Abstract
Fluorescent ‘intravital’ imaging is a new research technique by which the interior of living tissues and organs (in living bodies, if possible) can be observed, revealing the kinetics of cell and molecular processes in real time. Recent technological innovations in optical equipment and fluorescence imaging techniques have enabled a variety of cellular phenomena in different tissues and organs to be characterized under completely native conditions. This shift from static to dynamic biology constitutes the beginning of a new era in biomedical sciences.
Keywords
Cellular dynamics; Immune system; Inflammation; Intravital imaging; Multiphoton microscopy
Abbreviations
S1P, sphingosine-1-phosphate; S1PR1, sphingosine-1-phosphate receptor 1; S1PR2, sphingosine-1-phosphate receptor 2; GFP, green fluorescent protein
To see the unseen
Three hundred years ago, we started to be able to “see things invisible to the eye.” Specifically, Antonie van Leeuwenhoek in the Netherlands observed small organisms (microbes) invisible to the naked eye using his elaborate prototype of an optical microscope; he also discovered that these microorganisms cause human diseases, establishing the field of microbiology. At around the same time, Robert Hooke in Britain identified the basic unit of living organisms, the cell, using his originally developed microscope. These advances established key new concepts in the life sciences by making visible what had previously been invisible. Henceforth, the life sciences have continued to progress whenever technology for seeing the unseen has been developed.
Most of the basic structure of the optical microscope was completed by the end of the 19th century. At that time, Germany led the world in the field of microscopy; German scientists made dramatic advances in the field of pathology by identifying the causes of illness through the microscopic observation of cells and tissues from lesions.
Although the prototype of the modern microscope was largely complete by the 19th century, imaging technology continued to develop in the 20th century. One major advance in imaging technologies is the ability to see living tissues and organs. This was achieved using ‘multiphoton excitation microscopy,’ which enables one to see deep into the interior of tissues and organs with high spatiotemporal resolution while causing little damage.1 and 2
The development of multiphoton excitation imaging to visualize the dynamics and function of living cells
The first report of the biological application of multiphoton excitation microscopy was published by Denk and Webb in 1990,1 but a paper on calcium imaging in inner hair cells by Denk in 19952 had a major impact on the field of biological imaging. It led to multiphoton excitation-based analyses all over the world.
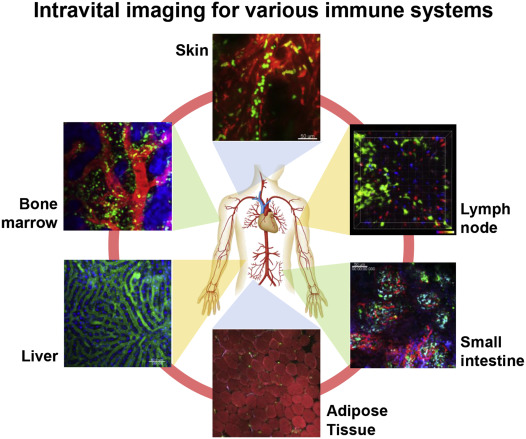
There are two types of live imaging: tissue explant imaging and intravital imaging. In tissue explant imaging, the tissues and organs to be imaged are removed from the body after sacrificing the experimental animal and kept in a viable state during observation in an oxygenated culture.3 and 4 In intravital imaging, experimental animals are kept alive under anesthesia, and the tissues and organs of interest are surgically exposed for imaging. Although this latter approach is methodologically more difficult than tissue explant imaging, it has many advantages. For example, the circulation of blood and cellular dynamics around blood vessels are intact in intravital imaging. This is particularly advantageous for determining the dynamic properties of living cells with an intact blood flow, which is critical for controlling different immune cells in vivo. Intravital imaging for revealing immune cell dynamics in lymph nodes was first reported around 2002 5 and 6; thereafter, the further development of intravital multiphoton imaging technology has enabled us to visualize various tissues and organs under intact conditions (Fig. 1). As part of this development, the present author has developed an imaging system for visualizing live bone tissues in vivo. 7 and 8
|
|
|
Fig. 1. Intravital imaging of various immune system components. Immune cells are highly dynamic and interconnect various tissues and organs by forming a ‘soft-wired’ network. We and other groups are elucidating the basic principles controlling the dynamic nature of immune cells by visualizing their behaviors in vivo using advanced imaging techniques. |
Intravital multiphoton imaging of live bone and bone marrow
Conventional bone histomorphological examinations provide only static information on the distributions of cells and molecules (i.e., not their temporal changes). Recently, a new system for visualizing the interior of bone tissues and bone marrow cavities in living animals under intact conditions using intravital multiphoton microscopy was established.7 and 8 Observation deep into the interior of bone tissues is difficult because not only visible but also infrared light is easily scattered by bone matrices containing calcium phosphate. Thus, we selected murine calvarial bone as an observation target, which is sufficiently thin (∼100–120 μm) to allow the passage of infrared lasers for multiphoton excitation. Using this system, we clarified the regulatory mechanism underlying the migration and localization of osteoclasts and their precursor monocytes in vivo. We demonstrated that sphingosine-1-phosphate (S1P), a lipid mediator that is abundant in blood, controls the migratory behavior of osteoclast precursors, dynamically regulating bone mineral homeostasis. Thus, we identified a critical factor in osteoclastogenesis control. Osteoclast precursors have been shown to express both S1PR1 and S1PR2, two cognate receptors for S1P, 9 and their migration was shown to be finely tuned by the local S1P concentration. 7, 9 and 10 Subsequently, this S1P-mediated regulatory mechanism of osteoclast precursor migration was shown to be therapeutically targeted by active vitamin D,11 confirming the biological significance of the system.
