Summary
Background
We previously reported our initial experience with laparoendoscopic single-site (LESS) retroperitoneal partial adrenalectomy using a custom-made single-port device and conventional straight laparoscopic instruments.
Methods
Between December 2010 and February 2012, LESS retroperitoneal partial adrenalectomies were performed in 11 patients. Six patients had aldosterone-producing adenomas (APAs) and five patients had nonfunctioning tumors. A single-port access was created with an Alexis wound retractor (Applied Medical, Rancho Santa Margarita, CA, USA) through an incision of 2–3 cm beneath the tip of the 12th rib. All procedures were performed with straight laparoscopic instruments.
Results
All LESS procedures were successfully completed without conversion to traditional laparoscopic conversion. The tumors ranged from 1 cm to 4.7 cm (mean, 2.3 cm). The operative time was 71–257 minutes (mean, 121 minutes). Most patients (n = 8) had minimal blood loss; the other three patients had a blood loss of 150 mL, 100 mL, and 100 mL. The mean hospital stay was 3 days (range, 1–6 days). There were no perioperative or postoperative complications. Pathological examinations revealed negative surgical margins in all specimens. All patients with Conns syndrome had an improvement in blood pressure and normalization of plasma renin activity and serum aldosterone levels; all patients were free of potassium supplementation.
Conclusion
Our results clearly demonstrate that LESS retroperitoneal partial adrenalectomy can be performed safely and effectively using a custom-made single-access platform and standard laparoscopic instruments.
Keywords
laparoendoscopic single-site;laparoscopy;partial adrenalectomy
1. Introduction
Two decades ago, laparoscopic access to the adrenal gland was described.1 Since then, it has replaced open surgery and become the standard procedure for adrenal surgery because of its high success rate, minimal morbidity, and rapid convalescence. In the past few years, advances in surgical instruments and/or techniques have increased the enthusiasm and interest in more minimally invasive surgery.2 Among these techniques, laparoendoscopic single-site surgery (LESS)—characterized by performing laparoscopic procedures through a single skin incision—has gradually gained popularity in recent years. Compared to conventional laparoscopic surgery, LESS has the advantage of a better cosmetic outcome3 and early convalescence.4 To date, LESS has been successfully applied in various operations,5 ; 6 and it was recently demonstrated as a feasible technique in total adrenalectomy.7; 8 ; 9
Partial adrenalectomy (i.e., adrenal-sparing surgery) was initially proposed with the intention to preserve the maximal postoperative adrenocortical function and to avoid steroid dependence in select patients (e.g., patients with a hereditary form of pheochromocytoma or a solitary adrenal gland).10 ; 11 Patients who undergo unilateral total adrenalectomy may not respond normally to stressful situations, even in patients with a normal contralateral adrenal gland.12 For this reason, several surgical groups also perform partial adrenalectomy in patients with aldosterone-producing adenomas (APAs) or small adrenal tumors.12; 13; 14; 15; 16; 17; 18; 19; 20 ; 21 A recent literature review covering the past 2 decades reveals an increasing trend worldwide toward partial adrenalectomy. Partial adrenalectomy can achieve a similar surgical outcome and perioperative complications as total adrenalectomy in various pathological conditions.13 These data encourage surgeons to develop and refine techniques for partial adrenalectomy, which is potentially more challenging than total adrenalectomy.
In the past, partial adrenalectomy has been successfully performed in several surgical platforms, including conventional laparoscopy,12 needlescopy,14 and robotic-assisted laparoscopy.22 ; 23 Yuge et al15 more recently reported the first case of LESS transperitoneal partial adrenalectomy. By using a custom-made single-access platform and standard laparoscopic instruments, we also completed 11 LESS retroperitoneal partial adrenalectomy procedures. In this paper, we report our series to evaluate the feasibility of this procedure.
