Abstract
In vivo imaging is a novel experimental approach for biological research. Multiphoton microscopy (MPM), a type of fluorescence microscopy, is a new tool for in vivo imaging analysis. MPM allows observation of both tissue structures and cell behaviors or cell–cell interactions in living animals in real time. Skin is an ideal tissue for MPM analysis as it is directly accessible to the microscope. In the skin, immune cells cooperate to maintain skin homeostasis or to exert immune responses against foreign antigens. In vivo imaging by MPM analysis provides precise information on cell dynamics in the skin, and has significantly expanded our knowledge of the cutaneous immune system. In this review, we will discuss recent insights related to the mechanisms of allergic skin inflammation that have been revealed by MPM analysis.
Keywords
Contact hypersensitivity; Dendritic cells; In vivo imaging; Multiphoton microscopy; T cells
List of abbreviations
MPM, Multiphoton microscopy; DC, Dendritic cell; MC, Mast cell; CHS, Contact hypersensitivity; LC, Langerhans cell; dDC, Dermal dendritic cell; dLN, Draining lymph node; YFP, Yellow fluorescence protein; LTB4, Leukotriene B4; CXCL2, Chemokine (C-X-C motif) ligand 2; iSALT, Inducible skin-associated lymphoid tissues; PD-1, Programmed death-1; CTLA-4, Cytotoxic T-lymphocyte-associated protein 4; Treg, Regulatory T cell
Introduction
The skin contains various kinds of immune cells, such as dendritic cells (DCs), T cells, and mast cells (MCs). Their dynamic interactions are essential for the maintenance of homeostasis and also for the induction/regulation of cutaneous immune responses.1 and 2 For example, DCs continuously migrate and scan antigens in the skin. When DCs capture antigens, they present them to effector T cells, which then activate and produce various cytokines to eliminate the antigens. Here, to exert their effector functions to an appropriate extent and with correct timing, their motility and the duration of interaction must be tightly regulated spatiotemporally. If not, these immune responses may cause unnecessary tissue damages as a result of excessive T cell activations, or they may instead fail to protect the host from the antigens due to inadequate T cell activations.
Until recently, cell dynamics analysis has mainly been performed by in vitro systems. For example, chemotaxis chamber assay is frequently performed to investigate cell migration ability in vitro. However, it remains unknown whether such cell dynamics in a culture system actually reflect in vivo cell behaviors, since tissue conditions, such as collagen fiber structures, blood supply, oxygen concentration and cell–cell interactions, significantly affect cell dynamics in vivo. Immunohistochemical analysis is the conventional method used to analyze cell localization and behaviors in tissues. Although this analysis can extract in vivo information, it provides a static picture of a certain specific moment in the continuous flow of biological phenomena, and it provides limited information related to cell dynamics. For the accurate evaluation of cell movement and cell–cell interaction in vivo, direct observation of the cells in a living animal is necessary.
Multiphoton microscopy (MPM) is a type of fluorescence microscopy that excites fluorophores with long-wavelength photons. Since the excitation of fluorophores with multiphotons occurs only on focal planes, the resolution of MPM is very high. In addition, long-wavelength photons penetrates deep into tissues with low phototoxicity, which enables long time-lapse observation with minimal tissue damages. Moreover, some tissue structures, such as collagen fibers and elastins, are visualized without fluorescent labeling by multiphoton excitation. Based on these characteristics, MPM is a useful tool for in vivo imaging with subcellular resolutions. Using this technique, we have investigated immune cell dynamics in various skin conditions. 3, 4, 5, 6 and 7 In this review, we will introduce how MPM analysis has expanded our knowledge of the cutaneous immune systems in allergic inflammation, with a focus on contact dermatitis.
