Abstract
Pioglitazone, a thiazolidinedione (TZD), is widely used as an insulin sensitizer in the treatment of type 2 diabetes. However, body weight gain is frequently observed in TZD-treated patients. Fish oil improves lipid metabolism dysfunction and obesity. In this study, we demonstrated suppression of body weight gain in response to pioglitazone administration by combination therapy of pioglitazone and fish oil in type 2 diabetic KK mice. Male KK mice were fed experimental diets for 8 weeks. In safflower oil (SO), safflower oil/low-dose pioglitazone (S/PL), and safflower oil/high-dose pioglitazone (S/PH) diets, 20% of calories were provided by safflower oil containing 0%, 0.006%, or 0.012% (wt/wt) pioglitazone, respectively. In fish oil (FO), fish oil/low-dose pioglitazone (F/PL), and fish oil/high-dose pioglitazone (F/PH) diets, 20% of calories were provided by a mixture of fish oil and safflower oil. Increased body weight and subcutaneous fat mass were observed in the S/PL and S/PH groups; however, diets containing fish oil were found to ameliorate these changes. Hepatic mRNA levels of lipogenic enzymes were significantly decreased in fish oil-fed groups. These findings demonstrate that the combination of pioglitazone and fish oil decreases subcutaneous fat accumulation, ameliorating pioglitazone-induced body weight gain, through fish oil-mediated inhibition of hepatic de novo lipogenesis.
Abbreviations
AOX , acyl-CoA oxidase ; ACC , acetyl-CoA carboxylase ; ATM , adipose tissue macrophage ; AUC , area under the curve ; BAT , brown adipose tissue ; CPT-1 , carnitine palmitoyl transferase 1 ; CT , computed tomography ; DHA , docosahexaenoic acid ; ELISA , enzyme-linked immunosorbent assay ; EPA , eicosapentaenoic acid ; FAS , fatty acid synthase ; FFA , free fatty acid ; G6pase , glucose-6-phosphatase ; GPAT , glycerol-3-phosphate acyltransferase ; HDL-C , high-density lipoprotein cholesterol ; H&E , hematoxylin and eosin ; HOMA-IR , homeostasis model assessment of insulin resistance ; Insig-1 , insulin-induced gene 1 ; IR , insulin resistance ; ITT , insulin tolerance test ; MCAD , medium-chain acyl-CoA dehydrogenase ; MCP-1 , monocyte chemoattractant protein-1 ; OGTT , oral glucose tolerance test ; PEPCK , phosphoenolpyruvate carboxykinase ; PPARα , peroxisome proliferator-activated receptor alpha ; PPARγ , peroxisome proliferator-activated receptor gamma ; RT-PCR , real-time polymerase chain reaction ; SCD-1 , stearoyl-CoA desaturase 1 ; SREBP , sterol regulatory element-binding protein ; TNF-α , tumor necrosis factor-α ; TLR-4 , toll-like receptor-4 ; TZD , thiazolidinedione ; UCP-2 , uncoupling protein 2 ; VLDL , very low-density lipoprotein ; WAT , white adipose tissue
Keywords
Fish oil ; Pioglitazone ; Adverse effect
1. Introduction
Lifestyle-related diseases, particularly type 2 diabetes, are increasingly becoming a major global health issue associated with excessive caloric intake and decreasing physical activity [1] . Type 2 diabetes epidemic has grown rapidly worldwide and is known to affect an estimated 387 million people (Diabetes Atlas 2014 update, International Diabetes Federation, IDF, 2014). The major pathogenic mechanism underlying type 2 diabetes is thought to be insulin resistance (IR) associated with abdominal obesity [2] . Excessive amounts of unconsumed calories result in triglyceride accumulation in adipocytes, particularly in visceral fat tissue, leading to cellular hypertrophy and stimulation of inflammatory cytokine production [3] , [4] and [5] . In addition, large amounts of free fatty acids (FFAs) derived from hypertrophic adipocytes are transferred to insulin responsive organs, such as the liver, skeletal muscle, and pancreas, as “ectopic fat” leading to decreased insulin sensitivity [6] and [7] . Adiponectin, an adipose tissue-derived secreted protein with insulin-sensitizing activity, has been shown to be decrease in abdominal obesity, with a negative correlation found between plasma adiponectin levels and body mass index [8] . Through the interactions of these complex mechanisms, obesity-induced accumulation of visceral fat is thought to contribute to IR through “lipotoxicity” [2] .
Thiazolidinediones (TZDs), potent and selective ligands for peroxisome proliferator-activated receptor gamma (PPARγ), are widely used clinically in the treatment of type 2 diabetes as insulin-sensitizing agents [9] . TZDs promote the differentiation of preadipocytes into mature adipocytes and apoptosis of hypertrophic adipocyte, a process termed “adipose tissue remodeling.” Further, TZDs increase plasma adiponectin levels and reduce the production of adipocytokines, such as tumor necrosis factor-α (TNF-α) and monocyte chemoattractant protein-1 (MCP-1), thereby ameliorating adipocytokine-mediated IR [10] , [11] , [12] and [13] . Thus, the antihyperglycemic effects of TZDs are thought to be mediated independently of increased insulin production, suggesting the greatest utility of TZDs lies in the treatment of type 2 diabetic patients without pancreatic beta cell failure [14] and [15] . Many studies have demonstrated TZDs have multifaceted utility including the improvement of lipid profiles [16] and [17] , amelioration of nonalcoholic steatohepatitis [18] and [19] , systemic anti-inflammatory effects [20] , and the prevention of arteriosclerosis [21] and [22] . And, TZD-induced “adipose tissue remodeling” is thought to lead increased absorption and storage of excess lipids by small adipocytes newly derived in response to the effects of TZDs [13] and [23] . However, body weight gain is considered a major adverse effect of TZD therapy following results from animal studies and several clinical trials in patients with type 2 diabetes [24] and [25] . Moreover, subcutaneous fat accumulation is frequently observed in patients administered TZDs [26] .
Fish oil contains eicosapentaenoic acid (EPA, 20-5) and docosahexaenoic acid (DHA, 22-6), which have been shown to have beneficial effects in hyperlipidemia, fatty liver, atherosclerosis, and cardiovascular disease in both animal models and clinical trials [27] , [28] , [29] and [30] . Hepatic de novo lipogenesis is mainly controlled by sterol regulatory element-binding proteins (SREBPs), transcription factors that regulate the expression of genes involved in lipogenesis. SREBP-1c, one of the SREBPs isoforms, plays a particularly critical role in fatty acid synthesis through the regulation of several target genes including fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), and stearoyl-CoA desaturase 1 (SCD-1) [31] . Fish oil consumption has been shown to decrease mRNA expression and mature protein levels of SREBP-1c [32] and [33] . Additionally, fatty acid oxidation has been shown to be stimulated by fish oil consumption through stimulation of peroxisome proliferator-activated receptor alpha (PPARα), a nuclear receptor involved in the regulation of numerous target genes, such as acyl-CoA oxidase (AOX) and uncoupling protein 2 (UCP-2) [32] and [34] . In our previous study of female KK mice, we demonstrated a diet with 25% of total calories from fish oil had numerous beneficial effects including suppression of body weight gain associated with whole-body adiposity, improvements in lipid metabolism dysfunction, the prevention of hyperinsulinemia due to increased fatty acid oxidation, and decreased lipid synthesis [35] . Therefore, we hypothesized that the combination of pioglitazone and fish oil prevents pioglitazone-induced body weight gain attributed to the accumulation of subcutaneous fat and exerts synergistic beneficial effects on glucose and lipid metabolism. In this study, we evaluated the combined effects of pioglitazone and fish oil in mice of type 2 diabetes to test the above hypothesis and inform the development of novel TZD treatment approaches.
