Summary
Background
Splenectomy is a common surgical procedure, but few reports focus on nontraumatic splenectomies. The aim of this study was to determine the predictors of morbidity and mortality of patients submitted to elective nontraumatic splenectomy.
Methods
A descriptive cross-sectional study of 152 consecutive, nonselected, nontraumatic patients operated on by splenectomy between 1996 and 2010 was carried out. Clinical, laboratory, and surgical data, histological findings, perioperative mortality, and postoperative complications according to Clavien-Dindo classification, were recorded. Factors related to morbidity and mortality were analyzed.
Results
Of the 152 patients (89 male and 63 female; mean age 49.8 ± 17.8 years), 74 (48.7%) were operated on for malignant hematologic disorders, 44 for benign hematologic process, and 34 for other nonhematologic disorders. The spleen was enlarged in 95 patients (62.5%) and 78 patients (51.3%) had hypersplenism. The overall complications rate was 40.1%: Grades I and II in 27 cases (17.7%), and Grades III and IV in 23 patients (15.1%). Perioperative mortality was 7.2% (11 patients). In univariate analysis, significant negative predictors for morbidity were age (p = 0.004), anemia (p = 0.03), leukocytosis (p = 0.016), and blood transfusions (p < 0.001). In the multivariate analysis, only the need for blood transfusion remained as an independent prognostic factor (p = 0.001). Related to mortality, negative prognostic factors were age (p = 0.003), leukocytosis (p = 0.048), American Society of Anesthesiologists (ASA) score (p < 0.001), blood transfusion (p < 0.001), pleural effusion (p = 0.031), and pneumonia (p = 0.001). Pneumonia remained an independent prognostic factor of mortality (p = 0.024).
Conclusion
Blood loss is the most important prognostic factor for postoperative complications after nontraumatic splenectomies. Pneumonia is the main prognosis factor for perioperative mortality.
Keywords
morbidity;mortality;nontrauma;prognosis factors;splenectomy
1. Introduction
Splenectomy is a common surgical procedure related to spleen trauma. However, many other elective surgical indications for splenectomy have been described in nontrauma patients including hematological disorders, lymphoma staging, and primary tumors of the spleen. In fact, splenic surgery is indicated in an interesting and wide variety of disease processes.1; 2 ; 3
There are many studies in which the prognosis factors related to the morbidity and mortality in traumatic splenectomies have been analyzed.4; 5 ; 6 However, very few studies of the prognosis factors in nontraumatic splenectomies have been reported.7; 8; 9 ; 10
The aim of this study was to identify and analyze factors related to the perioperative morbidity and mortality in elective, nontraumatic splenectomies.
2. Patients and methods
A descriptive cross-sectional study of 152 consecutive, nonselected, nontraumatic patients operated on by splenectomy between 1996 and 2010 was carried out. Registration of the patients medical data in an electronic database was recorded according to the guidelines of the Ethics Committee of our institution. The authorization to use these data was given by the patients in a written and informed consent. The setting was a tertiary-care hospital that attends a population of approximately 400,000 people. Exclusion criteria included incidental splenectomies, and splenectomies performed during en-bloc resection for malignancy in an adjacent organ, such as the stomach, colon, adrenal gland, or pancreas.
The following variables were recorded:
- Clinical data: age, sex, the anesthetic risk evaluation according to the American Society of Anesthesiologists score (ASA), splenic disorders that indicated the splenectomy, splenomegaly, and hypersplenism. In accordance with the European Association for Endoscopic Surgery (EAES) guidelines,11 splenomegaly was defined as a long axis exceeding 15 cm, as measured by ultrasound along its longer dimension. Hypersplenism12 was defined by thrombocytopenia and/or neutropenia resulting from blood cell sequestration in an enlarged spleen; in some cases, this can also be responsible for mild anemia. The definitive diagnostic of the splenic disorder was obtained from the histopathological study of the removed spleen.
- Preoperative laboratory variables: anemia (hemoglobin < 13 g/dL in males and < 12 g/dL in females), leukocytosis (> 11 × 109/L), leukopenia (< 4 × 109/L), and thrombopenia (< 100 × 109/L).
