Abstract
Background
We sought to identify prognostic factors of long-term mortality, specific for the underlying etiology of chronic systolic heart failure (CHF).
Methods and results
Between 1995 and 2009 baseline characteristics, treatment and follow-up data from 2318 CHF-patients due to ischemic (ICM; 1100 patients) or dilated cardiomyopathy (DCM; 1218 patients) were prospectively compared. To calculate hazard ratios with 95%-confidence intervals cox regression was used. We respectively established etiology-specific multivariable models of independent prognostic factors. During the follow-up period of up to 14.8 years (mean = 53.1 ± 43.5 months; 10,264 patient-years) 991 deaths (42.8%) occurred.
In the ICM-cohort, 5-year-survival was 53.4% (95% CI: 49.9–56.7%), whereas in DCM-patients it was higher (68.1% (95% CI: 65.1–71.0%)). Age, ejection fraction, or hyponatremia were independent predictors for mortality in both cohorts, whereas diabetes, COPD, atrial fibrillation and a heart rate of ≥ 80/min carried independent predictive power only in ICM-patients.
Conclusion
This study demonstrates the disparity of prognostic value of clinically derived risk factors between the two main causes of CHF. The effects of covariables in DCM-patients were lower, suggesting a less modifiable disease through risk factors considering mortality risk. An etiology-specific prognostic model may improve accuracy of survival estimations in CHF.
Abbreviations
ACE-I, Angiotensin-converting enzyme inhibitor;ARB, Angiotensin receptor blocker;BBL, Beta-blockers;CHF, Chronic systolic heart failure;CRT, Cardiac resynchronization therapy defibrillator;DCM, Dilated cardiomyopathy;EF, Left ventricular ejection fraction;HTX, Orthotopic heart transplantation;ICD, Implantable cardioverter-defibrillator;ICM, Ischemic cardiomyopathy;LBBB, Left bundle branch block;NT-proBNP, N-terminal pro-peptide of brain natriuretic peptide;NYHA, New York Heart Association
Keywords
Heart failure;Dilated cardiomyopathy;Ischemic cardiomyopathy;Prognosis
1. Introduction
During the past decade, many efforts have been made to generate reliable prognostic tools for patients suffering from chronic systolic heart failure (CHF) in order to identify patients who, in spite of adequate outpatient heart failure care and guideline adherent pharmacotherapy, are at high risk of rapid deterioration to a more severe stage of the disease and eventually to an earlier death. Although the clinical phenotype of systolic dysfunction and left ventricular dilation is shared by dilated cardiomyopathy (DCM) and ischemic cardiomyopathy (ICM), pathogenesis and genetic causes of these leading CHF etiologies evidently differ. Pre-clinical and clinical findings have pointed towards cause-related specific risks in CHF [1]; [2]; [3]; [4]; [5]; [6] ; [7], yet to date most contemporary risk prediction algorithms used for CHF prognostication derive from large data sets of general CHF populations [8]; [9] ; [10] and therefore fail to differentiate between DCM and ICM. Emerging concepts of risk prediction incorporate device therapy, novel biomarkers or common genetic variants into traditional prediction models of mortality in CHF and by this may further optimize risk prediction [11]; [12]; [13]; [14]; [15] ; [16]. Yet it is likewise conceivable that simple knowledge of influential differences of clinical indicators according to the sub-phenotype of CHF has the potential to increase the accuracy and robustness of prognostic quantification today and thus, set a higher performance benchmark for future risk models. We sought to improve prediction of long-term survival in patients suffering from CHF by identifying etiology-specific prognostic factors routinely available to clinicians.