2. Materials and methods
2.1. Animals and diets
All animal studies were approved by the Guidelines of Institutional Animal Care and Use Committee at the Josai University Life Science Center performed in accordance with the “Standards Relating to the Care and Management of Experimental Animals” (Notice No. 6 of the Office of the Prime Minister of Japan dated March 27, 1980). Male KK mice at 6 weeks of age were obtained from Tokyo Laboratory Animals Science Co. (Tokyo, Japan). All mice were individually housed and allowed free access to water and feed under the conditions of a 12-h light–dark cycle, a temperature of 22 ± 2 °C, and humidity of 55 ± 10% at Josai University Life Science Center. Mice were fed a standard commercial rodent diet for 1 week to stabilize metabolic conditions and divided into 6 groups (n = 5 in each group). All groups were fed experimental diets with contents composed of 60% carbohydrates, 20% fats, and 20% protein for 8 weeks. Dietary fats used safflower oil (Benibana Foods, Tokyo, Japan) and/or fish oil (NOF, Tokyo, Japan). Safflower oil contained 78 wt% oleic acid; fish oil contained 40.4 wt% polyunsaturated fatty acids, especially 6.6 wt% EPA and 24.7 wt% DHA. SO diet contained 20% of calories from safflower oil, and FO diet contained 10% of calories from safflower oil and 10% from fish oil. SO and FO diets were supplemented with 0.006% wt/wt (low-dose pioglitazone [PL]) or 0.012% wt/wt (high-dose pioglitazone [PH]) pioglitazone and designated S/PL, S/PH, F/PL, or F/PH accordingly. Dietary details are shown in Table 1 . The feed were changed once in every 2–3 days, and the residual amounts were recorded and showed as total food intake during experimental periods. All diets were stored at −30 °C until each meal is supplied freshly.
| Group | Ingredients (g) | |||||
|---|---|---|---|---|---|---|
| SO | S/PL | S/PH | FO | F/PL | F/PH | |
| Safflower oil | 8 | 8 | 8 | 4 | 4 | 4 |
| Fish oil | 4 | 4 | 4 | |||
| Casein | 20 | 20 | 20 | 20 | 20 | 20 |
| Sucrose | 10.37 | 10.37 | 10.37 | 10.37 | 10.37 | 10.37 |
| β-starch | 51.83 | 51.83 | 51.83 | 51.83 | 51.83 | 51.83 |
| Vitamin mixa | 1 | 1 | 1 | 1 | 1 | 1 |
| Mineral mixa | 3.5 | 3.5 | 3.5 | 3.5 | 3.5 | 3.5 |
| Cellulose powder | 5 | 5 | 5 | 5 | 5 | 5 |
| l-Cystin | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| t-Butylhydroquinone | 0.0016 | 0.0016 | 0.0016 | 0.0016 | 0.0016 | 0.0016 |
| Pioglitazone | 0.006 | 0.012 | 0.006 | 0.012 | ||
| Total | 100.00 | 100.01 | 100.01 | 100.00 | 100.01 | 100.01 |
| Energy (kcal/100g) | 374.02 | 374.00 | 373.99 | 374.02 | 374.00 | 373.99 |
| Fat energy (%) | 19.70 | 19.70 | 19.70 | 19.70 | 19.70 | 19.70 |
a. Vitamin and mineral mix were based on the AIN-93 M formation. Vitamin mix substituted 0.25% sucrose for choline bitartrate.
2.2. Computed tomography
Radiographic estimations of abdominal composition were performed by computed tomography (CT) for experimental animals using the mouse mode of the CT scanner (La Theta LCT100; ALOKA, Tokyo, Japan). At the end of the experiment, mice fasted for 3 h were anesthetized with intraperitoneal injections of pentobarbital sodium (Dainippon Sumitomo Pharma, Osaka, Japan) before scanning. Abdominal compositions of visceral and subcutaneous fats were estimated by fat slice images at 2 mm intervals between the second lumbar vertebra (L2) and L4 using La Theta software (version 2.10).
2.3. Collection of blood and tissue samples
After CT scanning, blood samples were obtained from tail veins of anesthetized mice. Glucose levels were measured using a blood glucose monitoring system (One Touch Ultra; Johnson & Johnson, Inc.). Mice were then immediately weighed prior to dissection. Blood samples were obtained from the inferior vena cava and treated with EDTA-2Na. Tissue samples, including liver, white adipose tissue (WAT) around the epididymis, and brown adipose tissue (BAT), were immediately removed, weighed, frozen in liquid nitrogen, and then stored at −80 °C until further use. Representative images of harvested whole livers from each group were taken using a digital camera to assess macroscopic appearance. For histopathological examination or morphometric analysis, samples of liver and WAT were collected from 4 to 5 mice per group, fixed in 10% neutral buffered formalin (Wako Pure Chemical Industries, Ltd., Osaka, Japan) and stained with hematoxylin and eosin (H&E, Kotobiken Medical Laboratoties, Inc., Tokyo, Japan). Blood samples were centrifuged at 900 × g for 10 min to collect plasma, which was stored at −80 °C until further use.
2.4. Oral glucose tolerance tests
Oral glucose tolerance tests (OGTTs) were performed in 3 h fasted mice at 14 weeks of age. Glucose solutions were prepared by diluting glucose in ultrapure water and orally administrated at a dose of 1 g glucose/kg body weight. Blood samples were obtained before (0 min) and 30, 60, and 120 min after glucose administration, and glucose levels were immediately measured using a blood glucose monitoring system (One Touch Ultra; Johnson & Johnson, Inc.).
2.5. Insulin tolerance tests
Insulin tolerance tests (ITTs) were performed in 3 h fasted mice at 14 weeks of age by intraperitoneal injection of insulin (Humulin R; Lilly USA, Indianapolis, USA) diluted in saline at a dose of 0.75 units/kg body weight. Blood glucose levels were determined using a similar protocol to OGTT. Blood glucose levels in response to insulin were expressed as percentages of initial values prior to insulin administration.
2.6. Measurement of hepatic lipid content and plasma biochemical parameters
Hepatic lipid extraction was performed using approximately 100 mg of liver tissue per mouse according to a previously described method [36] . Hepatic triglyceride and total cholesterol levels, and plasma triglyceride, total cholesterol, high-density lipoprotein cholesterol (HDL-C), and FFA levels, were quantified by an enzymatic colorimetric method using commercial kits (Wako E-Test kits; Wako Pure Chemical Industries Ltd., Osaka, Japan). Plasma insulin and leptin levels were determined by enzyme-linked immunosorbent assay (ELISA) using Insulin ELISA kits and Leptin/mouse ELISA kits (Morinaga Institute of Biological Science, Tokyo, Japan), respectively. Plasma adiponectin levels were measured using Mouse/rat adiponectin ELISA kits (Otsuka Pharmaceutical, Tokyo, Japan). Homeostasis model assessment of insulin resistance (HOMA-IR) indices and plasma non-HDL-C levels were calculated using the following formula: HOMA-IR, fasting blood glucose (mg/dl) × fasting plasma insulin (μU/ml)/405; plasma non-HDL-C, plasma total cholesterol (mg/dl) − plasma HDL-C (mg/dl).
2.7. Liver and WAT histopathology and adipocyte percentage area quantification
H&E-stained liver and epididymal WAT sections were examined under a microscope at 100-fold magnification. For morphometric analysis of epididymal WAT, adipocytes were manually traced and quantitated with an image analysis system (ImageJ, Wayne Rasband, NIH). Four different fields per group were randomly captured and white adipocyte areas were measured from more than 900 cells per group.
2.8. RNA isolation and measurement of mRNA levels by real-time polymerase chain reaction
TRIzol Reagent (Invitrogen Co.) was used to extract total RNA from liver and WAT samples from each mouse according to the manufacturer’s protocol. Quantitative real-time polymerase chain reaction (RT-PCR) was performed using 1 μg of total RNA per sample on an ABI Prism 7500 Sequence Detection System (Applied Biosystems, Foster City, CA) using QuantiTect SYBR Green RT-PCR kits (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. Primer sequences used in the present study are shown in Table 2 . Thermal cycling conditions were set as follows: 1 cycle of reverse transcription at 50 °C for 30 min, initial activation at 95 °C for 15 min, then 40 cycles of denaturation at 94 °C for 15 s, annealing at 55 °C for 30 s and extension at 72 °C for 1 min.