- Surgical variables: surgical procedure (laparoscopic vs. nonlaparoscopic approach), total surgical time (defined as the time from the skin incision for portal placement to the end of skin suturing), and perioperative blood transfusions.
- Macroscopic histological findings: size (centimeters) and weight (grams) of the specimen of spleen removed.
- Postoperative variables: postoperative hospital stay, perioperative complications, and perioperative mortality. Perioperative mortality was defined either as any death occurring within 30 days of surgery or any later death that was considered to be a direct consequence of a postoperative complication. To evaluate the severity of the complications, the Clavien-Dindo classification13 ; 14 was used: Grade I and Grade II are complications that include only minor deteriorations from the normal postoperative course and can be easily treated with drugs, blood transfusion, and nutritional supply; Grade III complications require interventional treatment; Grade IV complications are life-threatening complications with intensive care unit management; and Grade V means death of the patient.
Univariate analyses were performed to compare each independent variable (age, sex, ASA, splenic disorders, splenomegaly, hypersplenism, surgical procedure, surgical time, blood transfusions, and size and weight of the removed spleen) with either the morbidity (Clavien-Dindo Grades I and II vs. Grades III and IV) and mortality variables. Two models of multivariate analysis were created in order to identify risk factors of postoperative morbidity and perioperative mortality, respectively.
2.1. Statistical analysis
Data were analyzed using the statistical package SPSS version 15.0 for Windows (SPSS Inc., Chicago, IL, USA). Results of continuous variables were given as mean [± standard deviation (SD), confidence interval (95% CI)]. Categorical variables were expressed as percentages. The Chi-square test or Fishers test was used to compare categorical data. For the parametric distribution, Student t test or one-way analysis of variance (ANOVA) test were used to compare the mean values of two or more groups, respectively. For nonparametric variables, the Mann-Whitney U test or the Kruskal-Wallis test were used to compare the median values of the response variable. Two stepwise logistic regression analyses were performed. In the first model, the outcome variable was “postoperative complications”, and in the second model, the outcome variable was “perioperative mortality”, both categorized as binary variables. Each model included as explanatory or predictor factors the variables that were suggested in the univariate analysis to be more strongly and significantly associated with complications or mortality. A p < 0.05 level was used for the inclusion of the variables in the model. When two or more explanatory variables were correlated, only the variable with the strongest relationship with the outcome variable was included in the model. These analyses were designed to establish independent risk factors of morbidity and mortality for nontraumatic splenectomy. The 95% CI was determined as an indication of the precision of an estimate of a population value. The odds ratio (OR) was calculated as an estimate of relative risk between two groups on the basis of the mortality as outcome. Statistical significance was defined as p < 0.05.
3. Results
Out of the 152 patients, 89 (58.6%) were male and 63 (41.4%) were female. The mean ± SD age was 49.8 ± 17.8 years (95% CI: 46.9–52.6), with an age range of 14–83 years, and the median age was 53 years (interquartile range: 29). The distribution of the anesthetic risk scale ASA was as follows: 15 patients were ASA I (9.9%), 63 patients were ASA II (41.4%), 53 patients were ASA III, and 21 patients were ASA IV (13.8%).
Seventy-four patients (48.7%) were operated on for malignant hematologic disorders: 64 lymphomas and 10 myeloproliferative syndromes. In 30 patients (19.7%), the indication for splenectomy was a benign hematologic disorder: idiopathic thrombocytopenic purpura (ITP). Fourteen splenectomized patients (9.2%) suffered hemolytic anemia: hereditary spherocytosis (10 cases), Louis-Ville hemoglobinopathy (two cases), autoimmune hemolytic anemia (one case), congenital dyserythropoiesis (one case). In nine patients (5.9%), a vascular disorder indicated the splenectomy: portal hypertension (six cases), and aneurysm of splenic artery (three cases). In another nine patients (5.9%), the splenectomy was performed because of a benign neoplasm: hemangioma (two cases) and splenic cysts (two cases). An infectious disease was diagnosed in eight cases (5.3%): splenic abscesses (four cases), leishmaniasis (one case), chronic granulomatous disease (one case), mycotic splenic artery aneurysm (one case), and splenic atypical mycobacterial disease (one case). Another eight patients were included in a miscellaneous group: amyloidosis (one case), nonspecific lymphoid hyperplasia (two cases), metastasis of squamous cell carcinoma (one case), systemic lupus erythematosus (one case), Wegeners granulomatosis (one case), and normal spleen (three cases).
