussion section.
2.3.3. Study of DZN effects on isolated rat diaphragm and ileum
The study was conducted on 30 rats, divided into two experimental groups (n = 10) and the control one. DZN was administered p.o. at 1/10 of acute LD50 , for 28 days. Control rats were orally administered DZN solvent (corn oil) at 0.1 ml/100 g bw. At the end of the treatment period, on the 14th day (group 1) and the 28th day (group 2), 6 rats were randomly selected from both groups and euthanized with an overdose of pentobarbitone. Diaphragms and appropriate ileum segments from sacrificed rats were removed immediately, for further in vitro study.
The diaphragm preparation for in vitro study was arranged as described by Trailović et al. [40] . Diaphragm hemispheres were quickly excised and cut into strips of 1 × 0.5 cm, with incisions parallel to the direction of muscle fibres. Strips were mounted horizontally in an organ bath (with aerated Tyrode’s solution), such that one end was fixed to the bath base, and the other end attached to isometric force transducer, connected to SmartPlus 50 software (El Unit, Serbia). With a pair of platinum electrodes placed parallel to the muscles, EFS was performed by applying tetanic pulses (50 Hz frequency, 25 V voltage, width 15 μs, and 2 s duration) in trains of five pulses every 30 s, with rest interval of 3 min in between.
The preparation from isolated rat ileum was made of pre-terminal part of ileum. A sample of 4–5 cm length was taken at approximately 10 cm from ileocecal valve, and placed into Tyrode’s solution. After removal of fatty tissue, an intestine segment (2 cm length) was placed into organ bath filled with Tyrode’s solution, warmed to the temperature of 37 °C, pH value 7.4. Oxygenation of Tyrode’s solution in organ bath was performed by mixing oxygen and carbon dioxide (95% + 5%, respectively). Similar as with the diaphragm, one end of ileum was fixed to the bath base, and the other end attached to isometric force transducer, so that the ileum segment was located centrally between the two electrodes. Platinum electrodes were placed on the sides of the preparation at a distance of 3–4 mm and connected with stimulator BioSmart 150 (El Unit, Serbia). EFS was performed by applying pulses (50 Hz, 35 V, 2 μs and 2 s) in trains of five pulses every 60 s. After rest interval of 3 min, the same ileum segment was injected with increasing doses of acetylcholine (0.1, 0.3, 1, 3 and 10 μM), in order to determine the dose-response effect. Spontaneous ileum activity, its contractions induced by EFS and ACh level increase, were recorded by isometric transducers and BioSmart 150 software (El Unit, Serbia).
2.4. Statistical analysis
DZN median lethal dose (LD50 ) was calculated by probit analysis according to the method of Litchfield and Wilcoxon I, and the relative potency between the two lethal doses LD50 1/LD50 2, according to the method of Litchfield and Wilcoxon II Tallarida and Murray, [37] ).
Acetylcholine median effective doses (ED50 ) for ileum contractions in both non-treated and 1/10 LD50 DZN treated rats were determined by nonlinear regression and the differences between them tested with the F-test.
Statistical differences between the contractions of ileum and the diaphragm isolated from both non-treated and 1/10 LD50 DZN treated rats (on the 14th and 28th day), were analyzed by ANOVA and unpaired t -test.
Statistical analyses were performed by using commercial statistical software Pharm/PCS, Version 4.0 and GraphPad Prism, Version 4.0.
3. Results
3.1. Acute oral LD50 DZN in non-treated rats and 1/10 LD50 DZN treated rats for 28 days
Through the application of method Litchfield and Wilcoxon I, we determined acute oral LD50 DZN value of 558.70 mg/kg (safety margins from 475.69 to 656.19 mg/kg). DZN poisoning symptoms in rats occurred 2–3 h after the administration, depending on applied dose. Clinical picture of acute poisoning was dominated by classic muscarinic effects, excessive lacrimation and salivation (concluded on the base of wet and stringy hair around medial eye corners, mouth and ventral neck area). The following symptoms also occurred: difficulty breathing, pulsing cross-striated muscles, difficulty moving, with consequent gathering and resting. Another significant symptom was diarrhea.
Based on LD50 value, as determined in the previous section, 1/10 LD50 —55.8 mg/kg bw, was p.o. administered to a new group of rats, for 28 consecutive days. Clinical picture and the mortality of treated rats were monitored on daily basis.
First, the above mentioned, symptoms of DZN poisoning appeared on the 6th day from the beginning of administration. Almost all of treated rats developed behavioural changes, manifested through resting, gathering and loss of interest in environment. Soon after, in parallel with these changes, appeared the first symptoms of cholinergic hyperactivity—lacrimation and salivation, as well as difficulty breathing. Rats with most severe symptoms soon after experienced diarrhea, tremor of skeletal muscles, leading to first lethal outcomes. This was also the pattern of clinical symptoms in other rats that died afterwards. After the 15th treatment day, clinical symptoms of poisoning disappeared and the animals restored physiological conditions. Treated rats started to die on 7th and stopped on 14th day. Thirteen of 40 DZN treated rats died.