Furthermore, we successfully visualized the bone-destroying function of mature osteoclasts. To identify mature osteoclasts, we generated mice in which the a3 subunit of V-ATPase, an essential factor for acid secretion and bone resorption, was conjugated with green fluorescent protein (GFP) and expressed under the original promoter of V-ATPase a3 subunit (a3-GFP knock-in mice).8 By visualizing mature a3-GFP osteoclasts, we first identified different functional subsets of living mature osteoclasts, from ‘static, bone-resorptive’ (R-type) to ‘moving, non-resorptive’ (N-type). We also found that Th17 cells, which are reportedly involved in arthritic bone destruction, could switch from N-type to R-type by direct cell–cell contact, resulting in enhanced osteoclastic bone resorption at the site.8 and 12 In addition, we visualized the actual events during bone-destructive acidification in limited local areas by developing special chemical fluorescent probes.13 and 14
Intravital imaging of immune cell dynamics in various tissues and organs
Secondary lymphoid organs (peripheral lymph nodes)
An initial application of intravital multiphoton imaging was to visualize an explanted lymph node.5 and 6 Several lymphocytes (e.g., T and B cells) were visualized in lymph nodes by labeling with permeable fluorescent dyes and adoptively transferred to recipient mice. Naive T cells showed higher motility than B cells in lymph nodes. Further study revealed the mode of organized T cell migration, which, unlike ‘random-walking,’ is coordinated by structures composed of stromal cells and the reticular network.5 It was also revealed that T cells change their motility upon contact with antigen-presenting cells.6 When they encounter activated antigen-presenting dendritic cells (DCs) in lymph nodes, T cells form stable complexes that are sustained for a couple of hours, followed by reactivation of their motility. On the other hand, T cells exhibit shorter sustained contact with DCs upon tolerance induction. These results suggest that antigen presentation is a highly dynamic process and that the migratory behaviors and functions of the immune cells involved in this process are closely associated with each other.
It has been suggested that B cells expand only in germinal centers, and that activated T cells migrate towards B cell follicles to support antibody production. Intravital imaging has also revealed spatiotemporal aspects of these dynamic cellular interactions in lymph nodes.15
Thymus
Using thymal tissue cultures, multiphoton microscopy has enabled visualization of the dynamic interactions between thymocytes and thymic epithelial cells undergoing positive and negative selection.16 CD4+CD8+ double-positive immature thymocytes migrate to the central medulla, where they complete positive selection and become either CD4+CD8− or CD4−CD8+ single-positive thymocytes. The imaging of live thymic tissues has demonstrated that the motility of thymocytes is generally high and that their recognition of major histocompatibility complex/peptide complexes is associated with both stable and dynamic contacts with thymic epithelial cells, possibly representing different signals or different selection stages. After completing positive selection, the thymocytes migrate rapidly toward the medulla for negative selection.16 During negative selection, thymocytes migrate slowly in a limited area (∼30 μm in diameter).17
Blood vessels
Activated immune cells can enter inflammatory sites through blood vessels.18 Granulocytes are the first cells to be recruited to inflamed sites. Intravital imaging can detect dynamic inflammatory cell events, including the tethering, rolling, crawling, and invasion of granulocytes, which is controlled by adhesion molecules such as P- and L-selectins, as well as integrins on endothelial cell surfaces.19 Their attachment depends on interactions with several chemokines such as CX3C-chemokine ligand 1 and adhesion molecules such as intercellular adhesion molecule 1.
Inflammatory models of peripheral tissues and organs
Pathogenic T cells have been visualized in the spinal cord of a murine encephalitis model, EIA,20 in which T cells were shown to be highly motile but immobilized upon encountering cognate antigens, similar to the case in lymph nodes. In a diabetes model using NOD mice, interactions between antigen-specific T cells and DCs were observed in a draining lymph node.21
Future development of intravital imaging in immunology
Intravital or in vivo imaging has revealed the dynamic nature of immune and inflammatory systems. One of its most significant advantages is the ability to clarify spatiotemporal parameters in living cells and tissues that would not be achieved by other methodologies such as flow cytometry. Intravital imaging technology has continued to evolve, and trials on the detection of immune and inflammatory reactions in human patients have been performed. Although a range of limitations and obstacles impeding the advance of this technology remain, the analysis of an increasing number of subjects is falling within the scope of intravital imaging techniques.
Conflict of interest
The author received honoraria from Chugai Pharmaceutical and Daiichi-Sankyo, and research funding from Chugai Pharmaceutical, Daiichi-Sankyo, Novartis Pharma, Shionogi and Ono Pharmaceutical.