Contact dermatitis
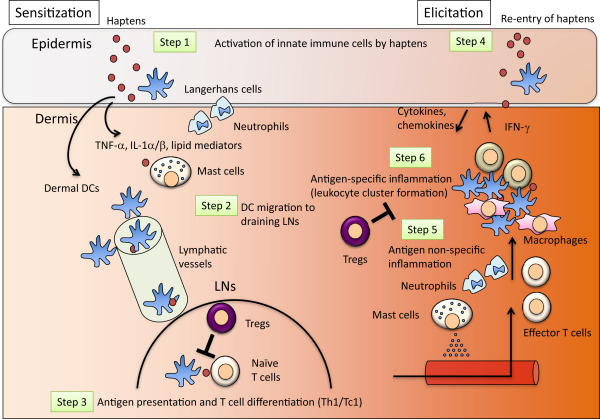
Contact dermatitis, such as metal allergy and plant allergy, is a kind of delayed-type hypersensitivity in skin, and is a common allergic skin disease affecting approximately 15–20% of the worlds populations.8 Contact hypersensitivity (CHS) is a frequently used mouse model of contact dermatitis. Chemicals that induce contact dermatitis and CHS are small compounds called haptens.9 and 10 Haptens binds to self-proteins, which become immunogenic neo-antigens.10 Haptens or haptenized self-proteins activate innate immune cells, such as keratinocytes, MCs, and macrophages, which produce various chemical mediators, and induce two important phases in CHS: the sensitization phase and the elicitation phase (Fig. 1). In the sensitization phase, skin DCs capture antigens (hapten-self complex), and migrate to draining lymph nodes (dLNs) to induce differentiation and proliferation of antigen specific T cells (mainly Th1 and Tc1 cells). When the same hapten enters the skin, the antigen-specific T cells are activated in the skin by antigen-captured cutaneous DCs. The activated T cells produce various cytokines/chemokines and induce skin inflammation.11 This inflammatory phase is called the elicitation phase. Using the CHS model, we have analyzed how DC and T cell dynamics are regulated in the skin during allergic inflammation.12, 13 and 14
|
|
|
Fig. 1. Overview of the immunological mechanisms of CHS. Step 1. Haptens activate innate immune cells (e.g. keratinocytes, mast cells) and induce the production of various chemical mediators. Step 2. Antigen-captured activated DCs migrate to the dLNs. Step 3. Migrated DCs present the antigen to naïve T cells, which in general differentiate them to Th1 and Tc1 cells. Step 4. Haptens generally induce subtle inflammation by activating innate immune cells, and recruit neutrophils. Haptens also activate macrophages, which lead to leukocyte cluster formation, which is called iSALT. Step 5. Leukocytes including antigen-specific effector T cells are recruited to form iSALT. Step 6. The antigen-specific effector T cells are activated in the skin by antigen-captured dermal DCs, which induce antigen-specific inflammation. Activation of effector T cells mainly occurs in leukocyte clusters, iSALT. Tregs play inhibitory roles in both the sensitization and elicitation phases. |
DC dynamics after hapten application
At least three DC subsets have been identified in the skin: Langerhans cells (LCs; located in the epidermal layer) and two dermal DC (dDC) populations. These two dDC subsets express different patterns of surface molecules and are classified as langerin-positive (or CD103-positive) DCs and langerin-negative (or CD11b-positive) dDCs.2 In the sensitization phase, dDCs, especially langerin-positive dDCs, are considered to be the essential cell populations that mediate the sensitization,15, 16 and 17 although other dDC subsets have the ability to exert the functions.18, 19, 20, 21 and 22
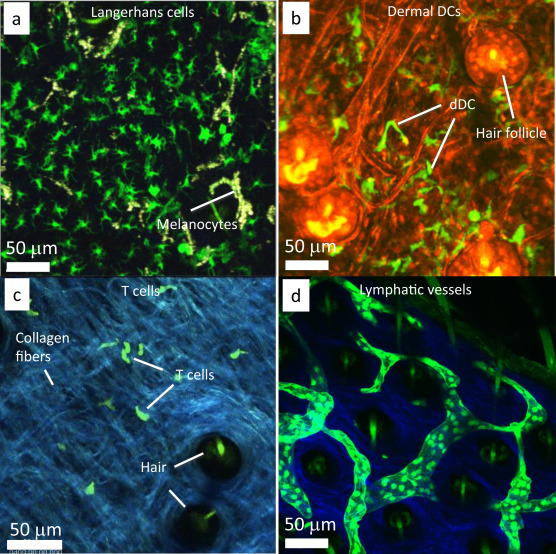
CD11c-YFP mice, which express yellow fluorescence protein (YFP) under the transcriptional control of mouse integrin alpha X (CD11c), are frequently used for the visualization of DCs by MPM.23 LCs and dDCs are clearly visualized in CD11c-YFP mice (Fig. 2). In the steady state skin, dDCs exhibit active motility with polarized morphology.4, 24 and 25 After hapten application, dDCs exhibit a transient increase in motility, and the average velocities increase almost twofold.4 and 25 Antigen-bearing DCs migrate from the skin to the dLNs, but some dDCs form cluster structures in the skin around 18–24 h after hapten application.5 This leukocyte cluster is essential for effector T cell activation in the skin, as discussed in the next section. The motility of dDCs in the skin appears to be regulated by G protein-coupled receptor-mediated signaling, because the administration of pertussis toxin significantly impairs dDC motility and probing behavior.25
|
|
|
Fig. 2. Representative MPM images of DCs and T cells in the skin. (a) LCs are visualized by green fluorescence in langerin-eGFP mice. Melanocytes are also visualized by auto-fluorescence by melanin. (b) dDCs are visualized by green fluorescence in CD11c-YFP mice crossed with DsRed mice. Skin structures, such as blood vessels and hair follicles, are visualized by red fluorescence (DsRed). (c) Effector OT-II GFP T cells are visualized by green fluorescence. Collagen fibers are visualized by the signals from second harmonic generation (blue). (d) Lymphatic vessels are visualized by green fluorescence in Prox1-GFP mice. |
Among the G-protein-coupled receptors, BLT1 receptor is one of the candidates considered to regulate DC motility in the skin.4 BLT1 is a receptor of leukotriene B4 (LTB4), a lipid mediator derived from arachidonic acid.26 and 27 Blockade of BLT1 signaling significantly down-regulates DC motility in both steady state skin and hapten-treated inflammatory skin. Consequently, DC migration from the skin to the dLNs and DC cluster formation in the skin are attenuated by BLT1 signaling blockade, which leads to suppressed CHS responses.4 Stimulation of BLT1 signaling causes actin filament reorganization in DCs through activation of Cdc42 and Rac1, and increases DC motility.4 Intriguingly, BLT1 signaling increases DC motility synergistically with chemokine (C–C motif) ligand 21 and chemokine (C-X-C motif) ligand 2 (CXCL2), both of which play essential roles in cutaneous DC migration.5 and 28 Thus, LTB4 may be an important accelerator of chemokine-induced cutaneous DC migration.