Upon the completion of treatment with 1/10 LD50 DZN for 28 days, survived rats were prescribed with acute p.o. LD50 . Through the application method of Litchfield and Wilcoxon I, we determined acute oral LD50 DZN value of 660.24 mg/kg (safety margins from 577.06 to 755.42 mg/kg).
As Fig. 1 shows, sigmoid curve of LD50 DZN values in rats treated for 28 days with 1/10 LD50 shifted to the right in relation to the curve of LD50 in non-treated rats. According to this study, the difference between calculated LD50 values was over 100 mg/kg bw, but the potency value of 0.84 did not reach statistical significance (safety margins from 0.68 to 1.04).
|
|
|
Fig. 1. Dose-response curves of DZN lethal effects in non-treated rats (LD50 = 558.70 mg/kg) (△) and in rats treated for 28 days with 1/10 LD50 (LD50 = 660.25 mg/kg) (О). |
3.2. Contraction of isolated rat diaphragm
We used the antagonists of nAChR—mecamylamine (100 and 300 μM) and pancuronium (2 μM), in order to prove that EFS technique induced muscle contractions only across neuromuscular end-plate.
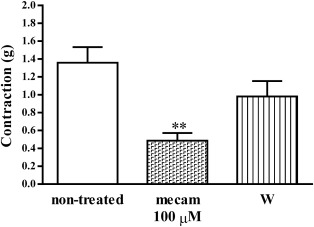
Fig. 2 demonstrates average contraction values of diaphragm preparations from non-treated rats induced with EFS, as well as the effect of 100 μM mecamylamine on the contractions.
|
|
|
Fig. 2. Contractions of diaphragm strips (g) isolated from non-treated rats, induced by EFS (50 Hz, 25 V, 15_μs, 2 s) and the effect of mecamylamine (100 μM) on the contractions (±SEM); W—wash. (**p = 0.0024 statistically significant difference compared to non-treated rats) |
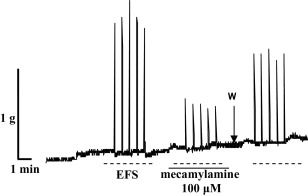
Fig. 3 shows representative records of diaphragm preparation contractions from non-treated rats, induced by EFS, as well as the effect of mecamylamine (100 μM) on the contractions.
|
|
|
Fig. 3. Representative recording of diaphragm strip contractions (g) from non-treated rats induced by EFS (50 Hz, 25 V, 15_μs, 2 s) and the effect of mecamylamine (100 μM) on the contractions; W—wash. |
Considering the fact that the effect of 300 μM mecamylamine was tested only on four diaphragm preparations, this part of study was not statistically analysed. However, obtained results indicated that 300 μM of mecamylamine completely blocked diaphragm contractions induced by EFS (data not shown).
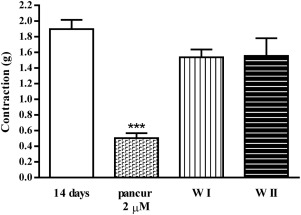
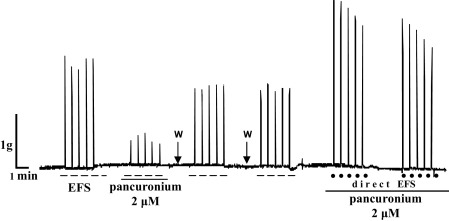
On the 14th day of oral administration of 1/10 LD50 DZN to the rats, diaphragm preparations from treated rats were stimulated with the same EFS parameters as the diaphragms from non-treated rats. Inhibition of nicotinic receptors, previously produced with 100 μM mecamylamine (data not shown), was reproduced with pancuronium which is demonstrated at Fig. 4 and Fig. 5 .
|
|
|
Fig. 4. Contractions of diaphragm strips (g) isolated from rats on the 14th day of treatment with 1/10 LD50 DZN induced by EFS (50 Hz, 25 V, 15_μs, 2 s) and the effect of 2 μM pancuronium on the contractions (±SEM); W—wash. (***p = 0.0009 statistically significant difference compared to non-treated rats) |
|
|
|
Fig. 5. Representative recording of diaphragm strip contractions (g) isolated from rats on the 14th day of treatment with 1/10 LD50 DZN induced by EFS (50 Hz, 25 V, 15 μs, 2 s) and the effect of 2 μM pancuronium on the contractions; W—wash; direct EFS (50 Hz, 25 V, 500 μs, 2 s) and absence of 2 μM pancuronium effect on the contractions. |
The effect of pancuronium also confirms indirect stimulation of diaphragm, occurring at neuromuscular synapses through nAChR (Fig. 5 ).