| Genes | Forward(5′ → 3′) | Reverse(3′ → 5′) |
|---|---|---|
| SREBP-1c | GGAGCCATGGATTGCACATT | GGCCCGGGAAGTCACTGT |
| Insig-1 | TCACAGTGACTGAGCTTCAGCA | TCATCTTCATCACACCCAGGAC |
| FAS | TCACCACTGTGGGCTCTGCAGAGAAGCGAG | TGTCATTGGCCTCCTCAAAAAGGGCGTCCA |
| SCD-1 | CCGGAGACCCCTTAGATCGA | TAGCCTGTAAAAGATTTCTGCAAACC |
| ACC | TGACAGACTGATCGCAGAGAAAG | TGGAGAGCCCCACACACA |
| GPAT | TCATCCAGTATGGCATTCTCACA | GCAAGGCCAGGACTGACATC |
| AOX | TCAACAGCCCAACTGTGACTTCCATTA | TCAGGTAGCCATTATCCATCTCTTCA |
| UCP-2 | GTTCCTCTGTCTCGTCTTGC | GGCCTTGAAACCAACCA |
| CPT-1 | CCAGGCTACAGTGGGACATT | GAACTTGCCCATGTCCTTGT |
| MCAD | GAAGATCAAGCGAGGCTCCA | CCTTCCTGCATTCCATCGTC |
| PEPCK | GTGCTGGAGTGGATGTTCGG | CTGGCTGATTCTCTGTTTCAGG |
| G6pase | ACTGTGGGCATCAATCTCCTC | CGGGACAGACAGACGTTCAGC |
| Adiponectin | GCAGAGATGGCACTCCTGGA | CCCTTCAGCTCCTGTCATTCC |
| TNF-α | TCTCATCAGTTCTATGGCCC | GGGAGTAGACAAGGTACAAC |
| IL-6 | TTCACAGAGGATACCACTCC | GTTTGGTAGCATCCATCATT |
| MCP-1 | GGCTCAGCCAGATGCAGT | GAGCTTGGTGACAAAAACTACAG |
| GAPDH | TGTGTCCGTCGTGGATCTGA | CCTGCTTCACCACCTTCTTGAT |
2.9. Statistical analysis
Values are presented as mean ± SD. Comparisons between experimental groups were performed using one-way ANOVA followed by the Tukey–Kramer test (Statview 5.0; SAS Institute Inc., USA). Means with different letters are different at P < 0.05.
3. Results
3.1. Combination of pioglitazone and fish oil suppresses weight gain associated with subcutaneous fat accumulation in response to pioglitazone in male KK mice
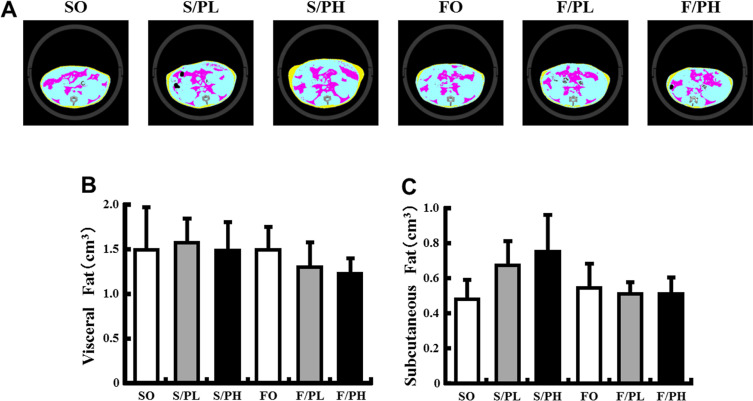
Greater body weight gain was observed in the pioglitazone-treated S/PL and S/PH groups compared with the non-treated SO group; however, F/PL and F/PH groups had approximately 3 g less body weight gain compared with the S/PL and S/PH groups, respectively (Table 3 ). Epididymal WAT weights in the S/PL, FO, F/PL, and F/PH groups were significantly lower than the SO group (Table 3 ). There was a trend toward lower epididymal WAT weights in S/PH compared with the SO group. In pioglitazone-treated groups, BAT weights were almost twice as great compared with non-treated mice. No significant difference in BAT weights was observed between the SO and FO groups (Table 3 ). CT scanning analysis revealed visceral fat mass weights were unchanged in response to either pioglitazone or fish oil (Fig. 1 A, B). However, subcutaneous fat mass in the S/PL and S/PH groups was increased by 40% and 57% compared with the SO group, respectively. The accumulation of subcutaneous fat in the F/PL and F/PH groups was comparable with the SO group, indicating amelioration of the effect of pioglitazone by fish oil (Fig. 1 A, C).
| Group | SO | S/PL | S/PH | FO | F/PL | F/PH |
|---|---|---|---|---|---|---|
| Total food intake (g/mouse) | 267±33 | 270±21 | 258±20 | 277±31 | 255±7 | 242±11 |
| Initial body weight (g) | 29.8±1.6 | 29.8±1.6 | 29.8±1.6 | 29.8±1.5 | 29.8±1.1 | 29.8±1.1 |
| Final body weight (g) | 42.3±2.4 | 45.3±4.1 | 45.9±3.4 | 40.8±2.3 | 42.3±1.7 | 43.2±1.7 |
| Body weight gain (g) | +12.5±1.3a,b | +15.6±2.7a | +16.1±2.6a | +11.0±3.1b | +12.5±2.1a,b | +13.4±1.5a,b |
| Liver weight (g) | 2.54±0.31b | 3.85±0.76a | 2.95±0.47a,b | 2.18±0.25b | 2.77±0.63b | 2.20±0.28b |
| WAT weight (g) | 1.52±0.25a | 1.14±0.25b | 1.21±0.18a,b | 1.11±0.18b | 0.94±0.09b | 1.02±0.06b |
| BAT weight (g) | 0.32±0.04b | 0.83±0.13a | 1.09±0.32a | 0.30±0.06b | 0.68±0.16a,b | 0.96±0.34a |
a. Data are represented as mean ± SD (n = 5).
b. Means with different letters are different at P < 0.05 by Tukey–Kramer test.
|
|
|
Fig. 1. X-ray computed tomography images, visceral fat, and subcutaneous fat in 15-week-old male KK mice. (A) CT images, (B) visceral fat mass, and (C) subcutaneous fat mass. Representative CT scanned cross-sections from mice fed diets containing safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Colored regions represent visceral fat (pink) and subcutaneous fat (yellow). Visceral fat and subcutaneous fat were evaluated in abdominal regions between the second and fourth lumbar vertebrae. Data are represented as mean ± SD (n = 4–5). |
3.2. Combination of pioglitazone and fish oil improves glucose metabolisms in male KK mice
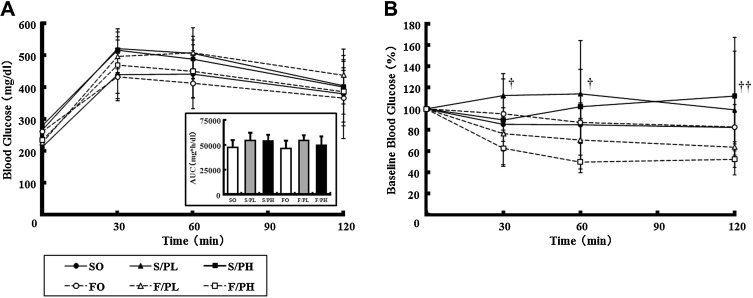
To investigate the effects of the combination of pioglitazone and fish oil on glucose metabolism, we performed OGTT and ITT on obese and diabetic male KK mice. In OGTT, no significant differences in blood glucose levels at 30, 60, or 120 min were observed between groups. No significant differences in blood glucose area under the curve (AUC) values were observed between groups (Fig. 2 A). For ITT, blood glucose levels at 30 min after insulin injections were reduced to approximately 80% and 60% of initial values in the F/PL and F/PH groups, respectively, with differences maintained until 120 min. In the F/PH group, blood glucose levels were significantly lower at 120 min after insulin injection than in the S/PH group (Fig. 2 B).