The spleen was enlarged in 95 patients (62.5%). This data could not be recorded in one patient. Hypersplenism was diagnosed in 78 patients (51.3%).
One hundred and four patients were operated by laparotomy (68.4%). In the remaining 48 patients, a laparoscopic approach was initially carried out, but the conversion of the procedure to open surgery was necessary in 11 cases (22.9%). The mean surgical time was 127.1 ± 44.6 minutes, the range was 45–285 minutes, and the median surgical time was 120 minutes (interquartile range: 55). Perioperatively, 66 patients (43.4%) underwent at least one blood transfusion.
The mean size of the spleen specimens removed was 18.1 ± 5.9 cm, range 7–32, and the median size was 18 cm. (interquartile range: 9). The mean weight ± SD of the specimens removed was 855.9 ± 779.3 g, the range was 39–3905 g, and the median weight was 581.5 g (interquartile range: 1062).
Seventeen patients (11.1%) were re-operated on. The overall postoperative complication rate following nontraumatic splenectomy in this series of 152 patients was 40.1%: pleural effusion (28 patients), subphrenic collection (15 cases), ileus (15 patients), pneumonia (13 cases), wound infection (10 cases), atelectasis (nine cases), hyperamilasemia (five cases), pancreas pseudocyst (four cases), and pneumothorax (three patients).
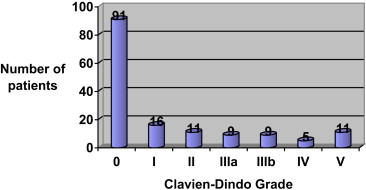
At least one complication was detected in 61 patients (Fig. 1). Clavien-Dindo Grade I and Grade II complications were found in 16 cases and 11 cases, respectively, Grade IIIa in nine patients (5,9%), Grade IIIb in 9 patients, and Grade IV in five patients (3.3%).
|
|
|
Figure 1. Overall postoperative complications after nontraumatic splenectomy according to the Clavien-Dindo score. |
Perioperative mortality (Grade V) was 7.2% (11 patients). The causes of death included septic shock in three patients, multiple organ failure in six patients, liver failure in one patient, and hypovolemic shock due to bleeding in one patient.
In the univariate analysis, the factors related to complications according Clavien-Dindo score are shown in Table 1. Significant univariate negative predictors were the variables age (p = 0.004), anemia (p = 0.03), leukocytosis (p = 0.016), and perioperative blood transfusions (p = 0.029). Significant favorable predictors were the presence of thrombocytopenic purpura (p = 0.01) and laparoscopic approach (p = 0.059). In the multivariate analysis ( Table 2) only the need for perioperative blood transfusion remained as an independent prognostic factor of morbidity (p = 0.001; OR: 3.50; 95% CI: 1.67–7.34).