2. Methods
2.1. Study setting and sample
Both hospitals included in our analysis (secondary hospital: Städtisches Klinikum, Ludwigshafen and tertiary hospital: University Hospital, Heidelberg) are located in the south-west of Germany and offer heart failure treatment for patients from the nearby community as well as patients referred from other sites. Data from the participating clinics was entered into a common registry in a prospective manner. This registry, the ‘HELUMA heart failure registry’ represents a multi-site cooperation between the specialized heart failure clinics of the University Hospital Heidelberg, the Klinikum Ludwigshafen and the TKH Mannheim, Germany. As the participating centers serve as major outpatient clinics for a region of approximately 900.000 inhabitants a broad representation of patients in a “real life” setting is ensured. In the present study, we included ambulatory patients with CHF due to ICM or DCM at the institutions mentioned above, who were diagnosed over a period of 14 years (1995 until 31 January 2009). Informed consent was obtained from each patient involved. Patients with acute coronary syndrome during initial evaluation, asymptomatic patients (NYHA functional class I), left ventricular ejection fraction (EF) of > 40% in echocardiographic measurement, those in whom in-hospital death occurred or patients with other etiologies of CHF such as heart failure secondary to valvular, hypertensive or a primary pulmonary disease were excluded from the present analysis. Included patients were followed up regularly during ambulatory examinations which took place at least once a year, as well as during every hospitalization in a participating center.
2.2. Data collection
Information about patient characteristics, cardiac history, current investigations (electrocardiography, echocardiography and others), laboratory measurements and chronic medication was collected during the index visit. The underlying cause of HF was categorized as ICM on the basis of a history of MI or the findings in coronary angiography such as 1-, 2-, 3-vessel CHD except arteriosclerosis without relevant stenosis or as DCM defined according to the 1995 WHO definition of cardiomyopathies [17]. DCM is well known to have various underlying causes such as genetic, infectious, autoimmune, and toxic predispositions and diseases, which finally end in the common pathway of ventricular dilatation and systolic dysfunction [18]. We defined DCM as a disease of genetic and/or idiopathic origin and excluded underlying infectious, autoimmune and/or toxic causes.
Venous blood samples were taken in the morning after overnight fasting. N-terminal pro-peptide of brain natriuretic peptide (NT-proBNP) was measured according to the standard protocol of the fully automated Elecsys® Roche Diagnostics analyzer at the tertiary center of Heidelberg since 1995 and at the secondary center of Ludwigshafen since 1999. Anemia was defined according to the WHO thresholds for hemoglobin used to define anemia in adult patients (12.0 g/dl in non-pregnant women; 13.0 g/dl in men) [19]. The left ventricular internal diameter was measured at end diastole (LVEDD) in echocardiography. Left ventricular systolic function was determined by the EF through one of the following methods: the biplane disc summation method (Simpson rule) in echocardiography or by quantitative measurement or qualitative estimation in contrast ventriculography during cardiac catheterization. For functional classification we used the well-known NYHA classification. Atrial fibrillation and left branch bundle block (LBBB) were diagnosed by using a 12-lead-electrocardiogram (ECG) with respect to the guidelines in ECG diagnostics. Patients were followed prospectively for a generally unlimited time period. Follow-up information on vital status, clinical events and medical treatment was obtained using standardized case report forms during re-examinations or by telephone calls at least on a yearly basis. No patient identification was recorded on the case report forms or transferred to the analysis center. Instead, the hospitals were instructed to keep a log of all included patients, in which their names, contact information, and study code were recorded, in order to enable follow-up. The study was performed according to the ethical standards of the Declaration of Helsinki.
2.3. Statistical analysis
Patient characteristics are described by percentages and compared by Chi2/Fisher exact test. The distributions of metrical variables are presented as median and quartiles, metrical or ordinal variables compared by Mann–Whitney–Wilcoxon U-Test. Death from any cause constituted the primary endpoint of the study. Density curves for age were smoothed using triangular kernels. Patients receiving orthotopic heart transplantation (HTX) were followed until their surgical procedure and then censored. Survival curves were created using the Kaplan–Meier method to present overall mortality and the composite of mortality and HTX stratified by disease etiology.
The prognostic value of single clinical variables was assessed using Cox regression by calculating adjusted hazard ratios (HR) with 95%-confidence intervals (95% CI), using the data from the total observation time. The effects were estimated for the both groups of ICM and DCM patients, and the difference between these groups was assessed with the Wald test for an interaction term in the model. Four basic predictors—age (linear for patients ≥ 50 years), gender, NYHA III/IV and LVEF ≤ 30%—were adjusted for center of patient inclusion and for inclusion period (2001–2009 vs. 1995–2000) and their interaction. For the further potential predictors of mortality, effects were adjusted for these basic predictors as well as center and inclusion period.