|
|
|
Fig. 2. Oral glucose tolerance tests (OGTTs) and insulin tolerance tests (ITTs) in 14-week-old male KK mice. (A) Blood glucose levels during OGTT and area under the curve values and (B) blood glucose levels during ITT. OGTT was performed by administration of a 10% glucose solution (1 g/kg) after 3 h of fasting. ITT was performed by intraperitoneal injection of an insulin solution (0.75 U/kg) after 3 h of fasting. Blood samples were obtained immediately prior to glucose or insulin treatment (0 min) and at 30, 60, and 120 min after glucose loading or insulin injection. Data are represented as mean ± SD (n = 5). The Tukey–Kramer test was used for statistical analyses. †, P < 0.05 for comparison between S/PL and F/PH groups; ††, P < 0.05 for comparison between S/PH and F/PH groups. |
Plasma insulin levels were significantly decreased in all pioglitazone-treated groups compared with the SO group. In the FO group, a trend toward decreased plasma insulin levels was observed compared with the SO group (Table 4 ). Similarly, HOMA-IR indices were significantly decreased in the S/PL, S/PH, F/PL, and F/PH groups, with a trend toward a decrease in the FO group, compared with the SO group (Table 4 ). No significant differences in plasma adiponectin levels were observed between the SO and FO groups. However, plasma adiponectin levels were higher in pioglitazone-treated groups compared with non-treated groups, with significant differences observed in some groups. In particular, the F/PH group had a 2.2-fold significant increase in plasma adiponectin levels compared with the S/PH group (Table 4 ).
| Group | SO | S/PL | S/PH | FO | F/PL | F/PH |
|---|---|---|---|---|---|---|
| Blood glucose (mg/dl) | 287±59 | 255±52 | 347±75 | 354±120 | 290±66 | 288±31 |
| Plasma insulin (ng/ml) | 50.2±25.3a | 19.8±7.2b | 6.2±3.9b | 31.4±17.1a,b | 16.9±8.8b | 9.8±4.6b |
| HOMA-IR | 1.00±0.45a | 0.36±0.13b,c | 0.15±0.09c | 0.68±0.35a,b | 0.36±0.22b,c | 0.20±0.10b,c |
| Plasma adiponectin (μg/ml) | 10.3±0.8c | 19.6±2.9b,c | 30.9±8.8b | 12.7±2.3c | 28.9±5.4b | 68.2±12.9a |
| Plasma leptin (ng/ml) | 68.3±14.7 | 64.7±10.1 | 67.8±9.9 | 57.9±9.5 | 63.1±8.1 | 67.7±19.3 |
| Plasma TG (mg/dl) | 193±52a | 146±29a,c | 168±25a,b | 187±24a | 116±23b,c | 93±23c |
| Plasma TC (mg/dl) | 176±21a | 167±15a | 163±15a | 115±11b | 86±9c | 76±5c |
| Plasma HDL-C (mg/dl) | 135±18a | 131±13a | 123±12a | 82±10b | 73±10b | 61±6b |
| Plasma non HDL-C (mg/dl) | 41±7a | 36±8a | 41±9a | 33±3a | 14±5b | 15±2b |
| Plasma FFA (mEq/l) | 0.74±0.12a | 0.56±0.12a,b | 0.50±0.09b | 0.48±0.08b | 0.41±0.10b,c | 0.28±0.05c |
a. Data are represented as mean ± SD (n = 5).
b. HOMA-IR are shown as ratio to SO group.
c. Means with different letters are different at P < 0.05 by Tukey–Kramer test.
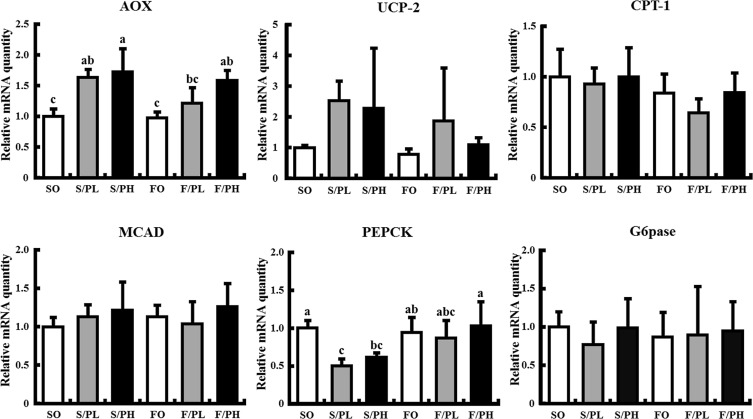
Hepatic expression levels of genes related to gluconeogenesis are shown in Fig. 6 . Phosphoenolpyruvate carboxykinase (PEPCK) mRNA levels were significantly decreased in the S/PL and S/PH groups compared with the SO group, whereas no changes were observed in fish oil-fed groups. No significant differences in glucose-6-phosphatase (G6pase) mRNA levels were observed between all groups.
3.3. Combination of pioglitazone and fish oil improves plasma lipid profiles in male KK mice
To examine the effects of the combination of pioglitazone and fish oil on plasma lipid profiles, plasma lipid levels were measured, with the results shown in Table 4 . No significant differences in triglyceride levels were observed among the SO, S/PL, S/PH, and FO groups. Triglyceride levels were significantly lower in the F/PL and F/PH groups compared with the SO group. No significant differences in total plasma cholesterol levels were observed between pioglitazone-treated and untreated safflower oil-fed groups. Total plasma cholesterol levels were significantly decreased in fish oil-fed groups treated with pioglitazone (F/PL and F/PH) compared with the FO group. HDL-cholesterol levels were significantly decreased in fish oil-fed groups compared with safflower oil-fed groups, regardless of pioglitazone treatment (Table 4 ). Plasma FFA levels were significantly decreased in all groups, except the S/PL group, compared with the SO group. Plasma FFA levels in the F/PH group were significantly lower than in the S/PH group.
3.4. Pioglitazone treatment induces accumulation of liver triglyceride, but fish oil feeding suppresses that in male KK mice
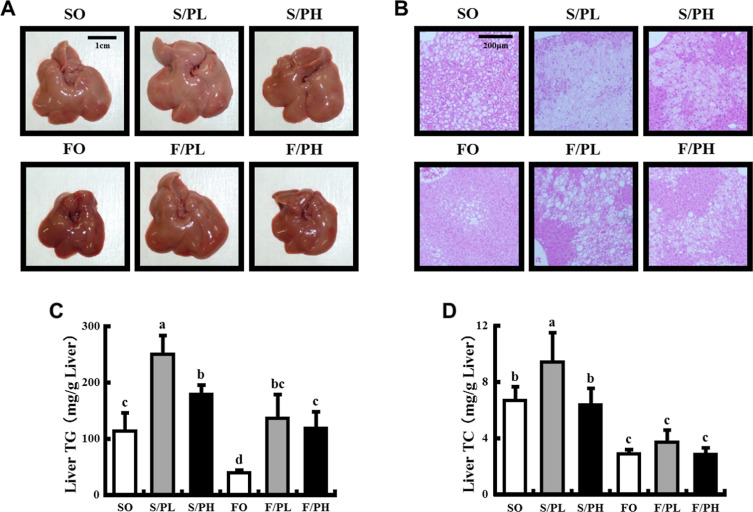
To examine the effects of the combination of pioglitazone and fish oil on hepatic lipid accumulation, we performed histological analysis on liver specimens and measured hepatic lipid contents. Liver tissues had the palest appearance, characteristic of severe steatosis, in the S/PL group (Fig. 3 A). A greater severity steatosis was observed in liver sections from the S/PL and S/PH groups than the SO group, with minimal steatosis observed in the FO group (Fig. 3 B). Similarly, liver triglyceride levels were significantly increased in pioglitazone-treated S/PL (2.2-fold) and S/PH (1.6-fold) groups compared with those in the SO group. Liver triglyceride levels in the FO group were significantly decreased compared with those in the SO group. Increased liver triglyceride levels in response to pioglitazone were significantly reduced to the level of the SO group by the combination of pioglitazone and fish oil (Fig. 3 C). Although liver total cholesterol levels were significantly increased in the S/PL group than the SO group, liver total cholesterol levels in fish oil-fed groups (FO, F/PL, and F/PH) were significantly lower than in the SO group (Fig. 3 D).