| Parameter | N (%) | Clavien-Dindo Grade 0n = 91 (59.9%) | Clavien-Dindo Grades I and IIn = 27 (17.8%) | Clavien-Dindo Grades III–Vn = 34 (22.4%) | p |
|---|---|---|---|---|---|
| Preoperative status | |||||
| Age (mean ± SD) | 152 | 46.0 ± 17.1 | 53.9 ± 16.0 | 56.6 ± 18.7 | 0.004∗ |
| Male sex | 89 (58.6) | 50 (40.9) | 17 (63.0) | 22 (64.7) | 0.539 |
| Anemia | 98 (64.5) | 551 (56.0) | 21 (77.8) | 26 (76.5) | 0.03∗ |
| Leukocytosis | 42 (27.6) | 20 (22.0) | 6 (22.2) | 16 (47.1) | 0.016∗ |
| ASA III–IV | 74 (48.7) | 38 (41.8) | 14 (51.9) | 22 (64.7) | 0.07 |
| Splenomegaly | 95 (62.9) | 55 (61.1) | 19 (70.4) | 21 (61.8) | 0.674 |
| Hypersplenism | 78 (51.3) | 44 (48.4) | 19 (70.4) | 15 (44.1) | 0.084 |
| Splenic disorder | |||||
| Malignant hematologic disorder | 74 (48.7) | 41 (45.1) | 18 (66.7) | 15 (44.1) | 0.119 |
| Thrombocytopenic purpura | 30 (19.7) | 25 (27.5) | 1 (3.7) | 4 (11.8) | 0.01∗ |
| Hemolytic anemia | 14 (9.2) | 10 (11.0) | 3 (11.1) | 1 (2.9) | 0.360 |
| Surgical variables | |||||
| Laparoscopic approach | 37 (24.3) | 29 (31.9) | 4 (14.8) | 4 (11.8) | 0.029∗ |
| Perioperative transfusions | 66 (43.4) | 26 (28.6) | 14 (51.9) | 26 (76.5) | < 0.001∗ |
| Surgical time (mean ± SD) | 152 | 123.8 ± 46.2 | 130.2 ± 48.0 | 133.4 ± 37.6 | 0.524 |
| Specimen removed | |||||
| Size (cm) (mean ± SD) | 152 | 17.6 ± 5.7 | 19.5 ± 6.2 | 17.9 ± 6.2 | 0.351 |
| Weight (g) (mean ± SD) | 152 | 770.1 ± 683.9 | 986.7 ± 739.9 | 981.5 ± 1010.6 | 0.255 |
∗Statistically significant.
| Parameter | B | SE | Wald | p | OR (95% CI) |
|---|---|---|---|---|---|
| Age | 0.014 | 0.012 | 1.330 | 0.249 | 1.01 (0.99–1.04) |
| Leukocytosis | 0.516 | 0.408 | 1.596 | 0.206 | 1.68 (0.75–3.73) |
| Thrombocytopenic purpura | −0.624 | 0.588 | 1.125 | 0.289 | 0.54 (0.17–1.70) |
| Laparoscopic surgery | −0.466 | 0.507 | 0.846 | 0.358 | 0.63 (0.23–1.69) |
| Blood transfusion | 1.252 | 0.378 | 10.953 | 0.001∗ | 3.50 (1.67–7.34) |
| Constant | −1.637 | 0.705 | 5.397 | 0.020 |
∗ Statistically significant.
B = regression coefficient; CI = confidence interval; OR = odds ratio; SE = standard error; Wald = test statistic.
Univariate analyses of mortality-related factors are summarized in Table 3. Age (p = 0.003), leukocytosis (p = 0.048), ASA score (p < 0.001), perioperative blood transfusion (p = 0.001), postoperative pleural effusion (p = 0.031), and postoperative pneumonia (p = 0.001) were associated with mortality in a statistically significant way. Nevertheless, only pneumonia as a postoperative complication was identified as independent risk of mortality in multivariate analysis (p = 0.024; OR: 5.59; 95% CI: 1.2–24.85; Table 4).