In order to assess the discriminatory ability of prediction models including all relevant available clinical variables by ROC curves and C statistics etiology-specific multivariable Cox regression models were developed. The same variables were included for both etiologies, and only variables with p > 0.15 in both models were removed. Three variables, QRS duration, anemia and NTproBNP were not considered for this multivariable analysis due to the incomplete composition of data sets in the overall cohort, specifically between the years 1995 and 1999, and means were imputed for missing values of the other covariates. All effects were adjusted for center and inclusion period, so we used a “minimal model” including only center and inclusion period as factors for comparison purposes. In this setting, ROC curves depend on the time for which the prediction is made, and they were calculated for 5 years after index discharge using the method of Heagerty et al. [20]. Estimates of the C-statistics and their difference between the full models and the minimal models together with their standard errors were calculated using the method of Uno et al. [21]. All tests were two-tailed and a p-value of less than 5% was regarded statistically significant. Statistical analysis was performed with SAS version 9.2 (Cary, NC, USA).
3. Results
3.1. Characteristics and pharmacotherapy stratified by etiology of CHF
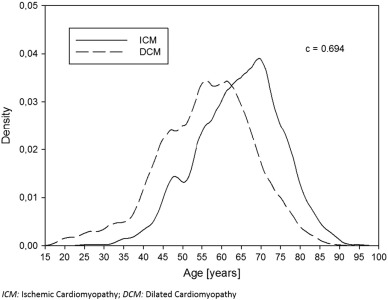
Of all 2318 patients who fulfilled the inclusion criteria, those suffering from ICM (47.5%) presented in a more severe NYHA clinical stage and more often carried well known risk factors commonly associated with CAD: Diabetes, hyperlipidemia, arterial hypertension and smoking. These patients also more likely had renal insufficiency in comparison to the group of patients with DCM (15.5% versus 9.8%, respectively, p < 0.0001). A comparison of age distribution between the two groups is presented in Fig. 1. Patients in whom DCM was the underlying cause of heart failure (52.5%), were significantly younger (median age 56.8 years in patients with DCM versus 65.3 years in those with ICM, p-value < 0.0001) and were more likely female (24.6% in DCM versus 18.1% in ICM, p < 0.001). Further, those with DCM were more likely to have chronic obstructive pulmonary disease (COPD), atrial fibrillation and LBBB. These patients more frequently presented with more severely impaired ejection fractions measured in echocardiography, yet with fewer limitations in functional capacity assessed by a 6-min walk test. Patient demographics and clinical characteristics are described in more detail in Table 1. Regarding pharmacotherapy according to heart failure guidelines, high percentages of beta-blockers (BBL), angiotensin-converting enzyme inhibitors (ACE-I) and angiotensin II receptor blockers (ARB) prescribed at discharge were found throughout both cohorts. Yet in direct comparison, the proportion of DCM patients receiving ACE-I/ARB, aldosterone antagonists, digitalis or oral anticoagulation was significantly higher, whereas the proportion of patients with ICM who received aspirin and statin therapy was expectably larger than in the cohort of patients with DCM (Table 2).