2. Materials and methods
Between December 2010 and February 2012, 11 adult patients with benign adrenal tumors underwent LESS retroperitoneal partial adrenalectomy at our institute. All operations were performed by a single surgeon (YCT). The diagnoses of the 11 patients were Conns syndrome (n = 6) and nonfunctioning adrenal tumors (n = 5). Table 1 lists the patients' data.
| Patient | Age (y) | Sex | Site | Tumor size (cm) | Diagnosis | Operation time (min) | Blood loss (mL) | Hospital stay (d) | Clinical outcome | Postoperative laboratory test results | Follow-up period (mo) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 52 | Female | R, medial limb | 1.4 | Conns syndrome | 105 | 0 | 2 | Resolution of HTN | Normal aldosterone/renin ratio and serum K level | 6 |
| 2 | 71 | Female | R, medial limb | 3.4 | Nonfunctioning tumor | 136 | 150 | 6 | N.A. | 27 | |
| 3 | 66 | Male | R, medial limb | 1 | Conns syndrome | 105 | 0 | 5 | Resolution of HTN | Normal aldosterone/renin ratio and serum K level | 3 |
| 4 | 54 | Female | R, medial limb | 3.2 | Conns syndrome | 84 | 0 | 4 | Resolution of HTN | Normal aldosterone/renin ratio and serum K level | 3 |
| 5 | 47 | Male | L, lateral limb | 1.7 | Nonfunctioning tumor | 71 | 0 | 1 | N.A | 3 | |
| 6 | 56 | Female | L, medial limb | 2.5 | Conns syndrome | 80 | 0 | 1 | Improvement of HTN | Normal aldosterone/renin ratio and serum K level | 5 |
| 7 | 52 | Female | L, medial limb | 4.7 | Nonfunctioning tumor | 257 | 100 | 2 | N.A | 3 | |
| 8 | 57 | Male | L, medial limb | 3 | Nonfunctioning tumor | 132 | 100 | 6 | N.A | 3 | |
| 9 | 78 | Female | L, medial limb | 1.3 | Conns syndrome | 136 | 0 | 3 | Improvement of HTN | Normal aldosterone/renin ratio and serum K level | 19 |
| 10 | 45 | Female | R, lateral limb | 1.7 | Conns syndrome | 95 | 0 | 2 | Resolution of HTN | Normal aldosterone/renin ratio and serum K level | 22 |
| 11 | 66 | Male | R, lateral limb | 1.5 | Nonfunctioning tumor | 132 | 0 | 2 | N.A. | 21 |
HTN = hypertension; K = potassium; L = left; N.A. = not applicable; R = right.
After a full description and explanation of the procedure, all patients signed an informed consent form. The patients' data, operative time, estimated blood loss, perioperative and postoperative complications, and outcome were prospectively collected.
2.1. Operative techniques
Under general anesthesia, the patient was placed in a prone jackknife position. The operation started with the placement of a custom-made single port and the creation of the pneumoretroperitoneum, as described previously.9 A skin incision of 2–3 cm was formed just beneath the tip of the 12th rib. The subcutaneous tissues and fascia were dissected sharply. With blunt finger dissection, a retroperitoneal space was created for the placement of the single port. An Alexis wound retractor (Xsmall; Applied Medical, Rancho Santa Margarita, CA, USA) was placed in position through the incision with the bottom ring inside the abdomen. Double-layered sterile surgical gloves were then snapped onto the external ring, and the upper half of the glove parts (including the five fingers) were ligated and truncated.
A 5-mm laparoscopic standard trocar was inserted through the gloves and secured by a purse-string suture around the trocar. The capnoretroperitoneum was created by a carbon dioxide pressure of 20–30 mmHg.24; 25 ; 26 After establishing the pneumoretroperitoneum, a rigid 5-mm 30° laparoscope was inserted for inspecting the retroperitoneal cavity. Two additional 5-mm operating trocars were inserted using the same procedure. Standard straight laparoscopic instruments were used for subsequent manipulation.