| N (%) | Aliven = 141 (92.8%) | Deadn = 11 (7.2%) | p | OR (95% CI) | |
|---|---|---|---|---|---|
| Preoperative status | |||||
| Age (mean ± SD) | 152 | 48.6 ± 17.3 | 64.7 ± 17.4 | 0.003∗ | — |
| Male sex | 89 (58.6) | 80 (56.7) | 9 (81.8) | 0.124 | 0.29 (0.06–1.39) |
| Anemia | 98 (64.5) | 88 (62.4) | 10 (90.9) | 0.05 | 6.02 (0.75–48.39) |
| Leukocytosis | 42 (27.6) | 36 (25.5) | 6 (54.5) | 0.048∗ | 3.50 (1.01–12.16) |
| ASA III–IV | 74 (48.7) | 63 (44.7) | 11 (100) | < 0.001∗ | 1.18 (1.07–1.29) |
| Splenomegaly | 95 (62.9) | 86 (61.4) | 9 (81.8) | 0.153 | 2.83 (0.59–13.58) |
| Hypersplenism | 78 (51.3) | 72 (51.1) | 6 (54.5) | 0.824 | 1.15 (0.34–3.94) |
| Splenic disorder | |||||
| Malignant hematologic disorder | 74 (48.7) | 67 (47.5) | 7 (63.6) | 0.303 | 1.93 (0.54–6.90) |
| Thrombocytopenic purpura | 30 (19.7) | 29 (20.6) | 1 (9.1) | 0.320 | 0.39 (0.05–3.14) |
| Hemolytic anemia | 14 (9.2) | 14 (9.9) | 0 (0.0) | 0.332 | 0.92 (0.88–0.97) |
| Surgical variables | |||||
| Laparoscopic approach | 37 (24.3) | 36 (25.5) | 1 (9.1) | 0.200 | 0.29 (0.04–2.36) |
| Perioperative transfusions | 66 (43.4) | 55 (39.0) | 11 (100) | < 0.001∗ | 1.2 (1.08–1.34) |
| Surgical time (mean ± SD) | 152 | 126.5 ± 44.6 | 135 ± 46.2 | 0.543 | — |
| Specimen removed | |||||
| Size (cm) (mean ± SD) | 152 | 18.0 ± 6.1 | 17.8 ± 4.4 | 0.914 | — |
| Weight (g) (mean ± SD) | 152 | 854.7 ± 796.4 | 871.2 ± 537.9 | 0.946 | — |
| Postoperative complications | |||||
| Pleural effusion | 28 (18.4) | 23 (16.3) | 5 (45.5) | 0.031∗ | 4.28 (1.20–15.20) |
| Subphrenic collection | 15 (9.9) | 12 (8.5) | 3 (27.3) | 0.079 | 4.03 (0.94–17.20) |
| Ileus | 15 (9.9) | 12 (8.5) | 3 (27.3) | 0.079 | 4.03 (0.94–17.20) |
| Pneumonia | 13 (8.6) | 8 (5.7) | 5 (45.5) | 0.001∗ | 13.85 (3.47–55.33) |
| Wound infection | 10 (6.6) | 8 (5.7) | 2 (18.2) | 0.156 | 3.69 (0.68–20.03) |
∗Statistically significant.
| Parameter | B | SE | Wald | p | OR (95% CI) |
|---|---|---|---|---|---|
| Age (y) | 0.016 | 0.025 | 0.416 | 0.519 | 1.02 (0.97–1.07) |
| ASA classification | 1.007 | 0.568 | 3.143 | 0.076 | 2.74 (0.90–8.33) |
| Blood transfusion | 1.654 | 1.131 | 2.140 | 0.144 | 5.23 (0.57–47.95) |
| Pneumonia | 1.721 | 0.761 | 5.116 | 0.024∗ | 5.59 (1.26–24.85) |
| Constant | −7.886 | 2.007 | 15.445 | 0.000 |
∗Statistically significant.
B = regression coefficient; CI = confidence interval; OR = odds ratio; SE = standard error; Wald = test statistic.
4. Discussion
Splenectomy in nontrauma patients is a frequent surgery for diagnosis and therapeutic indications, and is related to a high rate of complications, but very few papers have been published on this topic.9 ; 10
The prevalence of morbidity associated with nontraumatic splenectomies ranges between 27% and 52%.7; 8; 9 ; 10 This wide range could be because a lack of uniform reporting of negative outcomes, which makes interpretation of surgical literature difficult. Clavien and Dindo have13 ; 14 have introduced an interesting grading system of postoperative complications, i.e., a five-scale classification system with the aim of presenting an objective, simple, reliable, and reproducible way of reporting all complications after surgery. Few studies have used this score for postoperative complications in splenectomy.15 We consider that this classification represents an objective and simple way of reporting all complications in patients undergoing splenectomies. In our study, the overall morbidity rate was found to be 40%. Nevertheless, complications of the Clavien-Dindo Types III and IV were only 22.4%.