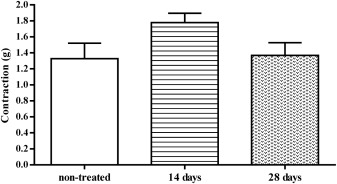
On the 28th day of oral administration of 1/10 LD50 DZN, we examined diaphragm contractions. In this part of study we did not test the effects of mecamylamine and pancuronium, but we monitored the level of diaphragm contractions and compared it with diaphragm contractions from non-treated rats and rats treated for 14 days with 1/10 LD50 DZN. The results of this comparison are shown in Table 1 and Fig. 6 .
| non-treated (n = 6) | DZN 14 days (n = 6) | DZN 28 days (n = 6) |
|---|---|---|
| 1.33± 0.19 | 1.78±0.12 | 1.36±0.16 |
|
|
|
Fig. 6. Comparison of the main values of diaphragm contractions (g) induced by EFS (50_Hz, 25 V, 15 μs, 2 s) isolated from non-treated rats and rats treated for 14 and 28 days with 1/10 LD50 DZN (±SEM). |
The highest average level of diaphragm contractions induced by EFS was recorded in the preparations isolated from rats treated with DZN for 14 days. The contractions were in average for 0.4 g (or by 25%) higher than contractions of diaphragms isolated from non-treated rats and rats treated with DZN for 28 days. Contractions of diaphragms from non-treated rats and those treated 28 days p.o. with DZN showed approximately the same values. Changes recorded in this study did not reach statistically significant level, but due to apparent differences and consistency of results, the changes will be considered under the Discussion section.
3.3. Contractions of isolated rat ileum
We evaluated EFS induced contractions of isolated rat ileum and amplitudes of spontaneous ileum contractions. We used (1 μM) atropine – antagonist of muscarinic receptors – in order to prove that EFS technique induces smooth-muscle contractions of ileum only through the activation of muscarinic receptors.
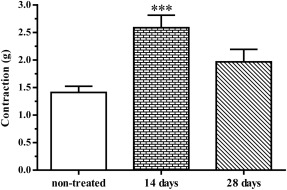
As Fig. 7 shows, statistically significant difference (p = 0.0010) compared with non-treated rats was recorded only in ileum preparations from rats treated for 14 days.
|
|
|
Fig. 7. Comparison of the main values of ileum contractions (g) induced by EFS (50 Hz, 35 V, 2 μs and 2 s) isolated from non-treated rats and rats treated with 1/10 LD50 DZN for 14 days and 28 days (±SEM). (***p = 0.0010—statistically significant difference comparing with non-treated) |
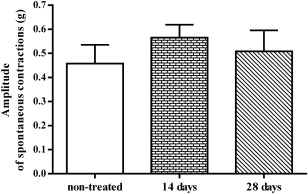
The same ileum preparations were also used to measure the amplitude of spontaneous contractions. Fig. 8 shows that after 14 days of DZN treatment, the amplitude of spontaneous contractions in treated rats increased comparing with non-treated rats, but this difference did not reach statistically significant level. Two weeks later, on the 28th treatment day, the amplitude level slightly decreased.
|
|
|
Fig. 8. The amplitudes of spontaneous contractions of ileum (g) isolated from non-treated rats and rats treated with 1/10 LD50 DZN for 14 and 28 days (±SEM). |
Differences in amplitudes of ileum spontaneous contractions correspond to the differences in ileum contractions induced by EFS, at the same time intervals (Fig. 7 and Fig. 8 ).
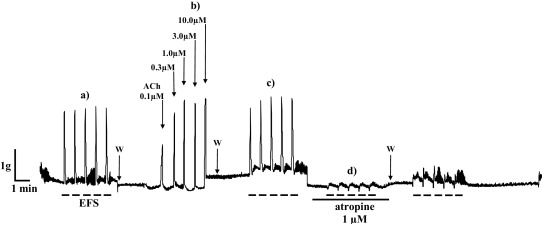
We also tested the effect of increasing concentrations of acetylcholine on contractility of ileum isolated from non-treated rats and the rats treated orally for 14 and 28 days with 1/10 LD50 DZN. Administered ACh doses caused dose-dependent contractions in all tested ileum preparations. Fig. 9 shows representative recordings of the contractions of isolated ileum from rats treated with 1/10 LD50 DZN for 14 days.
