Dear Editor,
Pregabalin is commonly used to treat patients with various neuropathic pain syndromes through its ligand action for α 2-δ subunits of the voltage-gated calcium channels in the nervous system.1 Pregabalin is known to cause cutaneous adverse events very rarely and there have been only two case reports on cutaneous adverse events by pregabalin.2 and 3 Herein we report a case of maculopapular type drug eruption caused by pregabalin. We also review a case of pregabalin-related drug eruption in English and Japanese literatures.
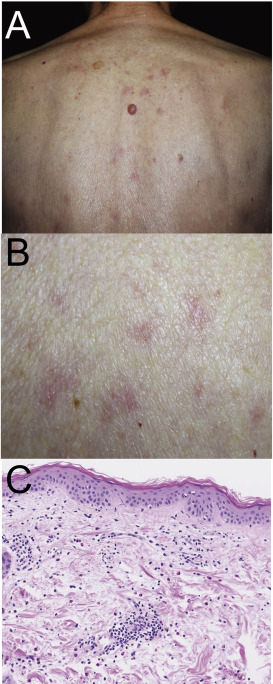
An 86 year-old male developed multiple papules and erythema on his trunk and extremities, 2 weeks after pregabalin administration for his peripheral neuralgia, and was referred to our department for evaluation of his eruption. Physical examination revealed palpable erythema and papules occurring on his trunk and extremities without the involvement of mucous membrane (Fig. 1A,B). Laboratory examination revealed that all the results including liver and renal functions were within normal. Skin biopsy taken from erythema on his back revealed lymphocyte infiltration around vessels (Fig. 1C). Eosinophils were also observed in the upper dermis. One week after the first visit, patch testing with pregabalin using vaseline vehicle or water-soluble ointment was negative.
|
|
|
Fig. 1. Clinical manifestation and histology. (A, B) Clinical manifestation. (A) Low magnification view of clinical feature showing erythema and papules on his trunk. (B) High manifestation views of clinical feature on back. (C) Histological examination exhibiting infiltration of lymphocytes and eosinophils around vessels. |
At the first visit, we performed lymphocyte stimulation test (LST) with pregabalin as described previously.4 and 5 Because of its insoluble characteristics, we dissolved pregabalin into culture medium by treatment of ultrasonic wave (US cleaner (US-4A); As One, Osaka, Japan) for 10 min. 3H-thymidine incorporation was significantly increased by the addition of 3.9 × 10−5 M pregabalin (corresponding to Cmax) to the peripheral lymphocyte culture with stimulation index of 2.1. Based on a clinical course and laboratory examination, we diagnosed his skin eruption as maculopapular type drug eruption caused by pregabalin. Five days after topical application of betamethasone butyrate propionate ointment and discontinuation of pregabalin, his eruption remarkably improved.
Although the frequency is relatively low, several cases of drug eruption due to pregabalin have been reported. To examine its characteristics, we reviewed the English and Japanese reported cases of drug eruption caused by pregabalin. There have been 3 reported cases, including our case2 and 3 (Table 1). Skin eruption types were as follow; 1 case of erythematous edema, 2 cases of maculopapular type. All cases developed relatively mild clinical manifestation and each skin eruption improved approximately 1 week after discontinuation of the drug. Interestingly, two cases of our department showed a positive result of LST,3 while patch testing with pregabalin was negative in both cases. It might depend on the characteristic of pregabalin, which is nearly insoluble for water and lipid. We were finally able to dissolve pregabalin into culture medium by the help of the ultrasonic waves. Taken together, it should be kept in mind that test reagents of pregabalin should be prepared appropriately to determine the causative drug for drug eruption.
| Author | Age/Sex | Eruption type | To identify causative drug | Duration of therapy |
|---|---|---|---|---|
| Smith TL et al.2 | 35/Female | Maculopapular type | N.D | 7 days |
| Yoshioka M et al.3 | 44/Male | Erythematous edema | LST: positivePatch test: negative | 10 days |
| Our case | 86/Male | Maculopapular type | LST: positivePatch test: negative | 5 days |
N.D, not described.
Conflict of interest
The authors have no conflict of interest to declare.
References
- 1 J.G. McGivern; Targeting N-type and T-type calcium channels for the treatment of pain; Drug Discov Today, 11 (2006), pp. 245–253
- 2 T.L. Smith, A. Baldwin, L.L. Cunningham Jr., A.M. Cook; Rash associated with pregabalin use; Ann Pharmacother, 42 (2008), pp. 1899–1902
- 3 M. Yoshioka, R. Okura, R. Hino, M. Nakamura; [A case of drug eruption due to pregabalin]; [Jpn J Clin Dermatol], 67 (2013), pp. 569–572 (in Japanese)
- 4 A. Inoue, Y. Sawada, S. Ohmori, D. Omoto, S. Haruyama, M. Yoshioka, et al.; Maculopapular type drug eruption caused by silodosin; Allergol Int, 65 (2016), pp. 219–220
- 5 N. Saito-Sasaki, Y. Sawada, S. Ohmori, D. Omoto, S. Haruyama, M. Yoshioka, et al.; Multiple fixed drug eruption caused by ropinirole in a patient with Parkinsons disease; Allergol Int, 65 (2016), pp. 221–222
Document information
Published on 05/04/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?