In contrast to dDCs, LCs are basically immotile in the steady state skin, although they constantly extend and retract dendrites, which is called “dendrite surveillance extension and retraction cycling habitude”.29 LCs have long been considered as the essential antigen-presenting cells in CHS. However, recent studies using LCs-deficient mice have suggested the regulatory roles of LCs in the sensitization phase of CHS via the production of IL-10 or the induction of regulatory T cells.30, 31 and 32 Hapten application increases LC motility in the epidermis slightly, but the average velocity is much lower than that of dDCs.25 and 33 Consistently, the kinetics of hapten-induced migration from the skin to the dLNs is much slower in LCs than in dDCs.34 The slow motility of LCs may generate the regulatory characteristics of LCs.
DC cluster formation
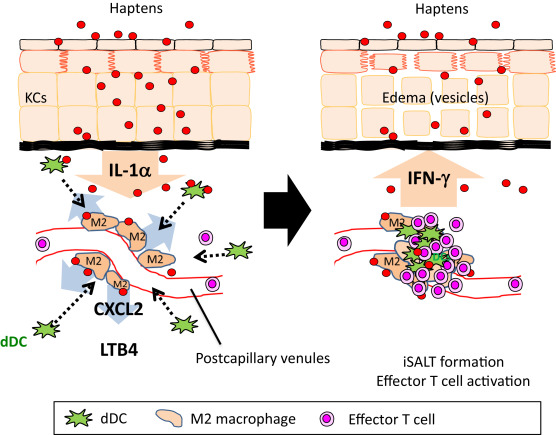
As mentioned above, dDCs form clusters in the dermis after hapten application. What is the formation mechanism and biological significance of DC clusters? MPM analysis provides important clues to answer these questions.5 Time-lapse imaging by MPM revealed that DCs migrate toward perivascular macrophages on post-capillary venules. IL-1α, CXCL2, and LTB4 are important molecules that create the clusters, and blockade of these molecules thus significantly impairs cluster formation.4 and 5 Keratinocytes are supposed to be the major source of IL-1α. IL-1α activates perivascular macrophages, which attract dDCs through production of CXCL2. Infiltrated effector T cells then accumulate around the clusters and are activated by antigen-bearing DCs in the cluster (Fig. 3). The inhibition of the cluster formation significantly attenuates effector T cell activation. These results indicate that DC clusters serve as essential structures for efficient T cell activation in the skin. We propose that these structures should be termed “inducible skin-associated lymphoid tissues (iSALT)” since they do not exist in the steady state but are induced in inflammatory conditions.5 and 35
|
|
|
Fig. 3. Schematic illustration of DC cluster formation mechanisms. (Left) Haptens induce IL-1α production by KCs, which stimulates M2-type macrophages located around postcapillary venules. The stimulated macrophages then produce CXCL2, which attract dDCs. LTB4 also plays an important role in DC accumulation by increasing DC motility. (Right) Effector T cells are activated within the DC clusters (called iSALT) and produce cytokines, such as IFN-γ, to induce skin inflammation. |
T cell dynamics and its regulation during the elicitation phase
Effector T cells infiltrate inflamed tissues with limited dependency on their antigen specificity.3 and 36 Without their cognate antigens, effector T cells exhibit active motility in the dermis.3 and 37 When effector T cells recognize their cognate antigens presented by DCs, T cells initiate stable contact with DCs, and reduce their motility.3 and 37 After the stable interaction with DCs, T cells produce various cytokines and provoke inflammation.9 Here, to avoid excessive tissue damages, the magnitude and quality of T cell activity must be tightly regulated. The precise T cell dynamics and the regulatory mechanisms involved in these processes remain unclear, however, due to the lack of appropriate systems to control the timing of initial antigen recognition by T cells in the skin.