|
|
|
Fig. 3. Liver morphology, histological sections, and liver lipid parameters in 15-week-old male KK mice. (A) Liver morphology, (B) liver histology, (C) liver triglyceride levels, and (D) liver total cholesterol levels. Representative macroscopic liver images and tissue sections from mice fed safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Sections were stained by H&E and examined under a microscope at 100-fold magnification. Data are represented as mean ± SD (n = 5). Groups with different letters are different at P < 0.05 by Tukey–Kramer test. |
3.5. Combination of pioglitazone and fish oil ameliorates adipocyte hypertrophy and increases the number of small adipocytes in male KK mice
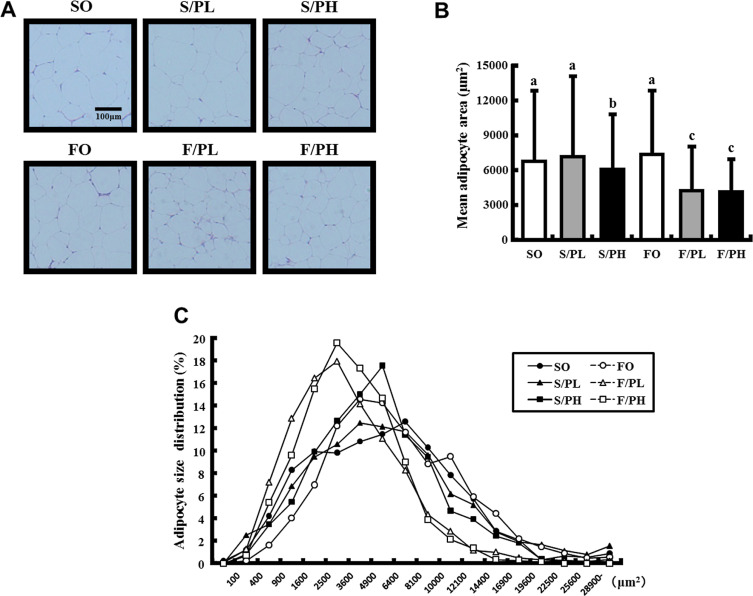
To clarify the interaction between the amelioration of insulin resistance in response to pioglitazone and fish oil treatment and changes in visceral fat parameters, we performed histological analysis on WAT sections (Fig. 4 A). Mean adipocyte areas were significantly lower in the S/PH, F/PL, and F/PH groups compared with groups not treated with pioglitazone. No difference in mean adipocyte areas was observed between the FO group and SO group. Mean adipocyte areas were significantly lower in the F/PH group compared with the S/PH group (Fig. 4 B). Evaluation of adipocyte size distributions demonstrated lower peak sizes in the F/PL and F/PH groups (2500–3600 μm2 ) compared with other groups. The percentages of small adipocytes (400–3600 μm2 ) and larger adipocytes (>6400 μm2 ) were increased and decreased, respectively, in the F/PL and F/PH groups compared with other groups (Fig. 4 C).
|
|
|
Fig. 4. WAT histological sections, mean adipocyte areas, and cell size distributions in 15-week-old male KK mice. (A) Epididymal WAT sections stained with H&E, (B) mean adipocyte areas, and (C) adipocyte size distributions. Representative WAT tissue sections from mice fed safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Sections were stained by H&E and examined under a microscope at 100-fold magnification. Data are represented as mean ± SD (n = 5). Groups with different letters are different at P < 0.05 by Tukey–Kramer test. |
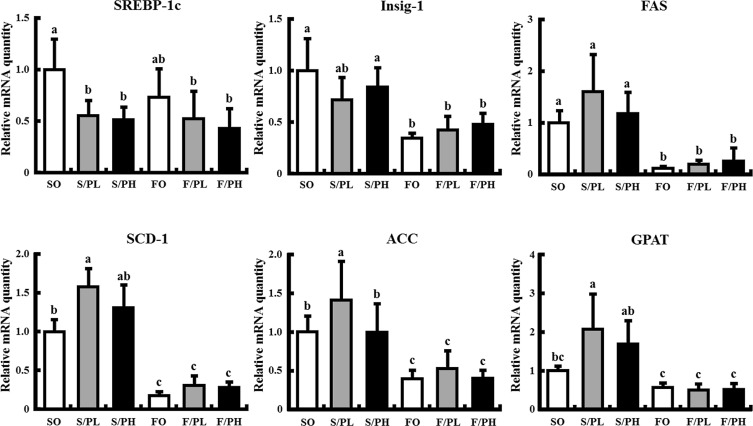
3.6. Fish oil feeding decreases hepatic mRNA levels of genes involved in lipogenesis with or without pioglitazone treatment
To elucidate the mechanisms underlying the amelioration of weight gain and subcutaneous fat accumulation in response to pioglitazone by the combination of pioglitazone and fish oil, we measured hepatic mRNA expression of genes involved in de novo lipogenesis ( Fig. 5 ). SREBP-1c mRNA levels were significantly decreased in all pioglitazone-treated groups compared with the SO group, with no difference observed between the SO and FO groups. Insulin-induced gene 1 (Insig-1) mRNA levels were significantly decreased in fish oil-fed groups. Insig-1 mRNA levels in the F/PL and F/PH groups were approximately 60% lower than in the S/PL and S/PH groups, respectively. Expression levels of SREBP-1c target genes, such as FAS, SCD-1, and ACC, were significantly decreased in fish oil-fed groups, regardless of pioglitazone treatment (Fig. 5 ). In addition, mRNA levels of glycerol-3-phosphate acyltransferase (GPAT), a lipogenic rate-controlling enzyme, were increased in the S/PL and S/PH groups compared with the SO group; however, increased expression of GPAT was markedly suppressed by the combination of pioglitazone with fish oil (Fig. 5 ).
|
|
|
Fig. 5. Hepatic mRNA levels related to lipogenesis in 15-week-old male KK mice. Mice were fed safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Panels show mRNA levels of SREBP-1c, Insig-1, FAS, SCD-1, ACC and GPAT. mRNA levels were determined by real-time quantitative PCR. Results are expressed as the ratio of the obtained value to that of the Safflower oil (SO) group. Data are represented as mean ± SD (n = 5). Groups with different letters are different at P < 0.05 by Tukey–Kramer test. |
|
|
|
Fig. 6. Hepatic mRNA levels related to fatty acid β-oxidation and metabolism, and gluconeogenesis in 15-week-old male KK mice. Mice were fed safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Panels show mRNA levels of AOX, UCP-2, CPT-1, MCAD, PEPCK, and G6pase. mRNA levels were determined by real-time quantitative PCR. Results are expressed as the ratio of the obtained value to that of the Safflower oil (SO) group. Data are represented as mean ± SD (n = 5). Groups with different letters are different at P < 0.05 by Tukey–Kramer test. |
3.7. Pioglitazone, but not fish oil, increases hepatic mRNA levels of genes involved in β-oxidation
To elucidate the association between β-oxidation and suppression of fat accumulation by the combination of pioglitazone and fish oil, we measured hepatic mRNA expression of genes involved in fatty acid β-oxidation and metabolism. AOX mRNA levels were significantly increased in the S/PL, S/PH, and F/PH groups compared with the SO group. No difference in AOX mRNA levels was observed between the SO and FO groups (Fig. 6 ). Although pioglitazone and fish oil did not significantly affect UCP-2 mRNA expression, there was a trend toward increased levels of UCP-2 mRNA levels in the S/PL and S/PH groups compared with the SO group (Fig. 6 ). Carnitine palmitoyltransferase 1 (CPT-1) and medium-chain acyl-CoA dehydrogenase (MCAD) mRNA levels were unaffected by pioglitazone or fish oil (Fig. 6 ).
3.8. Pioglitazone and fish oil decreases cytokine mRNA expression in WAT
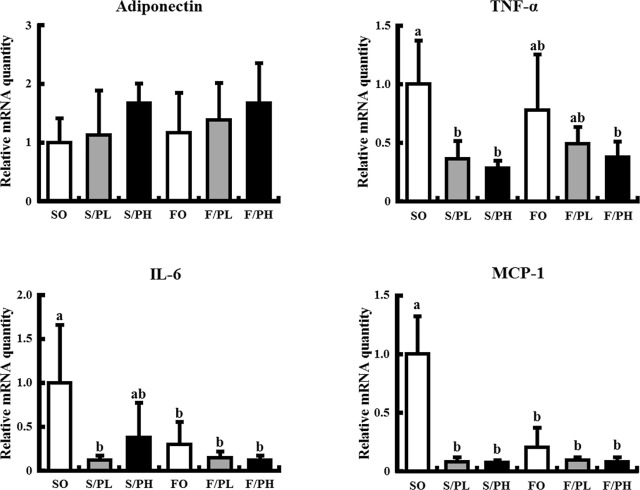
To elucidate the relationship between histological changes and chronic inflammation in response to the combination of pioglitazone and fish oil, cytokine mRNA expression levels were measured in epididymal fat. No significant differences in adiponectin mRNA levels were observed between groups; however, there was a trend toward increased adiponectin expression in the S/PH and F/PH groups compared with the SO group. TNF-α mRNA levels were lower in pioglitazone-treated groups compared with the SO group, with significant differences observed in some groups. No significant difference in TNF-α mRNA levels was observed in response to fish oil. Interleukin 6 (IL-6) mRNA levels were significantly decreased in the S/PL, FO, F/PL, and F/PH groups and tended to attenuate in the S/PH group compared with the SO group. MCP-1 mRNA levels were significantly lower in pioglitazone and/or fish oil treated groups compared with the SO group (Fig. 7 ).