After creating the retroperitoneal space by blunt dissection, Gerotas fascia and the adrenal gland were then well exposed. A careful dissection was performed to identify the tumor, based on preoperative image studies. The central adrenal vein remained intact, unless the tumor was close to a venous origin in which case the vein was controlled by Hem-o-lock clips (Weck, Research Triangle Park, NC, USA). When the tumor was exposed adequately, the tissue surrounding the normal part of the adrenal gland was dissected as minimally as possible to preserve the blood supply to the remaining tissue. Dissection was performed by a 5-mm LigaSure V Sealer/Divider (Covidien, Valleylab, Boulder, CO) to free the tumor from the remaining adrenal tissue. The operative field was inspected for hemostasis. In patients with active bleeding along the surgical margin (n = 8), fibrin glue (Floseal; Baxter Healthcare, Irvine, CA, USA) was used for adequate hemostasis. The resected adrenal tumor and its surrounding tissue were placed in a retrieval bag that was inserted directly after the detachment of the snapped double surgical gloves, and removed through the Alexis wound retractor.
2.2. Postoperative follow-up examination
The patients were regularly followed in our clinic with the first endocrine follow-up at 3 months, and then every 6 months for computed tomography imaging. The follow-up period ranged 3–27 months with a median follow up of 5 months. Follow-up computed tomography imaging detected tumor recurrence.
3. Results
Table 1 summarizes the perioperative data. No patient required conversion to an open or conventional retroperitoneoscopic approach. All adrenalectomies were successfully completed with two conventional laparoscopic instruments. The total operation time ranged 71–257 minutes (mean, 121 minutes). Bleeding was minimal in most patients (n = 8). Only three patients (Cases 2, 7, and 8) had a greater amount of blood loss, amounting to 150 mL, 100 mL, and 100 mL, respectively; none of these three patients required a blood transfusion. The fluctuation in blood pressure was easily controlled during all 11 procedures. The total drug dose for pain control postoperatively was minimal (mean meperidine use, 0.61 mg/kg per patient; mean acetaminophen use, 66.7 mg/kg per patient). The mean 2-hour, 24-hour, and 1-week postoperative visual analog scale (VAS) pain scores were 3.8, 1.5, and 0, respectively, at rest; and 5.5, 4.2, and 0.7, respectively, with activity. None of the 11 patients required pain medication after more than 12 hours postoperatively.
Pathological examination revealed that all adrenal tumors were removed en bloc ( Figure 1 ; Figure 2). No peri- or postoperative complications occurred. All patients started oral intake and ambulation on the day of the operation. The early cosmetic results were excellent. The hospital stay was 1–6 days (mean, 3 days). None of the patients experienced tumor recurrence during the follow-up period. Fig. 3 shows a representative pre- and postoperative computed tomography of a single patient.
|
|
|
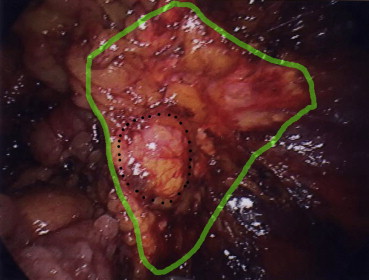
Figure 1. A right-sided adrenal tumor in a patient with an aldosterone-producing adenoma (prior to tumor resection). The green line indicates the margin of the adrenal gland and the broken line delineates the margin of the adrenal tumor. |
|
|
|
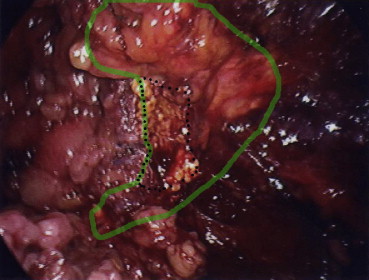
Figure 2. The green line indicates the margin of the adrenal gland and the broken line indicates the resection margin after partial adrenalectomy. |
|
|
|
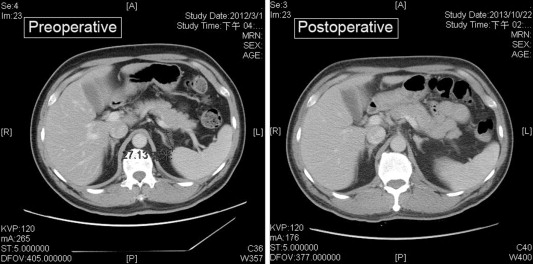
Figure 3. Pre- and postoperative (at 19 months) computed tomography images of a single patient. The postoperative image confirmed the successful removal of the adrenal tumor and showed no tumor recurrence. |
4. Discussion
Total adrenalectomy has historically been the standard procedure for treating all adrenal tumors, regardless of their size and site.13 It is effective for radically removing a causative tumor; however, it may cause the adverse effects of adrenal insufficiency and the need for steroid replacement in some scenarios such as solitary adrenal glands, bilateral adrenal involvement, or hereditary adrenal tumors. For example, one study demonstrated that 33% of patients who underwent bilateral adrenalectomy for multiple endocrine neoplasia type 2 had significant medical problems related to the lack of endogenous steroid production and required hospital admission.27 ; 28 Partial adrenalectomy was initially performed in patients with hereditary