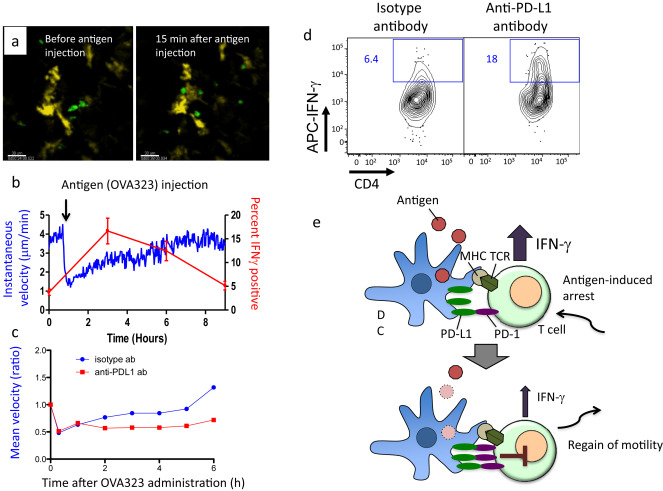
Using a delayed-type hypersensitivity model in combination with MPM analysis, we established a method that enables the synchronization of effector T cell activation, and analyzed the regulatory mechanisms that control effector T cell dynamics and effector function.3 After antigen recognition, effector T cells rapidly cease motility and initiate stable interaction with DCs within 10 min. They gradually regain motility 1–2 h after antigen recognition, and regain their original speed by 6–8 h. Intriguingly, the cytokine production ability reaches a peak within 3 h after the initial antigen contact, and then T cells cease making cytokine as they regain motility, indicating an inverse correlation between effector function and motility. Programmed death-1 (PD-1) signaling negatively regulates the antigen sensitivity of effector T cells and controls the duration of migration arrest and cytokine production (Fig. 4).3 These negative feedback mechanisms are responsible for the adequate T cell activation in inflammatory skin.38
|
|
|
Fig. 4. The relationship between T cell dynamics and the effector functions in the skin. (a) Antigen-induced T cell arrest by interacting with dDCs. Without cognate antigens, effector OT-II GFP T cells (green) exhibit active motility with elongated morphology. Within 15 min after peptibe antigen (OVA323) injection, T cells exhibit stable interaction with dDCs (yellow) with round shape morphology. dDCs are visualized using CD11c-YFP mice. (b) Time course of motility changes and the effector function in OT-II T cells before and after antigen stimulation. The mean instantaneous velocity of OT-II T cells is shown in blue line. The percentage of IFN-γ producing OT-II T cells is shown in red line. (c) The effect of anti-PD-1 ligand 1 (PD-L1) blocking antibody on the recovery of T cell motility after antigen-induced migration arrest. Anti-PD-L1 antibody treatment delays the recovery. (d) Flow cytometry analysis of intracellular IFN-γ staining of OT-II T cells at 8 h after antigen administration with (right panel) or without (left panel) anti-PD-L1 antibody. (e) Schematic illustration of T cell dynamics and the activation state in inflammatory skin. (Upper panel) Upon antigen recognition, effector T cells rapidly cease motility and produce cytokines through TCR activation. (Lower panel) T cells gradually upregulate PD-1, which limits sustained activation of T cells. Desensitized T cells regain motility and shuts down cytokine production. |
Similar PD-1-dependent regulatory mechanisms have been reported in other peripheral tissues,39 and 40 although different mechanisms may work depending on the tissue or experimental model.41 Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), another important negative regulator in the immune system, is also involved in the control of antigen-induced T cell arrest and T cell activation.42 and 43 Immune checkpoint molecules likely regulate T cell activation in peripheral tissues in a spatiotemporal manner.
Dynamics of regulatory T cells in the skin
Regulatory T cell (Treg) is a subset of CD4+ T cells with strong immunosuppressive activity.44 During the elicitation phase in CHS, the number of skin Tregs significantly increases, and the skin homing Tregs posses stronger immunosuppressive activity than LNs-resident Tregs.45 and 46 In addition, mice lacking skin Tregs spontaneously exhibit severe dermatitis,47 and loss of Tregs causes severe dermatitis in humans.48 These observations indicate that skin Tregs are essential for both the maintenance of skin homeostasis and the termination of inflammatory responses.49
Chow et al. analyzed the dynamics of Tregs in skin by MPM, and reported different patterns of migration ability between Tregs and effector CD4+ T cells.50 In contrast to the uniformly high motility of effector CD4+ T cells, most Tregs exhibited significantly lower motility in the steady state skin. In the inflammatory skin in the elicitation phase, although a subset of Tregs increased their motility, most Tregs remained immotile. Although the biological significance of such different migration patterns remains unclear, Tregs with low motility may be constantly interacting with DCs and interfere with the stable DC-effector T cell interaction in the skin, as observed in another model.51 In addition, Tregs with high motility may be in the process of migration from the skin to the dLNs, since skin Tregs constitutively migrate to the dLNs, and the number and proportion of the migratory Tregs increase in the elicitation phase.45 Elucidation of the factors that determine Treg motility may reveal the novel mechanisms by which Tregs exert their suppressive function in the skin.