According to the literature findings, the most common postoperative side-effects after this procedure are the pulmonary complications. Globally, these complications may arise in 32% of the patients,10 and include pleural effusion, atelectasis, pneumonia, and bronchitis. Subphrenic abscess is also a common complication reported in previous articles7; 8 ; 10 with a range of 3–6%, which is comparable with our findings.
In Dawson et als series,16 pneumonia occurred in 13% of patients, and Aksnes et al7 reported postoperative pneumonia in 17%. Wiseman et al17 also reported that splenectomy is associated with an increase in early postoperative infections after laparotomy for trauma, more specifically pneumonia (30%). This data support that infection diseases, especially pneumonia, are a common complication after splenectomy, probably due to immunosupression.
Splenectomy is recognized as a cause of portal, mesenteric, and splenic vein thrombosis, a potentially life-threatening complication that can occur within months after splenectomy18. The clinical spectrum ranges from undefined and nonspecific symptoms to intestinal infarction and portal hypertension. However, the exact incidence of the complications and its predisposing factors are not clearly defined. The reported rate of thrombosis widely varies depending on the diagnostic method used. In the review performed by Krauth et al,19 the overall risk of all reviewed studies combined was 3.3%. Risk factors were big spleens and hereditary hemolytic anemia, whereas the risk was low in autoimmune thrombocytopenia and trauma. To date, the role of the surgical approach, laparoscopic surgery, or open surgery is also unclear. Whereas some studies have shown no influence of surgical technique on the incidence of portal and splenic vein thrombosis, others have reported a significantly higher incidence of the complication after laparoscopic surgery.11 In our series, no portal/splenic vein thrombosis was detected in the early postoperative period.
Many preoperative factors have been described to predict complications. Age of patients has been reported to be a prognosis factor for morbidity.20 Elderly patients with hematological disorders requiring splenectomy are in a special group, in which the underlying disease by itself is often supposed to be associated with a higher surgical morbidity. In our study, we found that elderly patients had significantly more complications, as is described in other studies.7 ; 8
Regarding splenomegaly, the literature presents no unanimous use of this term. The normal spleen size is up to 12 cm, and anything larger than that is classified as splenomegaly, which could be moderate, within a range of 11–20 cm, or severe, more than 20 cm. However, we considered splenomegaly as a long axis exceeding 15 cm, according to the EAES guidelines11. This variable did not correlate with perioperative complications in our series. Several authors have defined splenomegaly in terms of weight. Horowitz et al10 reported a rate of complications of 63% for patients whose spleens after splenectomy for hematologic malignancies weighed greater than 2000 g, and 29% for patients whose spleens weighed less than 2000 g. Danforth et al21 reported that the postoperative complication rate after splenectomy from massive splenomegaly (≥ 1500 g) was 39.1%. In both cases, the splenic weight, as significant predictor of postoperative morbidity, was due to greater blood loss during surgery.
In fact, a spleen weight > 2000 g has been reported to stand out as the main determinant of operative blood loss resulting in more complications.10 ; 21 However, our findings are similar to that of Al Harbi et al8 and McAneny et al,22 who showed that the weight of the spleen has no effect on morbidity. Moreover, a definition by weight does not seem appropriate because the weight can be obtained only postoperatively and has no predictive value for the choice of surgical approach.11
Indication for splenectomy23 ; 24 is also a determinant factor for postoperative complications. Malignant diseases have more complications than autoimmune disease, which have less complications because of their underlying medical condition. In our series, patients with malignant hematological disease had more complications, compared with idiopathic thrombocytopenic purpura and hemolytic anemia. In particular, ITP was a predictor factor for fewer complications in univariate analysis. Many reports show that ITP is related to minimal blood loss, short hospital stay, and less negative outcome, especially when it is performed laparoscopically.23
In many centers, laparoscopic splenectomy has become the standard approach for most cases of elective splenectomy, and is associated with reduced morbidity, especially pulmonary, wound, and infectious complications.25 In our series, this approach was also related to fewer complications, similar to results reported by others. Maurus et al26 reported an overall complication rate much higher in open versus laparoscopic splenectomies, including less blood loss. Probably, the fewer complications could be related to there being more selected patients who underwent a laparoscopic approach. We had a relatively high rate of conversion (22.9%), which can be explained by the long time interval of our analysis series, between 1996 and 2010, which includes the learning curve of laparoscopy in our hospital.