pheochromocytomas to avoid the aforementioned complications and steroid dependence. A series by Yip et al21 included 26 patients who underwent cortical-sparing surgery for hereditary pheochromocytomas: 17 (65%) patients remained steroid-independent for a median follow-up period of 71 months and only three (10%) patients had a recurrence. In another study of 36 partial adrenalectomies for pheochromocytomas in 26 patients with von Hippel-Lindau, only three (11%) patients required steroid supplementation during the long-term follow up. Radiologic imaging detected recurrence in only three (11%) patients, and all were clinically asymptomatic.29 These data strongly indicate that partial adrenalectomy should be the treatment of choice in patients with potential postoperative adrenal insufficiency and steroid dependence.
However, the indication of partial adrenalectomy in APA with a normal contralateral adrenal gland is more controversial. Ishidoya et al30 prospectively randomized 92 patients with APA by laparoscopic total adrenalectomy (n = 63) or by partial adrenalectomy (n = 29), and compared the surgical outcomes of the two groups. Both groups postoperatively had significant improvement in blood pressure, plasma renin activity, and serum aldosterone levels. Only two of the 29 patients in the partial adrenalectomy group still experienced hypertension with high plasma aldosterone. Of the 63 extirpated specimens, 17 (27.0%) adrenal glands demonstrated multiple space-occupying lesions in addition to the main APA. Ishidoya et al 30 therefore concluded that primary hyperaldosteronism is highly associated with multiple adrenal space-occupying lesions and suggested total adrenalectomy should be performed in patients with unilateral APA. However, the APA multifocality in these patients may not compromise the clinical outcome after partial adrenalectomy. A recent literature review, which included 174 patients with APA undergoing partial adrenalectomy, reveals that no patient clinically experienced a recurrence.13 Fu et al31 more recently reported their long-term results of a prospective randomized trial comparing retroperitoneoscopic partial adrenalectomy (n = 104) and total adrenalectomy (n = 108) for APA. After a mean follow-up of 96 months, all patients in each group had an improvement in hypertension. Thirty-two (29.6%) patients in the total adrenalectomy group and 29 (27.9%) patients in the partial adrenalectomy group could decrease their dose of antihypertensive medications or use fewer antihypertensive medications. All patients' plasma renin activity and aldosterone level returned to normal after surgery. No patient required potassium supplements postoperatively. Fu et al 31 therefore concluded that the therapeutic results of partial adrenalectomy and total adrenalectomy are comparable in patients with primary hyperaldosteronism due to APA. By contrast, Nakada et al12 found that angiotensin II induces aldosterone release by activating tumor uninvolved cortical cells, and found that adrenal-sparing surgery may yield better functional results, compared to unilateral total adrenalectomy. Furthermore, APA is particularly amenable to adrenal-sparing procedures because the tumor is often small, solitary, and peripherally located. 13 ; 32 All previously mentioned evidence supports partial adrenalectomy as the first line treatment in APA.
Partial adrenalectomy is a more challenging technique than total adrenalectomy. Similar to other organ-sparing procedures, the keys to success include visualizing the tumor; inspecting for additional masses, negative margins, hemostasis; and minimizing damage to the remaining adrenal tissue. Some authors suggest fully exploring the whole gland prior to dissecting the adrenal mass.16 ; 20 In our practice, we limited the range of exploration to maintain the blood supply of the remaining tissue whenever it is enough to allow a radical tumor removal. This is because preserving the adrenal gland may have a better remnant function.32 ; 33 We believe that a whole adrenal gland exploration is only necessary in the following conditions: (1) a whole exploration is required for centrally located tumors to evaluate the margin status and to ensure a radical tumor resection; and (2) in tumors that are adjacent to the adrenal vein, we also expose the whole gland to evaluate whether adrenal vein ligation is necessary. However, in tumors located peripherally or away from the adrenal vein, we intended to minimize the dissection to preserve the blood supply and/or venous drainage.
