Summary
Gastritis cystica polyposa is relatively rare and characterized by polypoid hyperplasia and cystic dilatation of the gastric glands in stomach. Most cases are related to previous gastric surgeries, but a few cases have been reported in unoperated stomachs. We present a 34-year-old man who had anemic symptoms with melena and exertional dyspnea for 3 weeks. He denied any surgical history. An esophagogastroduodenoscopy revealed diffuse giant folds extending from the lower to the upper body of the stomach with nodularity and no obvious bleeding site. A pathologic diagnosis of a punch biopsy specimen from the giant folds revealed only moderately active chronic inflammation with a high Helicobacter pylori density. After serial studies, the patient received a whole layer gastric biopsy during a laparoscopy. Gastritis cystica polyposa was diagnosed on the pathology report. Our present case highlights the rare clinical and endoscopic condition of gastritis cystica polyposa in an unoperated stomach.
Keywords
Gastritis cystica polyposa ; Gastritis cystica profunda ; Neoplasm
Introduction
Gastritis cystica polyposa (GCP) is a rare disease characterized by polypoid hyperplasia of the gastric mucosa. GCP is commonly found as a polypoid mass on the gastric side of a gastroenterostomic anastomosis [1] . Only a few case reports have noted its development in an unoperated stomach. The presenting symptoms may include epigastric pain, abdominal fullness, gastrointestinal bleeding, and weight loss. The proposed etiologies of GCP in an operated stomach may be related to submucosal migration from mucosal cells at an anastomotic site or repair of preanastomotic gastric mucosa damage [2] . Differentiating GCP from cancer is usually difficult in radiological and endoscopic findings. The overall outcome also suggests that GCP may be a precancerous lesion, and therefore surgical intervention for tissue proof and removal of the lesions may be necessary [1] ; [3] .
We report a 34-year-old man with GCP because of the rare occurrence of this lesion unrelated to gastric surgery, the relative young age of this patient compared with most patients, and the presence of anemia rather than abdominal pain as the main clinical symptom. An esophagogastroduodenoscopy (EGD) showed diffuse dominant giant folds as presentation of polypoid hyperplasia. Abdominal computed tomography (CT), upper gastrointestinal series radiology, and endoscopic ultrasonography (EUS) did not show a definite diagnosis. Finally, we made a diagnosis with a whole layer gastric biopsy under laparoscopy.
Case report
A 34-year-old man complained of melena for 3 weeks. Other symptoms included exertional dyspnea and dizziness. Neither abdominal pain nor fullness was reported, and he had no special medical history or surgical history. On physical examination, his conjunctiva was anemic. His abdomen appeared soft and nontender with normoactive bowel sounds. Laboratory findings showed anemia with a hemoglobin of 5.4 g/dL, whereas other blood cell counts, biochemical tests, and the coagulation profile were within normal limits.
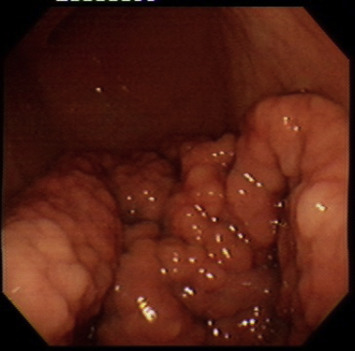
EGD showed diffuse giant folds extending from the lower to the upper body of the stomach with nodularity and an intact surface (Fig. 1 ), and the texture appeared soft during punch with a biopsy forceps. The pathologic diagnosis of the punch biopsy specimens revealed a moderately active chronic inflammation with a high Helicobacter pylori density.
|
|
|
Figure 1. Endoscopic finding in this gastritis cystica polyposa (GCP) patient. Diffuse giant folds extending from the lower to upper body with nodularity and intact surface. |
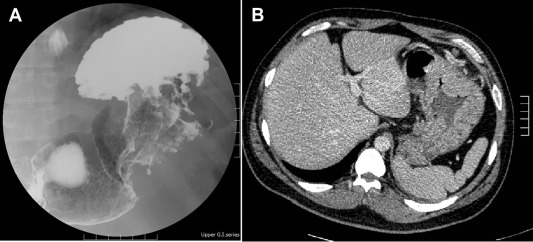
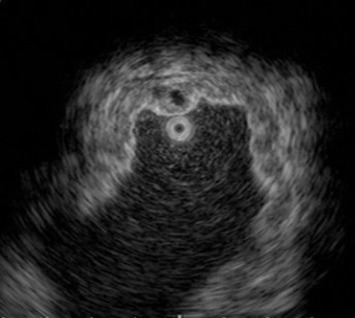
A barium upper gastrointestinal series radiography showed a suspected gastric tumor in the upper body with poor peristalsis and thickened mucosa (Fig. 2 A). An abdominal CT scan also revealed marked folds thickening in the gastric body without any obvious subserosal involvement. The impression from the radiologist was gastric cancer (Fig. 2 B). An EUS demonstrated circumferential thickening (up to 14.1 mm) of the second and third layers of the gastric wall with some anechoic cystic lesions (Fig. 3 ).