|
|
|
Fig. 1. Smoothed density curves of the age distribution of patients according to the underlying etiology of heart failure. The C-statistic estimates the probability that a patient with ICM is older than a randomly chosen patient with DCM (c = 0.694). |
| ICM (n = 1100) | DCM (n = 1218) | p-value | OR (95%-CI) | |
|---|---|---|---|---|
| Secondary center [%] | 47.1% | 29.5 % | < 0.0001 | 2.13 (1.80–2.53) |
| Age [years/IQR] | 65.3 (57.1–71.7) | 56.8 (48.4–64.4) | < 0.0001 | – |
| Female gender [%] | 18.1% | 24.6 % | < 0.001 | 0.68 (0.55–0.83) |
| Functional class [%] | < 0.0001* | |||
| NYHA class II | 37.6% | 45.0% | – | |
| NYHA class III | 50.2% | 46.9% | – | |
| NYHA class IV | 12.1% | 8.1% | – | |
| Severely impaired LVF (EF ≤ 30%) [%] | 67.1% | 76.1% | < 0.0001 | 0.64 (0.53–0.77) |
| Moderate/severe mitral regurgitation [%] | 25.7 % | 30.1 % | < 0.05 | 0.80 (0.65-0.98) |
| Heart rate [bpm] (IQR) | 75 (65–87) | 80 (70–95) | < 0.0001 | – |
| Atrial fibrillation [%] | 15.8% | 21.9% | < 0.001 | 0.67 (0.54–0.84) |
| LBBB [%] | 26.2% | 34.4% | < 0.0001 | 0.68 (0.56–0.82) |
| Diabetes [%] | 31.0 % | 17.7 % | < 0.0001 | 2.09 (1.72 -2.55) |
| COPD [%] | 28.4 % | 36.4 % | < 0.0001 | 0.70 (0.58–0.83) |
| Hypercholesterolemia [%] | 58.7 % | 27.7% | < 0.0001 | 3.72 (3.10–4.45) |
| BMI | 26.4 (24.1–29.0) | 26.4 (23.7–29.6) | n.s. | – |
| Sodium [mmol/l] | 139 (137–141) | 139 (137–141) | < 0.05 | – |
| NT-proBNP [ng/l] | 1781 (735–4387) | 1611 (627–3750) | n.s. | – |
| Creatinine > 1.5 mg/dl [%] | 15.5 % | 9.8 % | < 0.001 | 1.69 (1.29–2.22) |
| Hemoglobine [g/dl] | 13.7 (12.3–14.9) | 14.1 (12.9–15.2) | < 0.0001 | – |
| ICD/CRT [%] | 24.0% | 15.1% | < 0.001 | 1.77 (1.44–2.19) |
| Six-minute walk test [m] | 428 (344–493) | 464 (378–528) | < 0.0001 | – |
ICM: ischemic cardiomyopathy; DCM: dilated cardiomyopathy; OR: odds ratio; IQR: interquartile range; NYHA: New York Heart Association; LVF: systolic left ventricular function; EF: left ventricular ejection fraction; bpm: beats per minute; LBBB: left bundle branch block; ICD: implantable cardioverter-defibrillator; CRT: cardiac resynchronization therapy defibrillator; BMI: body mass index; COPD: chronic obstructive pulmonal disease; *NYHA: III-IV vs. II, II-III vs. IV.
| ICM (n = 1100) | DCM (n = 1218) | p-value | OR (95%-CI) | |
|---|---|---|---|---|
| Medication at discharge | ||||
| ACE-I or ARB | 94.3% | 96.6% | < 0.01 | 0.58 (0.39–0.87) |
| BBL | 68.4% | 67.0% | n.s. | 1.07 (0.89–1.27) |
| Aldosterone antagonists | 35.0% | 42.8% | < 0.001 | 0.72 (0.61–0.85) |
| Loop diuretics | 70.8% | 70.7% | n.s. | 1.01 (0.84–1.21) |
| Digitalis | 52.2% | 65.1% | < 0.0001 | 0.58 (0.49–0.69) |
| Aspirin | 41.4% | 14.4% | < 0.0001 | 4.19 (3.43–5.13) |
| Anticoagulant therapy | 53.4% | 65.4% | < 0.0001 | 0.61 (0.51–0.72) |
| Statins | 65.7% | 25.1% | < 0.0001 | 5.70 (4.76–6.83) |
ICM: ischemic cardiomyopathy; DCM: dilated cardiomyopathy; ACE-I: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; BBL: beta-blockers.