The role of MCs and the dynamics in CHS
MCs are generally considered to be innate immune cells, but there is now growing evidence that they play important functions in both innate and adaptive immune responses. In CHS, the role of MCs is controversial since conflicting results have been reported by different groups. However, recent data using a novel MC-specific depletion system indicate that MCs exert promoting roles and are essential for the development of CHS.52, 53 and 54 Furthermore, MPM analysis using MC reporter mice revealed the unique functions of MCs in CHS.55 and 56 MCs are located around blood vessels in the dermis, and are sessile in both the steady and inflammatory states. Their elongated or spindle-like morphology in steady state skin changes to a globular shape after an antigen challenge, however.56 An antigen challenge induces an increase in vascular permeability, which is dependent on MC-derived histamines. This increased permeability is essential for the initial neutrophil infiltration or effector T cell infiltration in the elicitation phase.57 In the sensitization phase, MC-derived TNF-α induces cutaneous DC migration and maturation, and promotes effector CD8+ T cell priming in dLNs.52, 54 and 56
Concluding remarks
The development and progress of intravital imaging techniques have significantly extended our knowledge of cell dynamics in vivo, including the morphological change, migration speed, and interaction time with other cells. Moreover, we have encountered unexpected phenomena that have not previously been observed by the conventional immunohistochemical method, and this has led to the discovery of novel regulatory mechanisms and concepts related to cutaneous immune system. MPM analysis is not just a tool for the statistical analysis of previously reported biological phenomenon, but is expected to be a great tool in the discovery of new biology.
Currently, the application of MPM is mainly restricted in animal studies, and the cell dynamics of human tissues remains unknown. There already exists an MPM machine that can be used on humans, and the morphological analysis of epidermal cells in some skin diseases has been performed.58, 59 and 60 The development of novel cell-labeling systems and further improvement in MPM will enable the analysis of cell dynamics in humans in the future.
Conflict of interest
The authors have no conflict of interest to declare.
References
- 1 M. Pasparakis, I. Haase, F.O. Nestle; Mechanisms regulating skin immunity and inflammation; Nat Rev Immunol, 14 (2014), pp. 289–301
- 2 W.R. Heath, F.R. Carbone; The skin-resident and migratory immune system in steady state and memory: innate lymphocytes, dendritic cells and T cells; Nat Immunol, 14 (2013), pp. 978–985
- 3 T. Honda, J.G. Egen, T. Lammermann, W. Kastenmuller, P. Torabi-Parizi, R.N. Germain; Tuning of antigen sensitivity by T cell receptor-dependent negative feedback controls T cell effector function in inflamed tissues; Immunity, 40 (2014), pp. 235–247
- 4 Y. Sawada, T. Honda, S. Hanakawa, S. Nakamizo, T. Murata, Y. Ueharaguchi-Tanada, et al.; Resolvin E1 inhibits dendritic cell migration in the skin and attenuates contact hypersensitivity responses; J Exp Med, 212 (2015), pp. 1921–1930
- 5 Y. Natsuaki, G. Egawa, S. Nakamizo, S. Ono, S. Hanakawa, T. Okada, et al.; Perivascular leukocyte clusters are essential for efficient activation of effector T cells in the skin; Nat Immunol, 15 (2014), pp. 1064–1069
- 6 K. Kabashima, G. Egawa; Intravital multiphoton imaging of cutaneous immune responses; J Invest Dermatol, 134 (2014), pp. 2680–2684
- 7 G. Egawa, S. Nakamizo, Y. Natsuaki, H. Doi, Y. Miyachi, K. Kabashima; Intravital analysis of vascular permeability in mice using two-photon microscopy; Sci Rep, 3 (2013), p. 1932