|
|
|
Figure 2. (A) Barium fluoroscopic upper gastrointestinal series and (B) upper abdominal computed tomography. (A) Barium fluoroscopic upper gastrointestinal series reports suspicious gastric tumor of high body with poor peristalsis and thickening mucosa. (B) Abdominal computed tomography reveals marked folds thickening at gastric body without obvious subserosal involvement. |
|
|
|
Figure 3. Endoscopic ultrasonography imaging. Circumferential thickening of second and third layers of gastric wall with some anechoic cystic lesions. |
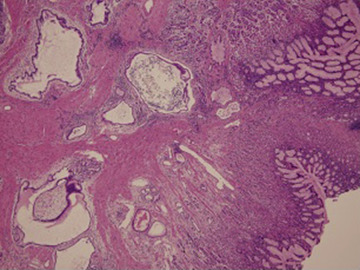
Because the studies above produced no definite diagnosis and there was a suspected malignancy, a whole layer gastric biopsy was performed under laparoscopy. Histological examination showed cystically dilated, bland-appearing, pyloric-like glandular structures in the mucosa and submucosa (Fig. 4 ). Based on these findings, GCP was diagnosed.
|
|
|
Figure 4. Laparoscopic gastric whole layer biopsy (hematoxylin and eosin, ×100). Cystically dilated, bland-appearing, pyloric-like glandular structures in the mucosa and submucosa. |
No melena was noted after laparoscopic intervention. The patient refused further surgical intervention and only received H. pylori eradication therapy. The patient was followed up for 3 months without symptoms, but he refused regular EGD surveys and was then lost to follow-up.
Discussion
GCP appears as a polypoid lesion under endoscopic view. Since 1972 when the entity was first described by Littler and Gleibermann [2] , several synonyms have been used, including gastritis cystica profunda, multiple polypoid cystic gastritis, gastric cystica polyposis, and heterotopic submucosal cysts. GCP involves elongation of the gastric foveola along with hyperplasia and cystic dilatation of the gastric glands [1] . It can be further divided into two histological patterns: (1) gastritis cystica superficialis, in which cystic glands are limited to the mucosal layer; and (2) gastritis cystica profunda, in which there is cystic gland infiltration into the submucosal layer [2] .
GCP is a rare diagnosis. Choi et al [4] reported a retrospective study of 10,728 patients with gastric cancer who underwent gastric cancer surgery between 2000 and 2009 in South Korea. The incidence of gastritis cystica profunda was 1.5% (n = 161), of which the incidence of gastritis cystica profunda was 9.1% (10 of 110 patients) in those with remnant gastric cancer, and 1.4% (151 of 10,618 patients) in those with nonremnant gastric cancer [4] . Laratta et al [5] reviewed published cases of gastritis cystica profunda from 1972 to 2011, and found that only 35% (n = 13) of 37 patients had no prior gastric surgery. The male/female ratio was 3.6:1, and the overall mean age was 60.5 years (range 39–81 years). Of all cases, 62% (n = 23) were located in the body and 24% (n = 9) were in the fundus. The presenting symptoms include epigastric pain and abdominal fullness (27%), gastrointestinal bleeding (16%), and weight loss (8%). Gastritis cystica profunda was an incidental finding in 19% of cases [5] .
GCP develops more often in patients with an operated stomach. Etiologies are presumably due to mucosal elements migrating into the submucosa at the anastomotic site or repair of the preanastomotic gastric mucosa after damage caused by the reflux of duodenal contents [2] .
Barium upper gastrointestinal series radiography may demonstrate obstruction in postgastrectomic patients. GCP generally lacks mucosal erosions, ulcerations, marked fibrosis, or firm consistency, which can occur with gastric adenocarcinoma, and less commonly with gastrointestinal stromal tumor (GIST) [5] . CT can show a heterogeneously iso- to hypoattenuating intraluminal lesion with multiple small cysts (several millimeters in diameter). The cysts in GCP may differentiate it from gastric adenocarcinoma, but the appearance may be similar to GIST [6] . On EUS, GCP can appear as a polymorphic, homogeneous cystic mass with a minimal solid component within the gastric mucosa [3] ; [7] . Because the mucosa is spared in GCP, a submucosal fine-needle aspiration may be required for a definite diagnosis, but nondiagnostic results are often reported [5] .
GCP is rare in the unoperated stomach, and the cause may be congenital in origin in patients with no prior gastric ulceration or trauma history [8] . Park et al [3] first published a case report of a pedunculated GCP in an unoperated stomach, which was removed by endoscopic polypectomy without complications. In another case report, an endoscopic polypectomy was performed on a sessile, wide-based GCP in an unoperated stomach, but late wound bleeding with symptoms of hematemesis and melena were noted 14 days after polypectomy [9] . An endoscopic follow-up conducted 6 months later demonstrated no recurrence in these two cases.