3.2. Mortality and need for heart transplantation
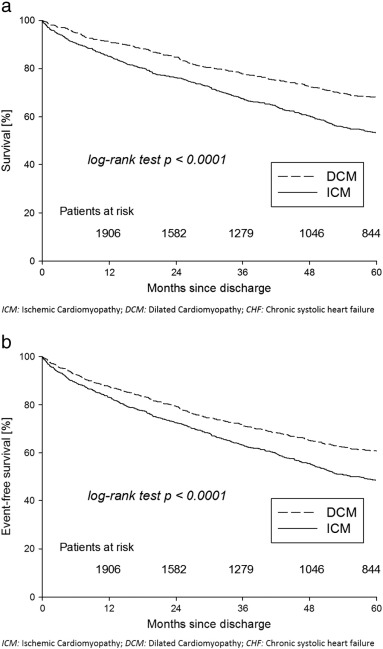
During the follow-up period of up to 14.8 years (mean = 53.1, SD = 43.5 months; 10,264 patient-years) 991 (42.8%) patients died and 197 (8.5%) patients received HTX. The median interval between index visit and HTX was 19.6 months (IQR = 7–40 months). Patient status at 5 years was available for 949/1100 (86.3%) patients of the ICM cohort (425 deaths, 67 HTX, 457 survivors) and 1079/1218 (88.6%) patients with DCM (322 deaths, 104 HTX, 653 survivors) who entered the study. Kaplan–Meier survival curves illustrating 5-year survival stratified by the underlying etiology of CHF are shown in Fig. 2a. In the group of patients suffering from ICM, unadjusted survival rates were 85.1% (95% CI: 82.8–87.1%) at 1 year, 67.5% (95% CI: 64.4–70.4%) at 3 years and 53.4% (95% CI: 49.9–56.7%) at 5 years. Overall survival was higher in the group of patients suffering from DCM (log rank test p < 0.0001). Unadjusted survival rates in this patient cohort were 91.2% (95% CI: 89.4–92.7%) at 1 year, 77.8% (95% CI: 75.2–80.2%) at 3 years and 68.1% (95% CI: 65.1–71.0%) at 5 years, respectively. Accordingly, for the composite of mortality and HTX, crude event-free survival differed by the underlying etiology of CHF and was notably higher in the group of patients with DCM (log rank test p < 0.0001, Fig. 2b).
|
|
|
Fig. 2. Kaplan–Meier survival curves: 5 year follow up. a: Endpoint mortality. Survival curves not adjusted. HTX censored. Kaplan–Meier curve of 5-year survival was significantly lower among patients with ICM compared to those with DCM as the underlying cause of CHF (log rank test p < 0.0001). b: Endpoint mortality or heart transplantation. Survival curves not adjusted. Five-year event-free survival was significantly lower among patients with ICM compared to those with DCM as the underlying cause of CHF (log rank test p < 0.0001). |
3.3. Multivariable predictors of mortality
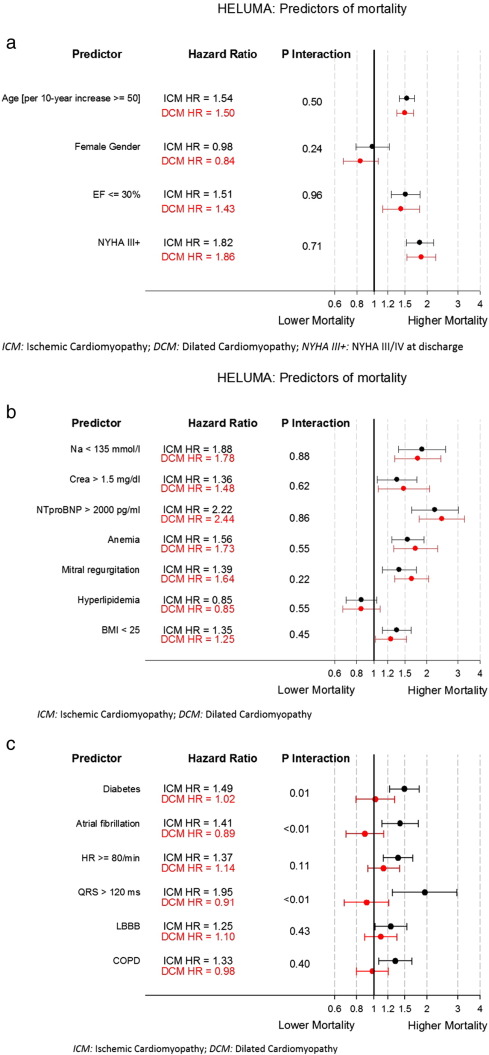
Clinical characteristics associated with mortality are presented in Fig. 3a–c. All available demographic information and baseline clinical characteristics that potentially affected survival were analyzed. After adjustment, advanced age, EF ≤ 30%, NYHA functional class III/IV, BMI < 25, renal insufficiency, hyponatremia, moderate/severe mitral regurgitation, anaemia (WHO definition) and NTproBNP values > 2000 pg/ml were found to be associated with shorter survival time in both patient cohorts. In contrary, among all patients, time to death was not