We also found that preoperative anemia and leukocytosis were predictive negative factors for morbidity. Anemia is not reported to be a related factor in previous papers about this issue, but patients with hematologic malignancy with leukocytosis are described to have more complications.10
Blood transfusion was the only independent risk factor for morbidity in the multivariate analysis. It has been reported in other previous studies7−10 as one of the main predictors of postoperative complications. Many studies have shown that blood transfusions suppress immune function27 and are related to an increased number of postoperative complications and length of hospital stay.28 This variable was also a significant risk factor for mortality in the univariate analysis, but it was not an independent prognostic factor in multivariate analysis.
In this series, other examined variables were found to have no significant impact on morbidity, such as surgical time and ASA score. This may be because the ASA score is a predictor for postoperative mortality rather than for complications.
Regarding mortality, our series had a rate of 7.2%, which compares well the 6-9% rate reported in the literature.8; 9 ; 10 The direct cause of death in our series was multiple organ failure and sepsis, probably due to immunosupression after splenectomy. This is according to Johnson and Deterling,29 who reported that the direct cause of mortality was sepsis in 36% of patients. However, bleeding was the main cause of death in other series reviewed,8 where many patients had portal hypertension.
Reported predictors factors for mortality in the literature7 ; 8 are age, blood transfusion, thoraco-abdominal approach, operative time, and chest infection. In this study, we found that significant predictors for mortality were age, leukocytosis, ASA score, blood transfusion, pleural effusion, and pneumonia. However, only pneumonia was an independent negative prognostic factor for perioperative death.
The main limitation of this study comes from its retrospective design. Moreover, this study included patients from a long period of time, during which there have been many improvements in surgery, antibiotics, and anesthetics, which could cause fewer rates of complications.
In conclusion, the morbidity and mortality in nontraumatic splenectomies are relatively high, probably due to immunosuppressed patients. Blood loss, as in other types of surgery, such as liver or pancreatic surgery, is the most important prognostic factor for postoperative complications. Infection is the main prognostic factor for perioperative mortality.
References
- 1 P. Kapoor, E. Singh, P. Radhakrishnan, P. Mehta; Splenectomy in plasma cell dyscrasias: a review of the clinical practice; Am J Hematol, 81 (2006), pp. 946–954
- 2 S.C. Katz, H.L. Pachter; Indications for splenectomy; Am Surg, 72 (2006), pp. 565–580
- 3 S.J. Russell, J.D. Richards; Medical indications for splenectomy; Br J Hosp Med, 42 (1989), pp. 120–126 127
- 4 A.M. Carlin, J.G. Tyburski, R.F. Wilson, C. Steffes; Factors affecting the outcome of patients with splenic trauma; Am Surg, 68 (2002), pp. 232–239
- 5 M.A. Malangoni, L.D. Dillon, T.W. Klamer, R.E. Condon; Factors influencing the risk of early and late serious infection in adults after splenectomy for trauma; Surgery, 96 (1984), pp. 775–783
- 6 P. Tornetta III, H. Mostafavi, J. Riina, et al.; Morbidity and mortality in elderly trauma patients; J Trauma, 46 (1999), pp. 702–706
- 7 J. Aksnes, M. Abdelnoor, O. Mathisen; Risk factors associated with mortality and morbidity after elective splenectomy; Eur J Surg, 161 (1995), pp. 253–258