|
|
|
Fig. 9. Representative recordings of (a) isolated rat ileum contractions (g) (rats treated with 1/10 LD50 DZN for 14 days) induced by EFS (50 Hz, 35 V, 2 μs and 2 s); W—wash (b) dose-dependent ileum contractions caused by increasing concentrations of ACh; W—wash (c) ileum contractions (g) induced by EFS and (d) 1 μM atropine effect on the contractions of isolated rat ileum induced by EFS; W—wash. |
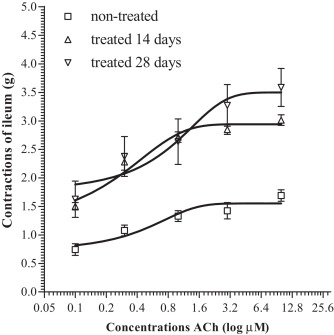
Dose-dependent effects upon ileum contractions induced by increasing doses of acetylcholine were determined by nonlinear regression method. Table 2 and Fig. 10 show obtained results.
| non-treated (n = 6) | treated 14 days (n = 6) | treated 28 days (n = 6) | |
|---|---|---|---|
| EC50 | 1.12 | 1.20 | 0.91 |
| (0.60–2.08) | (1.06–1.37) | (0.18–4.60) | |
| Max effect | 1.55 | 2.86 | 3.49 |
| (1.38–1.72) | (2.74–2.99) | (2.87–4.12) | |
| Hillslope | 0.93 | 2.65 | 0.48 |
| (0.09–1.76) | (1.45–3.86) | (−0.18 to 1.14) | |
|
|
|
Fig. 10. Dose-response curves of ACh effect on the contractions of ileum (g) isolated from non-treated rats and rats treated with 1/10 LD50 DZN for 14 and 28 days. |
EC50 acetylcholine levels in non-treated and treated rats did not differ significantly. The lowest level of EC50 acetylcholine was detected in ileum preparations from rats treated for 28 days and it is interesting that the highest EC50 level was detected in ileum of rats treated for 14 days. However, significantly highest contractions, as well as the highest maximum effect determined by nonlinear regression, were detected in ileums of rats treated for 28 days. The highest Hillslope was at sigmoid curve of relation between dose and effect on ileum of rats treated for 14 days.
4. Discussion
According to our knowledge, this study represents the first electrophysiological proof of downregulation of nicotinic and muscarinic receptors, as one of the adaptation mechanisms to subchronic/chronic intoxication with DZN, confirmed by the application of state-of-the-art EFS method.
In the DZN intoxication, as a consequence of AChE inhibition, ACh neurotransmitters are being accumulated in overstimulated nicotinic and muscarinic receptors. Overstimulation of nicotinic receptors resulted in diaphragm paralysis, and hyperstimulation of muscarinic receptors caused spasm of respiratory airways and hypersecretion of respiratory mucous, which is a life threatening condition for intoxicated organism. Because of this, after we confirmed in vivo the tolerance development to subchronic intoxication with DZN, we also conducted in vitro study on isolated diaphragm, as a nicotinic receptor model and on isolated rat ileum, as a muscarinic receptor model. In this part of the study, the electrophysiological results shown that significant role in tolerance development to subchronic intoxication with DZN, plays also downregulation of nicotinic and muscarinic receptors, with possible activation of some other adaptation mechanisms.
The literature acknowledges several different mechanisms of tolerance development to chronic organophosphate intoxication. The one of them is downregulation of nicotinic and muscarinic receptors [3] , [10] , [8] and [6] . Downregulation of nicotinic and muscarinic receptors has been proved mostly quantitatively, based on the binding of α-bungarotoxin (nAChR antagonist) [3] , [23] , [1] and [27] and quinuclidinyl benzilate (muscarinic receptor antagonist) [45] , [21] , [9] , [31] and [14] .
In the determination of acute oral LD50 DZN, administered doses induced dose-dependent lethality in rats and based on our results, acute p.o. LD50 DZN amounts to 558.7 mg/kg bw. The LD50 DZN complies with the literature values for rats ranging up to 600 mg/kg bw ([39] Toxicological profile for Diazinon US Department of Health and Human Services, www.atsdr.cdc.gov/toxprofiles/tp86.pdf ). Poisoning symptoms onset time and clinical picture of acute organophosphate poisoning, characterized by muscarinic, nicotinic and CNS effects, are also consistent with literature data [32] and [17] .