- 8 M. Peiser, T. Tralau, J. Heidler, A.M. Api, J.H. Arts, D.A. Basketter, et al.; Allergic contact dermatitis: epidemiology, molecular mechanisms, in vitro methods and regulatory aspects. Current knowledge assembled at an international workshop at BfR, Germany; Cell Mol Life Sci, 69 (2012), pp. 763–781
- 9 T. Honda, G. Egawa, S. Grabbe, K. Kabashima; Update of immune events in the murine contact hypersensitivity model: toward the understanding of allergic contact dermatitis; J Invest Dermatol, 133 (2013), pp. 303–315
- 10 D.H. Kaplan, B.Z. Igyarto, A.A. Gaspari; Early immune events in the induction of allergic contact dermatitis; Nat Rev Immunol, 12 (2012), pp. 114–124
- 11 T. Honda, K. Kabashima; Novel concept of iSALT (inducible skin-associated lymphoid tissue) in the elicitation of allergic contact dermatitis; Proc Jpn Acad Ser B Phys Biol Sci, 92 (2016), pp. 20–28
- 12 K. Kabashima, T. Murata, H. Tanaka, T. Matsuoka, D. Sakata, N. Yoshida, et al.; Thromboxane A2 modulates interaction of dendritic cells and T cells and regulates acquired immunity; Nat Immunol, 4 (2003), pp. 694–701
- 13 K. Kabashima, D. Sakata, M. Nagamachi, Y. Miyachi, K. Inaba, S. Narumiya; Prostaglandin E2-EP4 signaling initiates skin immune responses by promoting migration and maturation of Langerhans cells; Nat Med, 9 (2003), pp. 744–749
- 14 T. Honda, T. Matsuoka, M. Ueta, K. Kabashima, Y. Miyachi, S. Narumiya; Prostaglandin E(2)-EP(3) signaling suppresses skin inflammation in murine contact hypersensitivity; J Allergy Clin Immunol, 124 (2009), pp. 809–818 e802
- 15 L.F. Poulin, S. Henri, B. de Bovis, E. Devilard, A. Kissenpfennig, B. Malissen; The dermis contains langerin+ dendritic cells that develop and function independently of epidermal Langerhans cells; J Exp Med, 204 (2007), pp. 3119–3131
- 16 F. Ginhoux, M.P. Collin, M. Bogunovic, M. Abel, M. Leboeuf, J. Helft, et al.; Blood-derived dermal langerin+ dendritic cells survey the skin in the steady state; J Exp Med, 204 (2007), pp. 3133–3146
- 17 L.S. Bursch, L. Wang, B. Igyarto, A. Kissenpfennig, B. Malissen, D.H. Kaplan, et al.; Identification of a novel population of Langerin+ dendritic cells; J Exp Med, 204 (2007), pp. 3147–3156
- 18 T. Honda, S. Nakajima, G. Egawa, K. Ogasawara, B. Malissen, Y. Miyachi, et al.; Compensatory role of Langerhans cells and langerin-positive dermal dendritic cells in the sensitization phase of murine contact hypersensitivity; J Allergy Clin Immunol, 125 (2010), pp. 1154–1156 e1152
- 19 Y. Kumamoto, K. Denda-Nagai, S. Aida, N. Higashi, T. Irimura; MGL2 dermal dendritic cells are sufficient to initiate contact hypersensitivity in vivo; PLoS One, 4 (2009), p. e5619
- 20 M. Noordegraaf, V. Flacher, P. Stoitzner, B.E. Clausen; Functional redundancy of Langerhans cells and Langerin+ dermal dendritic cells in contact hypersensitivity; J Invest Dermatol, 130 (2010), pp. 2752–2759
- 21 B.T. Edelson, W. Kc, R. Juang, M. Kohyama, L.A. Benoit, P.A. Klekotka, et al.; Peripheral CD103+ dendritic cells form a unified subset developmentally related to CD8alpha+ conventional dendritic cells; J Exp Med, 207 (2010), pp. 823–836
- 22 L. Wang, L.S. Bursch, A. Kissenpfennig, B. Malissen, S.C. Jameson, K.A. Hogquist; Langerin expressing cells promote skin immune responses under defined conditions; J Immunol, 180 (2008), pp. 4722–4727
- 23 R.L. Lindquist, G. Shakhar, D. Dudziak, H. Wardemann, T. Eisenreich, M.L. Dustin, et al.; Visualizing dendritic cell networks in vivo; Nat Immunol, 5 (2004), pp. 1243–1250
- 24 L.G. Ng, A. Hsu, M.A. Mandell, B. Roediger, C. Hoeller, P. Mrass, et al.; Migratory dermal dendritic cells act as rapid sensors of protozoan parasites; PLoS Pathog, 4 (2008), p. e1000222