- 8 M. Al Harbi; Predictors for morbidity and mortality following non-traumatic splenectomy at the University Hospital, Jeddah, Saudi Arabia; Int Surg, 85 (2000), pp. 317–321
- 9 J.P. Arnoletti, J. Karam, J. Brodsky; Early postoperative complications of splenectomy for hematologic disease; Am J Clin Oncol, 22 (1999), pp. 114–118
- 10 J. Horowitz, J.L. Smith, T.K. Weber, M.A. Rodriguez-Bigas, N.J. Petrelli; Postoperative complications after splenectomy for hematologic malignancies; Ann Surg, 223 (1996), pp. 290–296
- 11 B. Habermalz, S. Sauerland, G. Decker, et al.; Laparoscopic splenectomy: the clinical practice guidelines of the European Association for Endoscopic Surgery (EAES); Surg Endosc, 22 (2008), pp. 821–848
- 12 B. Misselwitz, E.B. Bachli, P. Kaiser, J. Fehr, J.S. Goede; Diagnosis of hypersplenism with the epinephrine stimulation test - 23 years of experience at a tertiary care hospital; Swiss Med Wkly, 141 (2012), p. w13324
- 13 P.A. Clavien, J. Barkun, M.L. de Oliveira, et al.; The Clavien-Dindo classification of surgical complications: five-year experience; Ann Surg, 250 (2009), pp. 187–196
- 14 D. Dindo, N. Demartines, P.A. Clavien; Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey; Ann Surg, 240 (2004), pp. 205–213
- 15 E.M. Targarona, J.J. Espert, E. Bombuy, et al.; Complications of laparoscopic splenectomy; Arch Surg, 135 (2000), pp. 1137–1140
- 16 A.A. Dawson, P.F. Jones, D.J. King; Splenectomy in the management of haematological disease; Br J Surg, 74 (1987), pp. 353–357
- 17 J. Wiseman, C.V. Brown, J. Weng, A. Salim, P. Rhee, D. Demetriades; Splenectomy for trauma increases the rate of early postoperative infections; Am Surg, 72 (2006), pp. 947–950
- 18 K.M. Stamou, K.G. Toutouzas, P.B. Kekis, et al.; Prospective study of the incidence and risk factors of postsplenectomy thrombosis of the portal, mesenteric, and splenic veins; Arch Surg, 141 (2006), pp. 663–669
- 19 M.T. Krauth, K. Lechner, E.A. Neugebauer, I. Pabinger; The postoperative splenic/portal vein thrombosis after splenectomy and its prevention–an unresolved issue; Haematologica, 93 (2008), pp. 1227–1232
- 20 B. Palsson, M. Hallen, E. Nordenstrom, R. Andersson; Elective splenectomy in the elderly–perioperative and long-term course; Langenbecks Arch Surg, 386 (2001), pp. 339–345
- 21 D.N. Danforth Jr., D.L. Fraker; Splenectomy for the massively enlarged spleen; Am Surg, 57 (1991), pp. 108–113
- 22 D. McAneny, W.W. LaMorte, T.E. Scott, L.R. Weintraub, R.M. Beazley; Is splenectomy more dangerous for massive spleens?; Am J Surg, 175 (1998), pp. 102–107
- 23 M. Rosen, F. Brody, R.M. Walsh, M. Tarnoff, J. Malm, J. Ponsky; Outcome of laparoscopic splenectomy based on hematologic indication; Surg Endosc, 16 (2002), pp. 272–279
- 24 J.M. Ziemski, W.J. Rudowski, W. Jaskowiak, L. Rusiniak, R. Scharf; Evaluation of early postsplenectomy complications; Surg Gynecol Obstet, 165 (1987), pp. 507–514
- 25 L.S. Feldman; Laparoscopic splenectomy: standardized approach; World J Surg, 35 (2011), pp. 1487–1495
- 26 C.F. Maurus, M. Schafer, M.K. Muller, P.A. Clavien, M. Weber; Laparoscopic versus open splenectomy for nontraumatic diseases; World J Surg, 32 (2008), pp. 2444–2449
- 27 M.E. Brunson, J.W. Alexander; Mechanisms of transfusion-induced immunosuppression; Transfusion, 30 (1990), pp. 651–658
- 28 W.F. Bower, L. Jin, M.J. Underwood, Y.H. Lam, P.B. Lai; Peri-operative blood transfusion increases length of hospital stay and number of postoperative complications in non-cardiac surgical patients; Hong Kong Med J, 16 (2010), pp. 116–120
- 29 H.A. Johnson, R.A. Deterling; Massive splenomegaly; Surg Gynecol Obstet, 168 (1989), pp. 131–137
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?