Another major issue in partial adrenalectomy is the risk of intraoperative or postoperative bleeding.15 ; 31 A satisfactory hemostasis reportedly can be achieved simply by using electrocautery and an ultrasonic scalpel while resecting adrenal tumors from the normal adrenal parenchyma.14 Other authors have added fibrin glue for hemostasis.15 ; 16 In our experience, we routinely used 5-mm LigaSure V Sealer/Divider (Covidien) to resect the tumors. The pressure of the pneumoperitoneum could decrease intraoperative bleeding. To prevent further bleeding, we also applied fibrin glue in most of our patients (n = 8). With this method, most patients in our series had minimal hemorrhaging. Only three patients had a greater blood loss (150 mL, 100 mL, and 100 mL in Cases 2, 7, and 8, respectively), which was because of the relatively larger tumor size (3.4 cm, 4.7 cm, and 3 cm, respectively). The amount blood loss in our series was generally acceptable and comparable to those reported in previous series. 7; 8; 16; 17 ; 20 Based on the results of our series and previous studies, bleeding in partial adrenalectomy can be well controlled by adequate thermocoagulation with or without using fibrin glue, and it should not be a reason to preclude this procedure.
Partial adrenalectomy has been successfully performed by various approaches. In the past, it was performed through an open incision. Since the 1990s, it has been broadly performed through conventional laparoscopy.10; 12; 16; 18; 20; 30; 31 ; 32 The results of the more than 300 cases in the literature have proven that conventional laparoscopy is an effective approach for partial adrenalectomy. A team from the National Cancer Institute of the U.S. National Institutes of Health (Bethesda, MD, USA) more recently demonstrated the feasibility of robotic-assisted laparoscopic partial adrenalectomy.22 ; 23 The choice among the different techniques may depend on instrument availability, a surgeons preferences, a patients condition, the tumor characteristics (e.g., pathology, size, location), and the cost.
Our LESS surgery has several advantages, compared to the conventional technique or the robotic-assisted laparoscopic multiport technique. First, the single-port access has a better cosmetic result. Second, all instruments used in our technique, including the custom-made single-port device and straight laparoscopic instruments, are broadly available at institutes equipped with conventional laparoscopy. Third, the cost of our procedure is the same to conventional laparoscopic surgery.
Despite all of these advantages, our LESS partial adrenalectomy procedure does require an advanced technique. The major challenges of our technique are the clashing of instruments and the lack of triangulation. More experience and practice are required to overcome these difficulties.
In 2010, Yuge et al15 reported the first case of LESS partial adrenalectomy; they successfully performed LESS partial adrenalectomy for a left adrenal tumor as the first stage in the management of a case of bilateral APA. The tumor was successfully removed en bloc with an operative time of 123 minutes and with minimal blood loss. Between December 2010 and February 2012, we also performed LESS partial adrenalectomies successfully in 11 patients. To our knowledge, this is the first series of LESS partial adrenalectomy. Our method was in several aspects somewhat different from that of Yuge et al. 15 First, we favor retroperitoneoscopy to approach the adrenal gland because it has a shorter distance between the skin incision and the adrenal gland, and liver/spleen retraction is unnecessary in most patients.9 Second, we used a custom-made single-port device and straight laparoscopic instruments, which are more broadly available and cost less than specialized commercial devices and instruments for LESS.
Our data clearly demonstrate that LESS partial adrenalectomy can be safely and effectively performed with a custom-made single-port device and conventional straight laparoscopic instruments.