- 25 D. Sen, L. Forrest, T.B. Kepler, I. Parker, M.D. Cahalan; Selective and site-specific mobilization of dermal dendritic cells and Langerhans cells by Th1- and Th2-polarizing adjuvants; Proc Natl Acad Sci U S A, 107 (2010), pp. 8334–8339
- 26 T. Honda, K. Kabashima; Leukotrienes as key mediators and amplifiers in allergic inflammation: insights from the bench and clinic; Exp Dermatol, 23 (2014), pp. 95–96
- 27 T. Yokomizo; Leukotriene B4 receptors: novel roles in immunological regulations; Adv Enzyme Regul, 51 (2011), pp. 59–64
- 28 R. Forster, A. Schubel, D. Breitfeld, E. Kremmer, I. Renner-Muller, E. Wolf, et al.; CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs; Cell, 99 (1999), pp. 23–33
- 29 A. Nishibu, B.R. Ward, J.V. Jester, H.L. Ploegh, M. Boes, A. Takashima; Behavioral responses of epidermal Langerhans cells in situ to local pathological stimuli; J Invest Dermatol, 126 (2006), pp. 787–796
- 30 D.H. Kaplan, M.C. Jenison, S. Saeland, W.D. Shlomchik, M.J. Shlomchik; Epidermal langerhans cell-deficient mice develop enhanced contact hypersensitivity; Immunity, 23 (2005), pp. 611–620
- 31 B.Z. Igyarto, M.C. Jenison, J.C. Dudda, A. Roers, W. Muller, P.A. Koni, et al.; Langerhans cells suppress contact hypersensitivity responses via cognate CD4 interaction and langerhans cell-derived IL-10; J Immunol, 183 (2009), pp. 5085–5093
- 32 M. Gomez de Aguero, M. Vocanson, F. Hacini-Rachinel, M. Taillardet, T. Sparwasser, A. Kissenpfennig, et al.; Langerhans cells protect from allergic contact dermatitis in mice by tolerizing CD8(+) T cells and activating Foxp3(+) regulatory T cells; J Clin Invest, 122 (2012), pp. 1700–1711
- 33 M.R. Gaiser, T. Lammermann, X. Feng, B.Z. Igyarto, D.H. Kaplan, L. Tessarollo, et al.; Cancer-associated epithelial cell adhesion molecule (EpCAM; CD326) enables epidermal Langerhans cell motility and migration in vivo; Proc Natl Acad Sci U S A, 109 (2012), pp. E889–E897
- 34 A. Kissenpfennig, S. Henri, B. Dubois, C. Laplace-Builhe, P. Perrin, N. Romani, et al.; Dynamics and function of Langerhans cells in vivo: dermal dendritic cells colonize lymph node areas distinct from slower migrating Langerhans cells; Immunity, 22 (2005), pp. 643–654
- 35 S. Ono, K. Kabashima; Proposal of inducible skin-associated lymphoid tissue (iSALT); Exp Dermatol, 24 (2015), pp. 630–631
- 36 J.G. Egen, A.G. Rothfuchs, C.G. Feng, M.A. Horwitz, A. Sher, R.N. Germain; Intravital imaging reveals limited antigen presentation and T cell effector function in mycobacterial granulomas; Immunity, 34 (2011), pp. 807–819
- 37 G. Egawa, T. Honda, H. Tanizaki, H. Doi, Y. Miyachi, K. Kabashima; In vivo imaging of T-cell motility in the elicitation phase of contact hypersensitivity using two-photon microscopy; J Invest Dermatol, 131 (2011), pp. 977–979
- 38 E. Kugelberg; T cell responses: kiss and run; Nat Rev Immunol, 14 (2014), p. 134
- 39 D. Michonneau, P. Sagoo, B. Breart, Z. Garcia, S. Celli, P. Bousso; The PD-1 axis enforces an anatomical segregation of CTL activity that creates tumor niches after allogeneic hematopoietic stem cell transplantation; Immunity, 44 (2016), pp. 143–154
- 40 B.T. Fife, K.E. Pauken, T.N. Eagar, T. Obu, J. Wu, Q. Tang, et al.; Interactions between PD-1 and PD-L1 promote tolerance by blocking the TCR-induced stop signal; Nat Immunol, 10 (2009), pp. 1185–1192
- 41 B.H. Zinselmeyer, S. Heydari, C. Sacristan, D. Nayak, M. Cammer, J. Herz, et al.; PD-1 promotes immune exhaustion by inducing antiviral T cell motility paralysis; J Exp Med, 210 (2013), pp. 757–774
- 42 H. Schneider, J. Downey, A. Smith, B.H. Zinselmeyer, C. Rush, J.M. Brewer, et al.; Reversal of the TCR stop signal by CTLA-4; Science, 313 (2006), pp. 1972–1975
- 43 M.G. Ruocco, K.A. Pilones, N. Kawashima, M. Cammer, J. Huang, J.S. Babb, et al.; Suppressing T cell motility induced by anti-CTLA-4 monotherapy improves antitumor effects; J Clin Invest, 122 (2012), pp. 3718–3730
