Summary
Infection of a vascular prosthesis after a bypass surgery is relatively rare. However, once developed, serious complications can occur, such as bleeding, sepsis, and organ ischemia, occasionally resulting in leg amputation or even death in some cases. The treatment of a vascular prosthesis infection involves the necessary removal of the infected graft; subsequently, an extra-anatomical bypass surgery is often considered. We herein report a case in which postoperative methicillin-resistant Staphylococcus aureus infection caused dehiscence of the femoral vessels and exposure of the graft vessel and anastomosed area. The infected tissue was surgically removed (debridement), and the patients condition was successfully treated by the application of a nonadherent dressing and vacuum-assisted closure therapy combined with the bridging technique.
Keywords
infected prosthetic graft;methicillin-resistant Staphylococcus aureus;wound-closure techniques
1. Introduction
Infection of a vascular prosthesis following a bypass surgery is relatively rare (0.92–6.4%). However, once developed, serious complications can occur, such as bleeding, sepsis, and organ failure, resulting in leg amputation (10–70%) or even death (10–20%).1; 2; 3 ; 4
The treatment of a vascular prosthesis infection involves the necessary removal of the infected graft and then draining pus from the infected site (abscess). A subsequent extra-anatomical bypass surgery is often considered. Previously published reports suggest that graft preservation is possible in cases satisfying the following requirements: (1) absence of infection at the entire site of the graft; (2) absence of signs of sepsis; (3) patency of the unaffected parts of the graft; and (4) absence of Pseudomonas aeruginosa infection. 2 ; 5 Various reports have shown that vascular prosthesis was successfully preserved using a vacuum-assisted closure system (VAC; KCI, San Antonio, TX, USA). 6; 7 ; 8 We herein report a case in which methicillin-resistant Staphylococcus aureus (MRSA) infection following femoropopliteal artery bypass surgery caused dehiscence of the femoral and popliteal wounds and exposure of the graft. In this case, we successfully preserved the graft using a nonadherent dressing and a VAC system combined with the bridging technique.
2. Case report
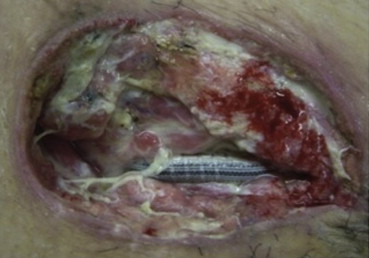
A 59-year-old man underwent a common femoral-to-above-knee popliteal artery bypass with a Dacron graft for the treatment of critical limb ischemia with first toe necrosis. An antibiotic (cefazolin) was administered by drip infusion 30 minutes prior to and 3 hours after the operation. He had a history of diabetes mellitus and had been receiving maintenance hemodialysis for the past 2 years. The patient had undergone coronary artery bypass surgery 1 month earlier. Three days after the bypass surgery, he developed a spike fever (>38°C). On the 8th postoperative day, pus discharge was observed from the femoral wound and the popliteal region that was accompanied by redness and pain in the femoral region to the flank. White blood cell count and C-reactive protein level were higher than normal limits (18,000/μL and 28.09 mg/dL, respectively). The following day, the femoral and popliteal wounds were exposed in the operating room. On exposing the graft, a massive milky white pus discharge was noted, which was accompanied in both regions by macroscopically visible poor granulation with wound infection (Fig. 1). Results of computed tomography scans did not reveal any abscess in the subcutaneous tunnel of the femoral region along the vascular prosthesis; therefore, the prosthesis was not removed. Instead, debridement was performed, and to begin the therapy, a VAC GranuFoam dressing (KCI) of a size to match that of the wound was inserted into the femoral and popliteal regions. During this insertion, the prosthesis was covered with a nonadherent dressing in order to avoid direct contact of the GranuFoam dressing with the prosthesis and the anastomosed area, as described by Dosluoglu et al.5 The GranuFoam dressing (KCI) in the femoral wound was connected to that in the popliteal wound using the bridging technique,9 followed by the serial application of negative pressure (125 mmHg; Fig. 2).
|
|
|
Figure 1. Macroscopically visible poor granulation and signs of infection observed on the 9th postoperative day. |
|
|
|
Figure 2. The GranuFoam dressing in the femoral wound is connected to that in the popliteal wound using the bridging technique to avoid direct exposure of the intact skin to the negative pressure. |
Incubation of the specimens from the wounds at the femoral and popliteal sites revealed MRSA infection. However, the blood culture was negative for MRSA infection. Vancomycin (VCM) treatment was initiated after the detection of pus discharge from the wound, given the possibility of MRSA infection. After the MRSA infection in the graft-exposed area was confirmed, VCM treatment was switched with linezolid. In subsequent cultures, MRSA was detected until the 16th postoperative day, and Klebsiella pneumoniae was isolated from the femoral site on the 25th postoperative day. The final culture at the time of the wound closure on the 46th postoperative day was negative. Because the patient did not show clinical signs of infection aggravation, the GranuFoam dressing (KCI) for VAC therapy was renewed every week, and the popliteal wound was closed during the 2nd week of VAC therapy. In the 5th week, the wound showed macroscopically healthy granulation; therefore, the patient was weaned from VAC therapy, and the femoral wound was closed. Antibiotics were administered for 2 months after the bypass surgery. Currently, approximately 7 months after wound closure, the patient has been free of wound abnormalities and shows no signs of reinfection.
3. Discussion
Treatments usually performed in cases of vascular prosthesis infection include: (1) complete removal of the prosthesis; (2) aggressive debridement; (3) cleansing to reduce the bacterial count and to ensure sufficient blood flow with granulation formation stimulation, use of a musculocutaneous flap, and so on; and (4) long-term antibacterial drug treatment.5 However, removal of the graft might cause ischemia, and an extra-anatomical bypass surgery to avoid ischemia in such cases is often technically difficult because of the low graft patency rate and the need to avoid the infected area.