References
- 1 M. Gagner, A. Lacroix, E. Bolte; Laparoscopic adrenalectomy in Cushings syndrome and pheochromocytoma; N Engl J Med, 327 (1992), p. 1033
- 2 R. Autorino, J.A. Cadeddu, M.M. Desai, et al.; Laparoendoscopic single-site and natural orifice transluminal endoscopic surgery in urology: a critical analysis of the literature; Eur Urol, 59 (2011), pp. 26–45
- 3 E. Liatsikos, P. Kallidonis, I. Kyriazis, A. Al-Aown, J.U. Stolzenburg; Urologic laparoendoscopic single-site surgery; Nat Rev Urol, 6 (2009), pp. 654–659
- 4 V.C. Lin, Y.C. Tsai, S.D. Chung, et al.; A comparative study of multiport versus laparoendoscopic single-site adrenalectomy for benign adrenal tumors; Surg Endosc, 26 (2012), pp. 1135–1139
- 5 M.M. Desai, A.K. Berger, R. Brandina, et al.; Laparoendoscopic single-site surgery: initial hundred patients; Urology, 74 (2009), pp. 805–812
- 6 J.U. Stolzenburg, P. Kallidonis, H. Till, M. Burchardt, T.R. Herrmann, E.N. Liatsikos; Current status of laparoendoscopic single-site surgery in urology; World J Urol, 27 (2009), pp. 767–773
- 7 M. Ishida, A. Miyajima, T. Takeda, M. Hasegawa, E. Kikuchi, M. Oya; Technical difficulties of transumbilical laparoendoscopic single-site adrenalectomy: comparison with conventional laparoscopic adrenalectomy; World J Urol, 31 (2013), pp. 199–203
- 8 M.K. Walz, H. Groeben, P.F. Alesina; Single-access retroperitoneoscopic adrenalectomy (SARA) versus conventional retroperitoneoscopic adrenalectomy (CORA): a case-control study; World J Surg, 34 (2010), pp. 1386–1390
- 9 S.D. Chung, C.Y. Huang, S.M. Wang, H.C. Tai, Y.C. Tsai, S.C. Chueh; Laparoendoscopic single-site (LESS) retroperitoneal adrenalectomy using a homemade single-access platform and standard laparoscopic instruments; Surg Endosc, 25 (2011), pp. 1251–1256
- 10 E.K. Diner, M.E. Franks, A. Behari, W.M. Linehan, M.M. Walther; Partial adrenalectomy: the National Cancer Institute experience; Urology, 66 (2005), pp. 19–23
- 11 M.M. Walther, H.R. Keiser, P.L. Choyke, W. Rayford, J.C. Lyne, W.M. Linehan; Management of hereditary pheochromocytoma in von Hippel-Lindau kindreds with partial adrenalectomy; J Urol, 161 (1999), pp. 395–398
- 12 T. Nakada, Y. Kubota, I. Sasagawa, T. Yagisawa, M. Watanabe, M. Ishigooka; Therapeutic outcome of primary aldosteronism: adrenalectomy versus enucleation of aldosterone-producing adenoma; J Urol, 153 (1995), pp. 1775–1780
- 13 D.R. Kaye, B.B. Storey, K. Pacak, P.A. Pinto, W.M. Linehan, G. Bratslavsky; Partial adrenalectomy: underused first line therapy for small adrenal tumors; J Urol, 184 (2010), pp. 18–25
- 14 C.H. Liao, S.C. Chueh, K.D. Wu, M.H. Hsieh, J. Chen; Laparoscopic partial adrenalectomy for aldosterone-producing adenomas with needlescopic instruments; Urology, 68 (2006), pp. 663–667
- 15 K. Yuge, A. Miyajima, M. Hasegawa, et al.; Initial experience of transumbilical laparoendoscopic single-site surgery of partial adrenalectomy in patient with aldosterone-producing adenoma; BMC Urol, 10 (2010), p. 19
- 16 K. Jeschke, G. Janetschek, R. Peschel, L. Schellander, G. Bartsch, K. Henning; Laparoscopic partial adrenalectomy in patients with aldosterone-producing adenomas: indications, technique, and results; Urology, 61 (2003), pp. 69–72 discussion 72
- 17 I. Sasagawa, Y. Suzuki, K. Itoh, et al.; Posterior retroperitoneoscopic partial adrenalectomy: clinical experience in 47 procedures; Eur Urol, 43 (2003), pp. 381–385