- 44 S. Sakaguchi, T. Yamaguchi, T. Nomura, M. Ono; Regulatory T cells and immune tolerance; Cell, 133 (2008), pp. 775–787
- 45 M. Tomura, T. Honda, H. Tanizaki, A. Otsuka, G. Egawa, Y. Tokura, et al.; Activated regulatory T cells are the major T cell type emigrating from the skin during a cutaneous immune response in mice; J Clin Invest, 120 (2010), pp. 883–893
- 46 T. Honda, A. Otsuka, H. Tanizaki, Y. Minegaki, K. Nagao, H. Waldmann, et al.; Enhanced murine contact hypersensitivity by depletion of endogenous regulatory T cells in the sensitization phase; J Dermatol Sci, 61 (2011), pp. 144–147
- 47 J.C. Dudda, N. Perdue, E. Bachtanian, D.J. Campbell; Foxp3+ regulatory T cells maintain immune homeostasis in the skin; J Exp Med, 205 (2008), pp. 1559–1565
- 48 H.D. Ochs, S.F. Ziegler, T.R. Torgerson; FOXP3 acts as a rheostat of the immune response; Immunol Rev, 203 (2005), pp. 156–164
- 49 T. Honda, Y. Miyachi, K. Kabashima; The role of regulatory T cells in contact hypersensitivity; Recent Pat Inflamm Allergy Drug Discov, 4 (2010), pp. 85–89
- 50 Z. Chow, S.N. Mueller, J.A. Deane, M.J. Hickey; Dermal regulatory T cells display distinct migratory behavior that is modulated during adaptive and innate inflammation; J Immunol, 191 (2013), pp. 3049–3056
- 51 Y. Onishi, Z. Fehervari, T. Yamaguchi, S. Sakaguchi; Foxp3+ natural regulatory T cells preferentially form aggregates on dendritic cells in vitro and actively inhibit their maturation; Proc Natl Acad Sci U S A, 105 (2008), pp. 10113–10118
- 52 A. Otsuka, M. Kubo, T. Honda, G. Egawa, S. Nakajima, H. Tanizaki, et al.; Requirement of interaction between mast cells and skin dendritic cells to establish contact hypersensitivity; PLoS One, 6 (2011), p. e25538
- 53 A. Dudeck, C.A. Suender, S.L. Kostka, E. von Stebut, M. Maurer; Mast cells promote Th1 and Th17 responses by modulating dendritic cell maturation and function; Eur J Immunol, 41 (2011), pp. 1883–1893
- 54 J. Dudeck, S.M. Ghouse, C.H. Lehmann, A. Hoppe, N. Schubert, S.A. Nedospasov, et al.; Mast-cell-derived TNF amplifies CD8(+) dendritic cell functionality and CD8(+) T cell priming; Cell Rep, 13 (2015), pp. 399–411
- 55 A. Kohler, V. Schmithorst, M.D. Filippi, M.A. Ryan, D. Daria, M. Gunzer, et al.; Altered cellular dynamics and endosteal location of aged early hematopoietic progenitor cells revealed by time-lapse intravital imaging in long bones; Blood, 114 (2009), pp. 290–298
- 56 A. Dudeck, J. Dudeck, J. Scholten, A. Petzold, S. Surianarayanan, A. Kohler, et al.; Mast cells are key promoters of contact allergy that mediate the adjuvant effects of haptens; Immunity, 34 (2011), pp. 973–984
- 57 T. Biedermann, M. Kneilling, R. Mailhammer, K. Maier, C.A. Sander, G. Kollias, et al.; Mast cells control neutrophil recruitment during T cell-mediated delayed-type hypersensitivity reactions through tumor necrosis factor and macrophage inflammatory protein 2; J Exp Med, 192 (2000), pp. 1441–1452
- 58 M.J. Koehler, S. Zimmermann, S. Springer, P. Elsner, K. Konig, M. Kaatz; Keratinocyte morphology of human skin evaluated by in vivo multiphoton laser tomography; Skin Res Technol, 17 (2011), pp. 479–486
- 59 M.J. Koehler, M. Speicher, S. Lange-Asschenfeldt, E. Stockfleth, S. Metz, P. Elsner, et al.; Clinical application of multiphoton tomography in combination with confocal laser scanning microscopy for in vivo evaluation of skin diseases; Exp Dermatol, 20 (2011), pp. 589–594
- 60 M. Klemp, M.C. Meinke, M. Weinigel, J. Rowert-Huber, K. Konig, M. Ulrich, et al.; Comparison of morphologic criteria for actinic keratosis and squamous cell carcinoma using in vivo multiphoton tomography; Exp Dermatol (2015) http://dx.doi.org/10.1111/exd.12912
Document information
Published on 05/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?