In rats treated for 28 days with 1/10 of previously determined LD50 DZN, first symptoms appeared on the 6th day, after cumulative dose of 6/10 LD50 . The manifested behavioural changes (decreased movement, loss of interest in environment) pointed to CNS function disorder. Soon after, hyperlacrimation and hypersalivation appeared, followed by muscle fasciculations. In the next few days, after the onset of diarrhea and few fatal issues, the clinical picture of poisoning was complete. In the further course of our study, we would emphasize the finding that until the 15th day, all the above mentioned clinical symptoms disappeared. Until the last day of the study (day 28th), treated rats restored physiological balance and it was not possible to notice any difference between treated and non-treated animals. As such, the finding that symptoms like hypersalivation, hyperlacrimation, muscle fasciculations and diarrhea, disappear with prolonged administration of organophosphates, complies with findings of Milatović and Dettbarn [28] . The authors treated rats with paraoxon at 0.12 mg/kg bw (≈50% LD50 ) for 20 consecutive days. Cholinergic hyperreactivity occurred after the 4th application, sustained until the 10th application and then disappeared, despite the fact that AChE activity was reduced. The authors explained this phenomenon by de novo AChE synthesis, which in tolerant rats increased the affinity for ACh; thereby the enzyme compensates for its significantly lower concentrations with more efficient ACh hydrolysis.
During the 28 days of treatment with 1/10 LD50 DZN, thirteen of 40 treated rats died (32.5%). As opposed to our results, within the study conducted by Milatović and Dettbarn [28] no rat died during 20 days of s.c. administration of organophosphate paraoxon at ≈50% LD50 per day. This difference could be partially explained by different administration method, as well as by the effect of different DZN toxicokinetics. Toxicokinetics of DZN after p.o. application is such that DZN is rapidly absorbed from the gastrointestinal tract and widely distributed throughout the body in both humans [33] and animals [20] and [30] . Both human and animal data demonstrate rapid metabolism of DZN and its oxon to DEP, DETP, and IMHP, which are predominantly excreted in the urine [13] , [25] , [30] , [33] , [43] and [44] . However, although it is generally thought that organophosphorous pesticides do not accumulate in the organism, being rapidly metabolized to their dialkylphosphates, Garcia-Repetto et al. [12] have found an endogenous reintoxication caused by this kind of compound. It is probably due to a release of the compound to blood from a prior deposit of it in adipose tissue. Garcia-Repetto et al. [12] reported detectable levels of DZN in blood, adipose tissue, muscle, liver, and brain of Wistar rats following oral dosing at 23.33 mg/kg. Olive oil was used as a vehicle (we used corn oil as a vehicle). In blood, adipose tissue, and brain, the levels of DZN decreased with time from 4 to 20 days postdosing. Levels in liver and muscle increased up to 8 and 12 days postdosing, respectively, and decreased thereafter. By 30 days postdosing, detectable levels were no longer observed in blood, adipose tissue, muscle, liver, or brain. Considering the fact that our experimental conditions were very similar to those in previous examinations and that we used higher p.o. dose (55.87 mg/kg bw), we may thus conclude that the daily DZN dose produced cumulative effect in organism. According to our study protocol, on the 13th treatment day, when the last death occurred, treated rats received cumulative dose by 30% higher than LD50 and only one rat died. Until the end of the study, rats cumulatively received almost three times higher LD50 DZN dose and no rat died. Described phenomena undoubtedly point to the development of tolerance to DZN toxic effects, confirmed by dose-response curve shifted to the right in rats treated for 28 days with 1/10 LD50 DZN (see Fig. 1 ).
In order to precisely explain the observed tolerance development to subchronic intoxication with DZN, we conducted in vitro studies on diaphragm and ileum isolated from treated rats by EFS method. EFS produced stable indirect contractions of diaphragm, as a sole consequence of nAChR activation at motor end-plate of neuromuscular junction. This was confirmed using nAChR antagonists—mecamylamine and pancuronium, both of which significantly blocked diaphragm contractions ( Fig. 3 and Fig. 5 ). After the pulse-width modification from 15 to 500 μs, mecamylamine and pancuronium had no effect; therefore the occurred contractions were induced by direct stimulation of muscle contractile machinery (Fig. 5 ). With regard to the above, similar results were obtained by Seeger et al. [36] , who confirmed the indirect EFS of diaphragm by the application of pancuronium.
In rats treated for 14 days with low DZN dose, average values of diaphragm contractions induced by EFS were over 25% higher than in non-treated rats, although the difference did not reach statistical significance (Table 1 , Fig. 6 ), which indicated decreased AChE activity. We spectrophotometrically determined lowest AChE activity in erythrocytes (RBC AChE) and diaphragm on the 14th treatment day, and on the 28th day, the AChE activity was significantly statistically higher (p < 0.001) comparing to the value on the 14th day. However, these findings are not presented under Results section. On the 28th treatment day, diaphragm contractions decreased to control values which matched with the “recovery” of enzyme activity, and together with nAChR downregulation, resulted in the absence of significant changes in diaphragm contractile response. As such, our electrophysiological findings comply with the results obtained by the application of other study methods proving tolerance development on the level of neuromuscular diaphragm junctions and other cross-striated muscles (m. soleus, m. extensor digitorum longus ) after chronic exposure of rats to reversible (neostigmine) and irreversible (DFP) AChE inhibitors [3] , [15] , [16] and [1] .