- 18 M.M. Walther, J. Herring, P.L. Choyke, W.M. Linehan; Laparoscopic partial adrenalectomy in patients with hereditary forms of pheochromocytoma; J Urol, 164 (2000), pp. 14–17
- 19 M.K. Walz, K. Peitgen, D. Diesing, et al.; Partial versus total adrenalectomy by the posterior retroperitoneoscopic approach: early and long-term results of 325 consecutive procedures in primary adrenal neoplasias; World J Surg, 28 (2004), pp. 1323–1329
- 20 X.J. Wang, Z.J. Shen, Y. Zhu, et al.; Retroperitoneoscopic partial adrenalectomy for small adrenal tumours (< or = 1 cm): the Ruijin clinical experience in 88 patients; BJU Int, 105 (2010), pp. 849–853
- 21 L. Yip, J.E. Lee, S.E. Shapiro, et al.; Surgical management of hereditary pheochromocytoma; J Am Coll Surg, 198 (2004), pp. 525–534 discussion 534–525
- 22 K.P. Asher, G.N. Gupta, R.S. Boris, P.A. Pinto, W.M. Linehan, G. Bratslavsky; Robot-assisted laparoscopic partial adrenalectomy for pheochromocytoma: the National Cancer Institute technique; Eur Urol, 60 (2011), pp. 118–124
- 23 R.S. Boris, G. Gupta, W.M. Linehan, P.A. Pinto, G. Bratslavsky; Robot-assisted laparoscopic partial adrenalectomy: initial experience; Urology, 77 (2011), pp. 775–780
- 24 R.M. Giebler, M. Behrends, T. Steffens, M.K. Walz, K. Peitgen, J. Peters; Intraperitoneal and retroperitoneal carbon dioxide insufflation evoke different effects on caval vein pressure gradients in humans: evidence for the starling resistor concept of abdominal venous return; Anesthesiology, 92 (2000), pp. 1568–1580
- 25 R.M. Giebler, M.K. Walz, K. Peitgen, R.U. Scherer; Hemodynamic changes after retroperitoneal CO2 insufflation for posterior retroperitoneoscopic adrenalectomy; Anesth Analg, 82 (1996), pp. 827–831
- 26 M.K. Walz, P.F. Alesina, F.A. Wenger, et al.; Posterior retroperitoneoscopic adrenalectomy—results of 560 procedures in 520 patients; Surgery, 140 (2006), pp. 943–948 discussion 948–950
- 27 M. Telenius-Berg, M.A. Ponder, B. Berg, B.A. Ponder, S. Werner; Quality of life after bilateral adrenalectomy in MEN 2; Henry Ford Hosp Med J, 37 (1989), pp. 160–163
- 28 T.C. Lairmore, D.W. Ball, S.B. Baylin, S.A. Wells Jr.; Management of pheochromocytomas in patients with multiple endocrine neoplasia type 2 syndromes; Ann Surg, 217 (1993), pp. 595–601 discussion 601
- 29 J.N. Benhammou, R.S. Boris, K. Pacak, P.A. Pinto, W.M. Linehan, G. Bratslavsky; Functional and oncologic outcomes of partial adrenalectomy for pheochromocytoma in patients with von Hippel-Lindau syndrome after at least 5 years of followup; J Urol, 184 (2010), pp. 1855–1859
- 30 S. Ishidoya, A. Ito, K. Sakai, et al.; Laparoscopic partial versus total adrenalectomy for aldosterone producing adenoma; J Urol, 174 (2005), pp. 40–43
- 31 B. Fu, X. Zhang, G.X. Wang, et al.; Long-term results of a prospective, randomized trial comparing retroperitoneoscopic partial versus total adrenalectomy for aldosterone producing adenoma; J Urol, 185 (2011), pp. 1578–1582
- 32 T. Imai, Y. Tanaka, T. Kikumori, et al.; Laparoscopic partial adrenalectomy; Surg Endosc, 13 (1999), pp. 343–345
- 33 Y. Ikeda, H. Takami, G. Tajima, et al.; Laparoscopic partial adrenalectomy; Biomed Pharmacother, 56 (suppl 1) (2002), pp. 126s–131s
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?