|
|
|
Fig. 7. Cytokine mRNA levels in epididymal fat of 15-week-old male KK mice. Mice were fed safflower oil or fish oil diets without pioglitazone treatment (SO, FO) and treated with low-dose pioglitazone (S/PL, F/PL) or high-dose pioglitazone (S/PH, F/PH) for 8 weeks. Panels show mRNA levels of adiponectin, TNF-α, IL-6, and MCP-1. mRNA levels were determined by real-time quantitative PCR. Results are expressed as the ratio of the obtained value to that of the Safflower oil (SO) group. Data are represented as mean ± SD (n = 5). Groups with different letters are different at P < 0.05 by Tukey–Kramer test. |
4. Discussion
Pioglitazone is widely used as an insulin sensitizer in the treatment of type 2 diabetes. However, body weight gain accompanied by an increase in subcutaneous fat is frequently observed in patients treated with pioglitazone and is considered a major adverse effect of pioglitazone. To address this substantial clinical issue, we examined the utility of fish oil consumption in ameliorating pioglitazone-induced body weight gain and further elucidated the beneficial effects of the combination of pioglitazone and fish oil on glucose and lipid metabolism.
In the present study, we found that the combination of pioglitazone and fish oil ameliorated body weight gain and subcutaneous fat accumulation in response to pioglitazone treatment. In addition, pioglitazone and/or fish oil-treated mice had significantly lower epididymal fat weight. However, no significant differences in visceral fat mass were observed between the treatment groups. Majority of FFAs released from the triglycerides of chylomicron and VLDL particles are re-esterified to triacylglycerol in adipose tissue for storage [37] . Thus, fish oil consumption is thought to decrease triacylglycerol supply to adipose tissue by reducing VLDL production, thereby inhibiting the accumulation of lipids in adipose tissue [33] and [35] . Our results demonstrate fish oil consumption potently decreases mRNA expression of lipogenic enzymes, including FAS, SCD-1, ACC, GPAT, and SREBP-1c. The majority of lipogenic gene expression levels were unchanged or increased in S/PL and S/PH groups despite decreased expression of SREBP-1c mRNA. These results are coincident with previous study used KKAy mice by Oribe et al. [38] . On the other hands, it has reported that pioglitazone inhibits nuclear translocation of SREBP-1c and represses lipogenic gene expression in lean C57BL6/J mice [39] . These data showed that lipogenic gene expressions in response to pioglitazone vary considerably depending on obesity or non-obesity.
Expression levels of fatty acid β-oxidation related genes, including AOX, UCP-2, CPT-1, and MCAD, were unaffected by fish oil consumption. These results suggest diets with 10% of total calories provided by fish oil had no effect on β-oxidation in the present study. Our previous studies demonstrated diets with 2% of total calories provided by fish oil significantly increased AOX and UCP-2 mRNA levels in C57BL/6 mice [40] ; however, diets with 2–25% of total calories provided by fish oil were not found to stimulate β-oxidation in obesity-induced KK mice fed a high fat diet [41] . These reports suggested fish oil-induced effects on stimulating fatty acid β-oxidation may be attenuated by obesity in female KK mice. In the present study used male KK mice, also, diets with 10% of total calories provided by fish oil were found to potently inhibit lipogenesis without activation of fatty acid β-oxidation, resulting in decreased accumulation of subcutaneous fat and body weight gain in response to pioglitazone treatment.
TZDs have been reported to ameliorate IR by increasing plasma adiponectin levels in obese and diabetes murine models and clinical trials [12] , [42] , [43] and [44] . Our data demonstrated plasma adiponectin levels were increased in pioglitazone-treated groups with significantly decreased HOMA-IR indices compared with corresponding pioglitazone-untreated groups. On the other hand, fish oil treatment alone was found to have no marked effect on these parameters. Fish oil supplementation reportedly increases circulating adiponectin levels in non-obese rodents [45] and [46] . However, our previous study demonstrated diets with 2.5%, 12.5%, and 25% of total calories provided by fish oil had no beneficial effects on plasma adiponectin and insulin levels in KK mice [41] . These findings suggest the antidiabetic effects of fish oil related to circulating adiponectin levels may be attenuated by obesity. In the present study, adiponectin levels in the F/PH group were 2.2-fold higher than in the S/PH group despite few notable effects on other parameters observed. Increased plasma adiponectin levels may contribute to the amelioration of IR in male KK mice; however, adiponectin levels above a certain level may not necessarily correspond to an insulin sensitizing effect.
The KK mouse model has significantly greater severity of hyperinsulinemia and IR compared with wild-type mice, with maximum dysfunction observed at 16–20 months of age [47] . In the present study, treatment of male KK mice with pioglitazone led to significantly decreased plasma insulin levels and improved IR compared with pioglitazone-untreated groups. However, these changes may be insufficient to greatly decrease blood glucose levels in OGTT.
Pro-inflammatory cytokines produced by adipose tissue macrophages (ATMs) in adipose tissue are closely associated with obesity and IR [48] . ATMs can be classified into two phenotypes: the pro-inflammatory M1 phenotype and the anti-inflammatory M2 phenotype. M1 ATMs are seen to accumulate in obese adipose tissue and secrete pro-inflammatory cytokines including TNF-α, IL-6, and MCP-1. M1 ATMs have been shown to promote adipose tissue lipolysis via activation of mitogen-activated protein kinases resulting in increased FFA production, particularly saturated fatty acids. In addition, the FFAs have been shown to act as a ligand for toll-like receptor-4 (TLR-4) expressed by macrophages and cause further exacerbation of chronic inflammation in adipose tissue [49] . Dietary n -3 polyunsaturated fatty acids, such as EPA and DHA, inhibited TLR-induced inflammatory signaling pathways [50] . And, pioglitazone treatment increased the ratio of M2-to-M1 ATMs in epididymal fat of diet-induced obese mice, and changes in the number of ATMs were closely correlated with IR [51] . In this study, fish oil consumption or/and pioglitazone treatment decreased expression of pro-inflammatory cytokines, such as TNF-α, IL-6, and MCP-1, in epididymal fat. Our results suggest that fish oil and/or pioglitazone ameliorated inflammation in adipose tissue and improved IR.
Hepatic lipid accumulation is closely related to IR and compensatory hyperinsulinemia. TZDs have been reported to potently stimulate the differentiation of adipocytes and induce a redistribution of FFAs from extra-adipose tissues, including the liver and pancreas, to newly derived adipocytes, thereby ameliorating whole-body IR and fatty liver [6] and [18] . In contrast, several studies have demonstrated the anti-steatotic effect of TZDs in both animals and humans [19] , [39] and [52] . Bedoucha et al. reported markedly increased hepatic levels of PPARγ mRNA and protein in diabetic KKAy and ob/ob mice compared with lean C57BL6/J mice [53] . Further, prominent microvesicular steatosis due to lipid accumulation was observed in KKAy mice treated with TZDs, suggesting obese mice are more susceptible to TZD-induced steatosis than lean mice. In the present study, pioglitazone monotherapy induced dramatic hepatic triglyceride accumulation in male KK mice, particularly in the S/PL group, in addition to stimulating expression of genes related to fatty acid synthesis. And, pioglitazone-induced hepatic lipogenesis was potently inhibited by fish oil consumption, contributing to the efficacy of the combination of TZD and fish oil in the treatment of type 2 diabetes.
5. Conclusions
In conclusion, we demonstrated that the combination of pioglitazone and fish oil may prevent the adverse effects of pioglitazone treatment on promoting excessive body weight gain and subcutaneous fat accumulation. Fish oil consumption was found to potently inhibit hepatic lipogenesis in KK mice. The results of this study suggest that the combination of pioglitazone and fish oil has particular efficacy in diabetic patients with fatty liver and adipose tissue inflammation. Thus, we provide novel data regarding the utility of nutritional therapy in patients with type 2 diabetes being treated with pioglitazone.