References
- 1 W. Denk, J.H. Strickler, W.W. Webb; Two-photon laser scanning fluorescence microscopy; Science, 248 (1990), pp. 73–76
- 2 W. Denk, J.R. Holt, G.M. Shepherd, D.P. Corey; Calcium imaging of single stereocilia in hair cells: localization of transduction channels at both ends of tip links; Neuron, 6 (1995), pp. 1311–1321
- 3 T. Nemoto, R. Kimura, K. Ito, A. Tachikawa, Y. Miyashita, M. Iino, et al.; Nat Cell Biol, 3 (2001), pp. 253–258
- 4 M. Matsuzaki, G.C.R. Ellis-Davies, T. Nemoto, Y. Miyashita, M. Iino, H. Kasai; Nat Neurosci, 4 (2001), pp. 1086–1092
- 5 S. Stoll, J. Delon, T.M. Brotz, R.N. Germain; Science, 296 (2002), pp. 1873–1876
- 6 M.J. Miller, S.H. Wei, I. Parker, M.D. Cahalan; Two-photon imaging of lymphocyte motility and antigen response in intact lymph node; Science, 296 (2002), pp. 1869–1873
- 7 M. Ishii, J.G. Egen, F. Klauschen, M. Meier-Schellersheim, Y. Saeki, J. Vacher, et al.; Sphingosine-1-phospate mobilizes osteoclast precursors and regulates bone homeostasis; Nature, 458 (2009), pp. 524–528
- 8 J. Kikuta, Y. Wada, T. Kowada, Z. Wang, G.-H. Sun-Wada, I. Nishiyama, et al.; Dynamic visualization of RANKL and Th17-mediated osteoclast function; J Clin Invest, 123 (2013), pp. 866–873
- 9 M. Ishii, J. Kikuta, Y. Shimazu, M. Meier-Schellersheim, R.N. Germain; Chemorepulsion by blood S1P regulates osteoclast precursor mobilization and obne remodeling in vivo; J Exp Med, 207 (2010), pp. 2793–2798
- 10 M. Ishii, J. Kikuta; Sphingosine-1-phosphate signaling controlling osteoclasts and bone homeostasis; Biochim Biophys Acta, 1831 (2013), pp. 223–227
- 11 J. Kikuta, S. Kawamura, F. Okiji, M. Shirazaki, S. Sakai, H. Saito, et al.; S1P-mediated osteoclast precursor monocyte migration is a critical point of control in antibone-resorptive action of active vitamin D; Proc Natl Acad Sci U S A, 110 (2013), pp. 7009–7013
- 12 J. Kikuta, M. Ishii; Osteoclast migration, differentiation, and function: novel therapeutic targets for rheumatic diseases; Rheumatology, 52 (2013), pp. 226–234
- 13 T. Kowada, J. Kikuta, A. Kubo, M. Ishii, H. Maeda, S. Mizukami, et al.; In vivo fluorescence imaging of bone-resorbing osteoclasts; J Am Chem Soc, 33 (2011), pp. 17772–17776
- 14 H. Maeda, T. Kowada, J. Kikuta, M. Furuya, M. Shirazaki, S. Mizukami, et al.; Real-time intravital imaging of pH variation associated with cell osteoclast activity and motility using designed small molecular probe; Nat Chem Biol (2016) http://dx.doi.org/10.1038/nchembio.2096
- 15 T.A. Schwickert, R.L. Lindquist, G. Shakhar, G. Livshits, D. Skokos, M.H. Kosco-Vilbois, et al.; In vivo imaging of germinal centres reveals a dynamic open structure; Nature, 446 (2007), pp. 83–87
- 16 P. Bousso, N.R. Bhakta, R.S. Lewis, E. Robey; Dynamic of thymocyte-stromal cell interactions visualized by two-photon microscopy; Science, 296 (2002), pp. 1876–1880
- 17 M.L. Borgne, E. Ladi, I. Dzhagalov, P. Herzmark, Y.F. Liao, A.K. Chakraborty, et al.; The impact of negative selection on thymocyte migration in the medulla; Nat Immunol, 10 (2009), pp. 823–830
- 18 M.J. Hickey, P. Kubes; Intravascular immunity: the host-pathogen encounter in blood vessels; Nat Rev Immunol, 9 (2009), pp. 364–375
- 19 C. Auffray, D. Fogg, M. Garfa, G. Elain, O. Join-Lambert, S. Kayal, et al.; Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior; Science, 317 (2007), pp. 666–670
- 20 N. Kawakami, U.V. Nagerl, F. Odoardi, T. Bonhoeffer, H. Wekerle, A. Flügel; Live imaging of effector cell trafficking and autoimmune encephalomyelitis lesion; J Exp Med, 201 (2005), pp. 1805–1814
- 21 Q. Tang, J.Y. Adams, A.J. Tooley, M. Bi, B.T. Fife, P. Serra, et al.; Visualizing regulatory T cell control of autoimmune responses in non-obese diabetic mice; Nat Immunol, 7 (2006), pp. 83–92
Document information
Published on 05/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?