Case reports have also revealed the possibility of a malignant transformation from GCP, even in an unoperated stomach, but the incidence of malignancy in GCP patients remains unknown [6] ; [10] . Choi et al [4] reported a significantly higher rate of Epstein– Barr virus in cancer tissue with gastritis cystica profunda (31.1%) than in that without gastritis cystica profunda (5.8%), and its possible role as a premalignant factor requires further clarification.
One case report from South Korea revealed a gastric adenoma associated with gastritis cystica profunda in an unoperated patient, and the sizes of both the gastritis cystica profunda and the adenoma overlying it had increased during a 10-year follow-up period. Of note, the gastritis cystica profunda enlarged from 3 cm to 4 cm in diameter over the last 3 years of this period). They performed endoscopic submucosal dissection of the adenoma, and no malignancy was noted in a specimen from that procedure [11] .
Surgical intervention for removal of GCP is considered for the following reasons: (1) potential malignancy, (2) obstruction of a gastroenterostomy site, (3) symptomatic anemia and (4) inability to make a definite diagnosis with radiologic study or endoscopic biopsy [6] . Endoscopic polypectomy for GCP [3] ; [9] and endoscopic submucosal dissection for GCP-related early cancer [10] are also possible in selected patients. Data on the long-term follow-up of patients with GCP removal are still lacking.
In conclusion, GCP is a rare gastric tumor found more often at a gastroenteric anastomosis and less often in an unoperated stomach. GCP is more common in men than women and typically presents with nonspecific symptoms. Imaging study including upper gastrointestinal series radiography, abdominal CT, and EUS may provide some information that can differentiate GCP from GIST and adenocarcinoma. Although GCP is benign, it may progress to malignancy, and no cases of local recurrence or distant metastasis for GCP following definitive surgical excision have been reported. Surgical excision may be considered under severe clinical symptoms or suspicious malignant change.
Conflicts of interest
All authors declare no conflicts of interest.
References
- [1] M.T. Wu, H.B. Pan, P.H. Lai, J.M. Chang, S.H. Tsai, C.W. Wu; CT of gastritis cystica polyposa; Abdom Imaging, 19 (1994), pp. 8–10
- [2] E.R. Littler, E. Gleibermann; Gastritis cystica polyposa; Cancer, 29 (1972), pp. 205–209
- [3] J.S. Park, S.J. Myung, H.Y. Jung, S.K. Yang, W.S. Hong, J.H. Kim, et al.; Endoscopic treatment of gastritis cystica polyposa found in an unoperated stomach; Gastrointest Endosc, 54 (2001), pp. 101–103
- [4] M.G. Choi, J.Y. Jeong, K.M. Kim, J.M. Bae, J.H. Noh, T.S. Sohn, et al.; Clinical significance of gastritis cystica profunda and its association with Epstein–Barr virus in gastric cancer; Cancer, 118 (2012), pp. 5227–5233
- [5] J.L. Laratta, T.N. Buhtoiarova, L.S. Sparber, R.S. Chamberlain; Gastritis cystica profunda: a rare gastric tumor masquerading as a malignancy; Surg Sci, 3 (2012), pp. 158–164
- [6] V.B. Kalra, J.W. Gilbert, K.A. Mitchell, R.R. Salem, G.M. Israel; AIRP best cases in radiologic–pathologic correlation: gastritis cystica polyposa; Radiographics, 33 (2013), pp. 109–114
- [7] R. Shudo, K. Horita, K. Takahashi, T. Saito, K. Waku, Y. Aoyanagi; A case of gastritis cystica polyposa showing a characteristic endoscopic ultrasonogram; Dig Endosc, 12 (2000), pp. 68–72
- [8] S. Gensler, B. Seidenberg, H. Rifkin, B.M. Rubinstein; Ciliated lined intramural cyst of the stomach: case report and suggested embryogenesis; Ann Surg, 163 (1966), pp. 954–956
- [9] K. Tuncer, M. Alkanat, A. Musoglu, A. Aydin; Gastritis cystica polyposa found in an unoperated stomach: an unusual case treated by endoscopic polypectomy; Endoscopy, 35 (2003), p. 882
- [10] C.H. Park, J.M. Park, C.K. Jung, D.B. Kim, S.H. Kang, S.W. Lee, et al.; Early gastric cancer associated with gastritis cystica polyposa in the unoperated stomach treated by endoscopic submucosal dissection; Gastrointest Endosc, 69 (2009), pp. e47–e50
- [11] J.H. Kim, S.Y. Jang, J.A. Hwang, S.H. Ha, W.G. Choi, J.S. Park, et al.; A ten-year follow-up of a case with gastric adenoma accompanied with gastritis cystica profunda treated by endoscopic submucosal dissection [Article in Korean]; Korean J Gastroenterol, 59 (2012), pp. 366–371
Document information
Published on 15/05/17
Submitted on 15/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?