Conflict of interest
The authors have nothing to declare.
Transparency document
Acknowledgement
This research was supported by JSPS KAKENHI Grant Number 25504012 .
References
- [1] D.M. Nathan; Clinical practice: initial management of glycemia in type 2 diabetes mellitus; N. Engl. J. Med., 347 (2002), pp. 1342–1349
- [2] K. Cusi; The role of adipose tissue and lipotoxicity in the pathogenesis of type 2 diabetes; Curr. Diabetes Rep., 10 (2010), pp. 306–315
- [3] G.S. Hotamisligil; Inflammation and metabolic disorders; Nature, 444 (2006), pp. 860–867
- [4] R.H. Eckel, S.M. Grundy, P.Z. Zimmet; The metabolic syndrome; Lancet, 365 (2005), pp. 1415–1428
- [5] A. Ito, T. Suganami, Y. Miyamoto, Y. Yoshimasa, M. Takeya, Y. Kamei, Y. Ogawa; Role of MAPK phosphatase-1 in the induction of monocyte chemoattractant protein-1 during the course of adipocyte hypertrophy; J. Biol. Chem., 282 (2007), pp. 25445–25452
- [6] P. Kovacs, M. Stumvoll; Fatty acids and insulin resistance in muscle and liver; Best Pract. Res. Clin. Endocrinol. Metabol., 19 (2005), pp. 625–635
- [7] R.H. Unger; Lipid overload and overflow: metabolic trauma and the metabolic syndrome; Trends Endocrinol. Metabol., 14 (2003), pp. 398–403
- [8] Y. Arita, S. Kihara, N. Ouchi, M. Takahashi, K. Maeda, J. Miyagawa, K. Hotta, I. Shimomura, T. Nakamura, K. Miyaoka, H. Kuriyama, M. Nishida, S. Yamashita, K. Okubo, K. Matsubara, M. Muraguchi, Y. Ohmoto, T. Funahashi, Y. Matsuzawa; Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity; Biochem. Biophys. Res. Commun., 257 (1999), pp. 79–83
- [9] B.J. Goldstein; Differentiating members of the thiazolidinedione class: a focus on efficacy; Diabetes Metab. Res. Rev. (Suppl. 2) (2002), pp. S16–S22
- [10] A. Okuno, H. Tamemoto, K. Tobe, K. Ueki, Y. Mori, K. Iwamoto, K. Umesono, Y. Akanuma, T. Fujiwara, H. Horikoshi, Y. Yazaki, T. Kadowaki; Troglitazone increases the number of small adipocytes without the change of white adipose tissue mass in obese zucker rats; J. Clin. Invest., 101 (1998), pp. 1354–1361
- [11] T. Yamauchi, J. Kamon, H. Waki, K. Murakami, K. Motojima, K. Komeda, T. Ide, N. Kubota, Y. Terauchi, K. Tobe, H. Miki, A. Tsuchida, Y. Akanuma, R. Nagai, S. Kimura, T. Kadowaki; The mechanisms by which both heterozygous peroxisome proliferator-activated receptor gamma (PPARgamma) deficiency and PPARgamma agonist improve insulin resistance; J. Biol. Chem., 276 (2001), pp. 41245–41254
- [12] T. Yamauchi, J. Kamon, H. Waki, Y. Terauchi, N. Kubota, K. Hara, Y. Mori, T. Ide, K. Murakami, N. Tsuboyama-Kasaoka, O. Ezaki, Y. Akanuma, O. Gavrilova, C. Vinson, M.L. Reitman, H. Kagechika, K. Shudo, M. Yoda, Y. Nakano, K. Tobe, R. Nagai, S. Kimura, M. Tomita, P. Froguel, T. Kadowaki; The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity; Nat. Med., 7 (2001), pp. 941–946
- [13] I. Bogacka, H. Xie, G.A. Bray, S.R. Smith; The effect of pioglitazone on peroxisome proliferator-activated receptor-gamma target genes related to lipid storage in vivo; Diabetes Care, 27 (2004), pp. 1660–1667
- [14] K. Yajima, H. Hirose, H. Fujita, Y. Seto, H. Fujita, K. Ukeda, K. Miyashita, T. Kawai, Y. Yamamoto, T. Ogawa, T. Yamada, T. Saruta; Combination therapy with PPARgamma and PPARalpha agonists increases glucose-stimulated insulin secretion in db/db mice; Am. J. Physiol. Endocrinol. Metabol., 284 (2003), pp. E966–E971
- [15] H. Ishida, M. Takizawa, S. Ozawa, Y. Nakamichi, S. Yamaguchi, H. Katsuta, T. Tanaka, M. Maruyama, H. Katahira, K. Yoshimoto, E. Itagaki, S. Nagamatsu; Pioglitazone improves insulin secretory capacity and prevents the loss of beta-cell mass in obese diabetic db/db mice: possible protection of beta cells from oxidative stress; Metabolism, 53 (2004), pp. 488–494
- [16] S. Aronoff, S. Rosenblatt, S. Braithwaite, J.W. Egan, A.L. Mathisen, R.L. Schneider; Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes: a 6-month randomized placebo-controlled dose-response study. The Pioglitazone 001 Study Group; Diabetes Care, 23 (2000), pp. 1605–1611
- [17] R. Kawamori, T. Kadowaki, M. Onji, Y. Seino, Y. Akanuma, PRACTICAL Study Group; Hepatic safety profile and glycemic control of pioglitazone in more than 20,000 patients with type 2 diabetes mellitus: postmarketing surveillance study in Japan; Diabetes Res. Clin. Pract., 76 (2007), pp. 229–235
- [18] R. Vuppalanchi, N. Chalasani; Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: selected practical issues in their evaluation and management; Hepatology, 49 (2009), pp. 306–317
- [19] R. Belfort, S.A. Harrison, K. Brown, C. Darland, J. Finch, J. Hardies, B. Balas, A. Gastaldelli, F. Tio, J. Pulcini, R. Berria, J.Z. Ma, S. Dwivedi, R. Havranek, C. Fincke, R. DeFronzo, G.A. Bannayan, S. Schenker, K. Cusi; A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis; N. Engl. J. Med., 355 (2006), pp. 2297–2307
- [20] F.M. Martens, T.J. Rabelink, J. op ‘t Roodt, E.J. de Koning, F.L. Visseren; TNF-alpha induces endothelial dysfunction in diabetic adults: an effect reversible by the PPAR-gamma agonist pioglitazone; Eur. Heart J., 27 (2006), pp. 1605–1609
- [21] T. Kadowaki, T. Yamauchi, N. Kubota, K. Hara, K. Ueki, K. Tobe; Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome; J. Clin. Invest., 116 (2006), pp. 1784–1792
- [22] H. Yki-Järvinen; Thiazolidinediones; N. Engl. J. Med., 351 (2004), pp. 1106–1118
- [23] C.J. de Souza, M. Eckhardt, K. Gagen, M. Dong, W. Chen, D. Laurent, B.F. Burkey; Effects of pioglitazone on adipose tissue remodeling within the setting of obesity and insulin resistance; Diabetes, 50 (2001), pp. 1863–1871
- [24] S.E. Nissen, S.J. Nicholls, K. Wolski, R. Nesto, S. Kupfer, A. Perez, H. Jure, R. De Larochellière, C.S. Staniloae, K. Mavromatis, J. Saw, B. Hu, A.M. Lincoff, E.M. Tuzcu, IPERISCOPE Investigators; Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes: the PERISCOPE randomized controlled trial; JAMA, 299 (2008), pp. 1561–1573
- [25] S. Hallakou, L. Doaré, F. Foufelle, M. Kergoat, M. Guerre-Millo, M.F. Berthault, I. Dugail, J. Morin, J. Auwerx, P. Ferré; Pioglitazone induces in vivo adipocyte differentiation in the obese Zucker fa/fa rat; Diabetes, 46 (1997), pp. 1393–1399
- [26] Y. Miyazaki, A. Mahankali, M. Matsuda, S. Mahankali, J. Hardies, K. Cusi, L.J. Mandarino, R.A. DeFronzo; Effect of pioglitazone on abdominal fat distribution and insulin sensitivity in type 2 diabetic patients; J. Clin. Endocrinol. Metabol., 87 (2002), pp. 2784–2791