Fourteen days after the administration of low DZN doses, contractions of ileum preparation were significantly higher comparing to non-treated rats (Fig. 7 ). Such a phenomenon could be explained by increased sensitivity of ileum to ACh, which complies with the results of Foley and McPhillips [10] and Hayashi et al. [18] . These authors proved that prolonged administration of sublethal doses of organophosphates induces apparent clinical symptoms of poisoning, leading to increased sensitivity of ileum to ACh. However, over the next two weeks ileum contractions reduced, and on the 28th treatment day, they were slightly higher than in control group (Fig. 7 ). We attributed the decrease in ileum preparation sensitivity to EFS stimulation to the downregulation of muscarinic receptors, which complies with previous findings [10] , [6] and [35] . On the same ileum segments, we also measured the amplitudes of spontaneous contractions. They were slightly higher than control values on the 14th treatment day (Fig. 8 ).
In addition to EFS application, in order to study tolerance development, we also examined the effect of exogenously applied increasing ACh concentrations to ileum contractions. Obtained EC50 ACh values did not differ significantly (Table 2 ). The lowest value of EC50 (0.91 μM) was recorded in ileum preparations after 28 days of treatment, which pointed to the highest ACh sensitivity exactly within this group. The supporting fact is that the highest maximum effect of ileum contractions was recorded exactly in rats treated for 28 days with diazinon (Fig. 10 ). Relatively different results of ileum contractions induced by EFS (sensitivity increased until the 14th day, then decreased until the 28th day), as well as by the outward application of increasing ACh concentrations, are definitely the consequence of adaptation mechanisms related only to endogenous structures of the myenteric plexus.
In conclusion, considering the fact that EFS could be applied to diaphragm biopsies from large animal species (ante mortem) , as well as to the organs of small animals (after autopsy), the results of our study could be useful in forensic diagnostics of organophosphate poisoning. This is particularly important if we have in mind the complexity of organophosphate poisoning diagnostics. There are no standardized tests for prompt detection of organophosphates and/or their metabolites in urine and blood serum. Further, a wide variation in normal cholinesterase values exists and there are no studies reporting a quantitative association between cholinesterase activity levels and exposure to diazinon. Thus, cholinesterase inhibition is not a specific biomarker of effect for diazinon exposure, but is indicative only of effect, and not useful for dosimetric analysis. Obtained results regarding AChE activity in RBC and diaphragm were in compliance with the results obtained through EFS technique—when diaphragm contractions were the highest, the activity of RBC ACh and diaphragm was the lowest. In this context, we consider that EFS technique in combination with analysis of reductions in the level of RBC AChE activity and clinical manifestations of poisoning characterized by a collection of cholinergic signs and symptoms, could contribute to more reliable diagnostics of poisoning with organophosphate compounds.
Nevertheless, having in mind that this study was conducted with rats as test animals, further studies require to be made on large animal samples and domestic animals.
Conflict of interest
The authors declare no conflict of interest.
Transparency document
Transparency Document.
Acknowledgment
This study was supported by the Ministry of Education, Science and Technological Development, Republic of Serbia (Project Nos. TR 31085 and TR 31087).
References
- [1] M. Adler, H.A. Manley, A.L. Purcell, S.S. Deshpande, T.A. Hamilton, R.K. Kan, G. Oyler, O. Lockridge, E.G. Duysen, R.E. Sheridan; Reduced acetylcholine receptor density, morphological remodeling, and butyrylcholin- esterase activity can sustain muscle function in acetylcholinesterase knockout mice; Muscle Nerve, 30 (2004), pp. 317–327