Despite serious concerns about using VAC therapy with exposed grafts, many recent reports have described cases that were completely cured by VAC therapy without omental transposition or muscle flap closure while preserving patency of the vascular prosthesis. Using this therapy, a curing rate of 82% has been reported for the inguinal region. Muscle flap closure was not used in the present case because reinfection had been reported in 35% of cases following this manipulation.6; 7; 8 ; 10 The VAC therapy may be considered the first choice of therapy for perivascular groin infections, leading to a high rate of vascular graft reconstruction preservation.7
Total and partial graft preservation has been reported in 42.5% and 35.8% of all cases, respectively, with a wound healing rate of 85% and 71%, respectively. Much lower (40%) success rates have been reported in cases in which P. aeruginosa was the cause of infection. 2; 5 ; 11
The most commonly isolated bacterial species was S. aureus (approximately 30%), including MRSA (12%). 1 ; 5 In the present case, although MRSA was isolated, we applied VAC therapy to observe its effects (bringing the wound edges closer together, adjusting the wound environments, reducing edema, and increasing blood flow around the wound). 12; 13 ; 14 In fact, VAC therapy was applied in a manner similar to that previously reported. 5; 6; 7; 9 ; 15
In the present case, we had the impression that the therapy stimulated the process of granulation tissue formation similar to the observation reported by Colwell et al.16 However, considering the reports on bleeding and pseudoaneurysms observed after VAC therapy for exposed vascular grafts,7; 8; 10; 11; 12 ; 13 adequate care during therapy and further data collection are needed.
References
- 1 W.H. Edwards Jr., R.S. Martin 3rd, J.M. Jenkins, W.H. Edwards Sr., J.L. Mulherin Jr.; Primary graft infections; J Vasc Surg, 6 (1987), pp. 235–239
- 2 K.D. Calligaro, F.J. Veith, M.L. Schwartz, et al.; Selective preservation of infected prosthetic arterial grafts. Analysis of a 20-year experience with 120 extracavitary-infected grafts; Ann Surg, 220 (1994), pp. 461–469
- 3 W.G. Liekweg Jr., L.J. Greenfield; Vascular prosthetic infections: collected experience and results of treatment; Surgery, 81 (1977), pp. 335–342
- 4 D.P. Newington, P.W. Houghton, R.N. Baird, M. Horrocks; Groin wound infection after arterial surgery; Br J Surg, 78 (1991), pp. 617–619
- 5 H.H. Dosluoglu, C. Loghmanee, P. Lall, G.S. Cherr, L.M. Harris, M.L. Dryjski; Management of early (<30 day) vascular groin infections using vacuum-assisted closure alone without muscle flap coverage in a consecutive patient series; J Vasc Surg, 51 (2010), pp. 1160–1166
- 6 H.H. Dosluoglu, D.K. Schimpf, R. Schultz, G.S. Cherr; Preservation of infected and exposed vascular grafts using vacuum assisted closure without muscle flap coverage; J Vasc Surg, 42 (2005), pp. 989–992
- 7 S. Svensson, C. Monsen, T. Kölbel, S. Acosta; Predictors for outcome after vacuum assisted closure therapy of peri-vascular surgical site infections in the groin; Eur J Vasc Endovasc Surg, 36 (2008), pp. 84–89
- 8 P. Impellizzeri, H. Dardik, H.J. Shah, A. Brotman-O'Neil, I.M. Ibrahim; Vacuum-assisted closure therapy with omental transposition for salvage of infected prosthetic femoral-distal bypass involving the femoral anastomosis; J Vasc Surg, 54 (2011), pp. 1154–1156
- 9 P.E. Banwell, D. Evison, I.M. Whitworth; Vacuum therapy in degloving injuries of the foot: technical refinements; Br J Plast Surg, 55 (2002), pp. 264–266
- 10 S.M. Taylor, D.A. Weatherford, E.M. Langan 3rd, J.S. Lokey; Outcomes in the management of vascular prosthetic graft infections confined to the groin: a reappraisal; Ann Vasc Surg, 10 (1996), pp. 117–122
- 11 B.E. Sumpio, D.E. Allie, K.A. Horvath, et al.; Role of negative pressure wound therapy in treating peripheral vascular graft infections; Vascular, 16 (2008), pp. 194–200
- 12 V. Saxena, C.W. Hwang, S. Huang, Q. Eichbaum, D. Ingber, D.P. Orgill; Vacuum-assisted closure: microdeformations of wounds and cell proliferation; Plast Reconstr Surg, 114 (2004), pp. 1086–1098
- 13 M.J. Morykwas, L.C. Argenta, E.I. Shelton-Brown, W. McGuirt; Vacuum-assisted closure: a new method for wound control and treatment: animal studies and basic foundation; Ann Plast Surg, 38 (1997), pp. 553–562
- 14 A.K. McNulty, M. Schmidt, T. Feeley, K. Kieswetter; Effects of negative pressure wound therapy on fibroblast viability, chemotactic signaling, and proliferation in a provisional wound (fibrin) matrix; Wound Repair Regen, 15 (2007), pp. 838–846
- 15 M.L. Venturi, C.E. Attinger, A.N. Mesbahi, C.L. Hess, K.S. Graw; Mechanisms and clinical applications of the vacuum-assisted closure (VAC) device: a review; Am J Clin Dermatol, 6 (2005), pp. 185–194
- 16 A.S. Colwell, M.C. Donaldson, M. Belkin, D.P. Orgill; Management of early groin vascular bypass graft infections with Sartorius and rectus femoris flaps; Ann Plast Surg, 52 (2004), pp. 49–53
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?