- [27] P.J. Nestel; Effects of N-3 fatty acids on lipid metabolism; Annu. Rev. Nutr., 10 (1990), pp. 149–167
- [28] M. Capanni, F. Calella, M.R. Biagini, S. Genise, L. Raimondi, G. Bedogni, G. Svegliati-Baroni, F. Sofi, S. Milani, R. Abbate, C. Surrenti, A. Casini; Prolonged n -3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: a pilot study ; Aliment Pharmacol. Ther., 23 (2006), pp. 1143–1151
- [29] A.T. Erkkilä, A.H. Lichtenstein, D. Mozaffarian, D.M. Herrington; Fish intake is associated with a reduced progression of coronary artery atherosclerosis in postmenopausal women with coronary artery disease; Am. J. Clin. Nutr., 80 (2004), pp. 626–632
- [30] A. Zampolli, A. Bysted, T. Leth, A. Mortensen, R. De Caterina, E. Falk; Contrasting effect of fish oil supplementation on the development of atherosclerosis in murine models; Atherosclerosis, 184 (2006), pp. 78–85
- [31] D. Eberlé, B. Hegarty, P. Bossard, P. Ferré, F. Foufelle; SREBP transcription factors: master regulators of lipid homeostasis; Biochimie, 86 (2004), pp. 839–848
- [32] H. Kim, M. Takahashi, O. Ezaki; Fish oil feeding decreases mature sterol regulatory element-binding protein 1 (SREBP-1) by down-regulation of SREBP-1c mRNA in mouse liver. A possible mechanism for down-regulation of lipogenic enzyme mRNAs; J. Biol. Chem., 274 (1999), pp. 25892–25898
- [33] T. Nakatani, H. Kim, Y. Kaburagi, K. Yasuda, O. Ezaki; A low fish oil inhibits SREBP-1 proteolytic cascade, while a high-fish-oil feeding decreases SREBP-1 mRNA in mice liver: relationship to anti-obesity; J. Lipid Res., 44 (2003), pp. 369–379
- [34] N. Tsuboyama-Kasaoka, M. Takahashi, H. Kim, O. Ezaki; Up-regulation of liver uncoupling protein-2 mRNA by either fish oil feeding or fibrate administration in mice; Biochem. Biophys. Res. Commun., 257 (1999), pp. 879–885
- [35] T. Arai, H. Kim, H. Chiba, A. Matsumoto; Anti-obesity effect of fish oil and fish oil-fenofibrate combination in female KK mice; J. Atheroscler. Thromb., 16 (2009), pp. 674–683
- [36] J. Folch, M. Lees, G.H. Sloane Stanley; A simple method for the isolation and purification of total lipides from animal tissues; J. Biol. Chem., 226 (1957), pp. 497–509
- [37] J. Bremer; The biochemistry of hypo- and hyperlipidemic fatty acid derivatives: metabolism and metabolic effects; Prog. Lipid. Res., 40 (2001), pp. 231–268
- [38] J. Oribe, T. Kakuma, M. Haranaka, K. Okamoto, M. Seike, H. Yoshimatsu; Intraperitoneal administration attenuates thiazolidinedione-induced hepatic steatosis in KKAy mice with increased hepatic peroxisome proliferator-activated receptor (PPAR)γ mRNA expression; Obes. Res. Clin. Pract., 6 (2012), pp. e175–262
- [39] A. Da Silva Morais, V. Lebrun, J. Abarca-Quinones, S. Brichard, L. Hue, B. Guigas, B. Viollet, I.A. Leclercq; Prevention of steatohepatitis by pioglitazone: implication of adiponectin-dependent inhibition of SREBP-1c and inflammation; J. Hepatol., 50 (2009), pp. 489–500
- [40] S. Hirako, H. Kim, S. Shimizu, H. Chiba, A. Matsumoto; Low-dose fish oil consumption prevents hepatic lipid accumulation in high cholesterol diet fed mice; J. Agric. Food Chem., 59 (2011), pp. 13353–13359
- [41] T. Arai, H. Kim, S. Hirako, M. Nakasatomi, H. Chiba, A. Matsumoto; Effects of dietary fat energy restriction and fish oil feeding on hepatic metabolic abnormalities and insulin resistance in KK mice with high-fat diet-induced obesity; J. Nutr. Biochem., 24 (2013), pp. 267–273
- [42] N. Maeda, M. Takahashi, T. Funahashi, S. Kihara, H. Nishizawa, K. Kishida, H. Nagaretani, M. Matsuda, R. Komuro, N. Ouchi, H. Kuriyama, K. Hotta, T. Nakamura, I. Shimomura, Y. Matsuzawa; PPARgamma ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein; Diabetes, 50 (2001), pp. 2094–2099
- [43] J.G. Yu, S. Javorschi, A.L. Hevener, Y.T. Kruszynska, R.A. Norman, M. Sinha, J.M. Olefsky; The effect of thiazolidinediones on plasma adiponectin levels in normal, obese, and type 2 diabetic subjects; Diabetes, 51 (2002), pp. 2968–2974
- [44] U.B. Pajvani, M. Hawkins, T.P. Combs, M.W. Rajala, T. Doebber, J.P. Berger, J.A. Wagner, M. Wu, A. Knopps, A.H. Xiang, K.M. Utzschneider, S.E. Kahn, J.M. Olefsky, T.A. Buchanan, P.E. Scherer; Complex distribution, not absolute amount of adiponectin, correlates with thiazolidinedione-mediated improvement in insulin sensitivity; J. Biol. Chem., 279 (2004), pp. 12152–12162
- [45] S. Neschen, K. Morino, J.C. Rossbacher, R.L. Pongratz, G.W. Cline, S. Sono, M. Gillum, G.I. Shulman; Fish oil regulates adiponectin secretion by a peroxisome proliferator-activated receptor-gamma-dependent mechanism in mice; Diabetes, 55 (2006), pp. 924–928
- [46] A.S. Rossi, Y.B. Lombardo, J.M. Lacorte, A.G. Chicco, C. Rouault, G. Slama, S.W. Rizkalla; Dietary fish oil positively regulates plasma leptin and adiponectin levels in sucrose-fed, insulin-resistant rats; Am. J. Physiol. Regul. Integr. Comp. Physiol., 289 (2005), pp. R486–R494
- [47] K. Srinivasan, P. Ramarao; Animal models in type 2 diabetes research: an overview; Indian J. Med. Res., 125 (2007), pp. 451–472
- [48] P. Dandona, A. Aljada, A. Bandyopadhyay; Inflammation: the link between insulin resistance, obesity and diabetes; Trends Immunol., 25 (2004), pp. 4–7
- [49] T. Suganami, Y. Ogawa; Adipose tissue macrophages: their role in adipose tissue remodeling; J. Leukoc. Biol., 88 (2010), pp. 33–39
- [50] J.Y. Lee, A. Plakidas, W.H. Lee, A. Heikkinen, P. Chanmugam, G. Bray, D.H. Hwang; Differential modulation of Toll-like receptors by fatty acids: preferential inhibition by n -3 polyunsaturated fatty acids ; J. Lipid Res., 44 (2003), pp. 479–486
- [51] S. Fujisaka, I. Usui, A. Bukhari, M. Ikutani, T. Oya, Y. Kanatani, K. Tsuneyama, Y. Nagai, K. Takatsu, M. Urakaze, M. Kobayashi, K. Tobe; Regulatory mechanisms for adipose tissue M1 and M2 macrophages in diet-induced obese mice; Diabetes, 58 (2009), pp. 2574–2582
- [52] I.A. Leclercq, V.A. Lebrun, P. Stärkel, Y.J. Horsmans; Intrahepatic insulin resistance in a murine model of steatohepatitis: effect of PPARgamma agonist pioglitazone; Lab. Invest., 87 (2007), pp. 56–65
- [53] M. Bedoucha, E. Atzpodien, U.A. Boelsterli; Diabetic KKAy mice exhibit increased hepatic PPARgamma1 gene expression and develop hepatic steatosis upon chronic treatment with antidiabetic thiazolidinediones; J. Hepatol., 35 (2001), pp. 17–23
Document information
Published on 12/05/17
Accepted on 12/05/17
Submitted on 12/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?