- [2] P.F. Burd, C.B. Ferry; A prolonged contraction at the end-plate region of the diaphragm of rats and mice after anticholinesterases in vitro; J. Physiol. (Lond.), 391 (1987), pp. 429–440
- [3] C.C. Chang, T.F. Chen, Sing-Tai Chuang; Influence of chronic neostigmine treatment on the number of acetylcholine receptors and the release of acetylcholine from the rat diaphragm; J. Physiol. (Lond.), 230 (1973), pp. 613–618
- [4] M.B. Čolović, D.Z. Krstić, T.D. Lazarević-Pašti, A.M. Bondžić, V.M. Vasić; Acetylcholinesterase inhibitors: pharmacology and toxicology; Current Neuropharmacol., 11 (2013), pp. 315–335
- [5] L.G. Costa; Current issues in organophosphate toxicology; Clin. Chim. Acta, 366 (2006), pp. 1–13
- [6] L.G. Costa, B.W. Schwab, S.D. Murphy; Differential alterations of cholinergic muscarinic receptors during chronic and acute tolerance to organophosphorus insecticides; Biochem. Pharmacol., 1 (1982), pp. 3407–3413
- [7] R.M. Eglen; Muscarinic receptor subtype pharmacology and physiology; Prog. Med. Chem., 43 (2005), pp. 105–136
- [8] J.F. Ehlert, N. Kokka, A.S. Fairhunt; Altered [3 H]-quinuclidinyl benzilate binding in the striatum of rats following chronic cholinesterase inhibition with diisopropylfluorophosphate ; Mol. Pharmacol., 17 (1980), pp. 24–30
- [9] B.B. Fitzgerald, L.G. Costa; Modulation of muscarinic receptors and acetylcholinesterase activity in lymphocytes and in brain areas following repeated organophosphate exposure in rats; Fundam. Appl. Toxicol., 20 (1993), pp. 210–216
- [10] D.J. Foley, J.J. McPhillips; Response of the rat ileum, uterus and vas deferens to carbachol and acetylcholine following repeated daily administration of a cholinesterase inhibitor; Br. J. Pharmacol., 48 (1973), pp. 418–425
- [11] A.D. Fryer, P.J. Lein, A.S. Howard, B.L. Yost, R.A. Beckles, D.A. Jett; Mechanisms of organophosphate insecticide-induced airway hyperreactivity; Am. J. Physiol.: Lung Cell. Mol. Physiol., 286 (2004), pp. 963–969
- [12] R. Garcia-Repetto, D. Martinez, M. Repetto; A biodisposition study of diazinon in the Wistar rat; Toxic Subst. Mech., 15 (1996), pp. 415–423
- [13] S.J. Garfitt, K. Jones, H.J. Mason, J. Cocker; Exposure to the organophosphate diazinon: data from a human volunteer study with oral and dermal doses; Toxicol. Lett., 134 (2002), pp. 105–113
- [14] S.X. Guo-Ross, J.E. Chambers, E.C. Meek, R.L. Carr; Altered muscarinic acetylcholine receptor subtype binding in neonatal rat brain following exposure to chlorpyrifos or methyl parathion; Toxicol. Sci., 100 (2007), pp. 118–127
- [15] C.R. Gupta, T.G. Patterson, W.D. Dettbarn; Mechanisms involved in the development of tolerance to DFP toxicity; Fundam. Appl. Toxicol., 5 (1985), pp. 17–28
- [16] C.R. Gupta, T.G. Patterson, D.W. Dettbarn; Mechanisms of toxicity and tolerance to diisopropylphosphorofluoridate at the neuromuscular junction of the rat; Toxicol. Appl. Pharmacol., 84 (1986), pp. 541–550
- [17] C.R. Gupta; Veterinary Toxicology—Basic and Clinical Principles; (first ed.)Academic Press, New York (2007)
- [18] E. Hayashi, H. Okudaira, S. Yamada; Protection by a carbamate against inactivation of cholinesterase and against the induction of supersensitivity of the ileum to acetylcholine produced by repeated administration of an organophosphate; Toxicol. Appl. Pharmacol., 48 (1979), pp. 111–116
- [19] B. Holmstedt; Pharmacology of organophosphorus cholinesterase inhibitors; Pharmacol. Rev., 11 (1959), pp. 567–688
- [20] N.F. Janes, A.F. Machin, M.P. Quick, H. Rogers, D.E. Mundy, A.J. Cross; Toxic metabolites of diazinon in sheep; J. Agric. Food Chem., 21 (1973), pp. 121–124
- [21] D.A. Jett, E.F. Hill, J.C. Fernando, M.E. Eldefrawi, A.T. Eldefrawi; Down-regulation of muscarinic receptors and the m3 subtype in white-footed mice by dietary exposure to parathion; J. Toxicol. Environ. Health, 39 (1993), pp. 395–415
- [22] M. Jokanovic; Biotransformation of organophosphorus compounds; Toxicology, 166 (2001), pp. 139–160
- [23] E.J. Katz, V.I. Cortes, M.E. Eldefrawi, A.T. Eldefrawi; Chlorpyrifos, parathion, and their oxons bind to and desensitize a nicotinic acetylcholine receptor: relevance to their toxicities; Toxicol. Appl. Pharmacol., 146 (1997), pp. 227–236
- [24] S.S. Kelly, C.B. Ferry, J.P. Bamforth; The effects of anticholinesterases on the latencies of action potentials in mouse skeletal muscles; Br. J. Pharmacol., 99 (1990), pp. 721–726
- [25] H.W. Klemmer, E.R. Reichert, W.L. Yauger; Five cases of intentional ingestion of 25 percent diazinon with treatment and recovery; Clin. Toxicol., 12 (1978), pp. 435–444
- [26] A.C. Kruse, J. Hu, A.C. Pan, D.H. Arlow, D.M. Rosenbaum, E. Rosemond, H.F. Green, T. Liu, P.S. Chae, R.O. Dror, D.E. Shaw, W.I. Weis, J. Wess, B.K. Kobilka; Structure and dynamics of the M3 muscarinic acetylcholine receptor; Nature, 22 (2012), pp. 552–556
- [27] D. Kudryavtsev, I. Shelukhina, C. Vulfius, T. Makarieva, V. Stonik, M. Zhmak, I. Ivanov, I. Kasheverov, Y. Utkin, V. Tsetlin; Natural compounds interacting with nicotinic acetylcholine receptors: from low-molecular weight ones to peptides and proteins; Toxins, 7 (2015), pp. 1683–1701
- [28] D. Milatović, W.D. Dettbarn; Modification of acetylcholinesterase during adaptation to chronic, subacute paraoxon application in rat; Toxicol. Appl. Pharmacol., 136 (1996), pp. 20–28
- [29] E. Mutch, F.M. Williams; Diazinon, chlorpyrifos and parathion are metabolised by multiple cytochromes P450 in human liver; Toxicology, 224 (2006), pp. 22–32
- [30] W. Mücke, K.O. Alt, O.H. Esser; Degradation of [14 C]-labeled diazinon in the rat ; J. Agric. Food Chem., 18 (1970), pp. 208–212
- [31] J. Myslivecek, S. Trojan, S. Tucek; Biphasic changes in the density of muscarinic and beta-adrenergic receptors in cardiac atria of rats treated with diisopropylfluorophosphate; Life Sci., 58 (1996), pp. 2423–2430
- [32] H.K. Plumlee; Clinical Veterinary Toxicology; (first ed.)Mosby, St. Louis (2004)
- [33] A. Poklis, F.W. Kutz, J.F. Sperling, D.P. Morgan; A fatal diazinon poisoning; Forensic Sci. Int., 15 (1980), pp. 135–140
- [34] V.K. Reddy, S.S. Deshpande, W.M. Cintra, G.T. Scoble, E.X. Albuquerque; Effectiveness of oximes 2-PAM and HI-6 in recovery of muscle function depressed by organophosphate agents in the rat hemidiaphragm: an in vitro study; Fundam. Appl. Toxicol., 17 (1991), pp. 746–760
- [35] B.W. Schwab, L.G. Costa, S.D. Murphy; Muscarinic receptor alterations as a mechanism of anticholinesterase tolerance; Toxicol. Appl. Pharmacol., 71 (1983), pp. 14–23
- [36] T. Seeger, F. Worek, L. Szinicz, H. Thiermann; Reevaluation of indirect field stimulation technique to demonstrate oxime effectiveness in OP-poisoning in muscles in vitro; Toxicology, 233 (2007), pp. 209–213
- [37] R.J. Tallarida, R.B. Murray; Manual of Pharmacologic Calculation with Computer Programs; (second ed.)Springer-Verlag, New York (1987)
- [38] H. Thiermann, P. Eyer, F. Worek, L. Szinicz; Effects of oximes on muscle force and acetylcholinesterase activity in isolated mouse hemidiaphragms exposed to paraoxon; Toxicology, 214 (2005), pp. 190–197
- [39] Toxicological Profile for Diazinon, US Department of Health and Human Services. Agency for Toxic Substances and Disease Registry; (2008) http://www.atsdr.cdc.gov/toxprofiles/tp86. pdf
- [40] S.M. Trailović, S.R. Ivanović, V.M. Varagić; Ivermectin effects on motor coordination and contractions of isolated rat diaphragm; Res. Vet. Sci., 91 (2011), pp. 426–433
- [41] O.L. Wolthuis, L.A. Kepner; Successful oxime therapy 1 h after soman intoxication in the rat; Eur. J. Pharmacol., 49 (1978), pp. 415–425
- [42] O.L. Wolthuis, R.A.P. Vanwersch, H.J. Van Der Wiel; The efficacy of some bis-pyridinium oximes as antidotes to soman in isolated muscles of several species including man; Eur. J. Pharmacol., 70 (1981), pp. 355–369
- [43] R.C. Wester, L. Sedik, J. Melendres, F. Logan, H.I. Maibach, I. Russell; Percutaneous absorption of diazinon in humans; Food Chem. Toxicol., 31 (1993), pp. 569–572
- [44] H.X. Wu, C. Evreux-Gros, J. Descotes; Diazinon toxicokinetics, tissue distribution and anticholinesterase activity in the rat; Biomed. Environ. Sci., 9 (1996), pp. 359–369
- [45] C.M. Yang, T.M. Dwyer, J.M. Farley; Muscarinic receptors and mucus secretion in swine tracheal epithelium: effects of subacute organophosphate treatment; Fundam. Appl. Toxicol., 17 (1991), pp. 34–42
Document information
Published on 02/05/17
Accepted on 02/05/17
Submitted on 02/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?