significantly prolonged in females and in those with hyperlipidemia (Fig. 3a, b). Further, diabetes, atrial fibrillation, heart rate ≥ 80/min, a wide QRS complex (> 120 ms), LBBB and COPD failed to carry predictive power in patients with DCM (Fig. 3c). Among all variables investigated, an NTproBNP value of > 2000 pg/ml was the most strongly associated predictor of reduced survival in both groups followed by hyponatremia and NYHA functional class III/IV. In multivariable analysis, all the above mentioned clinical variables except for hyperlipidemia proved to be independently associated with worse survival in patients suffering from ICM (Table 3). Yet when assessing patients with DCM in a multivariable model, diabetes, COPD, LBBB, atrial fibrillation and increased resting heart rate of ≥ 80 bpm failed to carry independent predictive power. Of the characteristics which correlated with improved survival, female gender and hyperlipidemia did not attain statistical significance.
|
|
|
Fig. 3. Predictors of mortality. a: Hazard ratios of single predictors for all-cause mortality by etiology, adjusted for center and inclusion period (1995–2000 vs. 2001–2008). b: Hazard ratios of single predictors for all-cause mortality by etiology, adjusted for age (linear > 50 years), gender, LVEF, NYHA class, center and inclusion period. c: Hazard ratios of single predictors for all-cause mortality by etiology, adjusted for age (linear > 50 years), gender, LVEF, NYHA class, center and inclusion period. |
| Variable | Multivariable HR (95%-CI) | |
|---|---|---|
| ICM | DCM | |
| Age [per 10 years above 50] | 1.46 (1.30–1.63) | 1.49 (1.31–1.68) |
| Female gender | 0.97 (0.76–1.24) | 0.80 (0.63–1.02) |
| EF ≤ 30% | 1.24 (1.01–1.52) | 1.33 (1.03–1.71) |
| NYHA III/IV at discharge | 1.49 (1.23–1.80) | 1.61 (1.32–1.97) |
| BMI < 25 | 1.32 (1.09–1.60) | 1.22 (0.99–1.49) |
| Diabetes | 1.45 (1.19–1.76) | 1.12 (0.87–1.44) |
| Hypercholesterolemia | 1 | 1 |
| Creatinine > 1.5 mg/dl | 1.36 (1.05–1.76) | 1.53 (1.09–2.15) |
| COPD | 1.29 (1.04–1.60) | 1.02 (0.82–1.26) |
| Sodium < 135 mmol/l | 1.73 (1.27–2.35) | 1.65 (1.22–2.24) |
| Heart rate ≥ 80 / min | 1.29 (1.06–1.57) | 1.12 (0.91–1.38) |
| Atrial fibrillation | 1.41 (1.11–1.78) | 0.89 (0.69–1.14) |
| Moderate / severe mitral regurgitation | 1.28 (1.02–1.60) | 1.52 (1.21–1.90) |
| LBBB | 1.22 (0.99–1.50) | 1.04 (0.85–1.26) |
HR: hazard ratio; other abbreviations as in Table 1.
The effect estimates are adjusted for center and inclusion period. Variables removed by the selection are marked with a hazard ratio of 1.
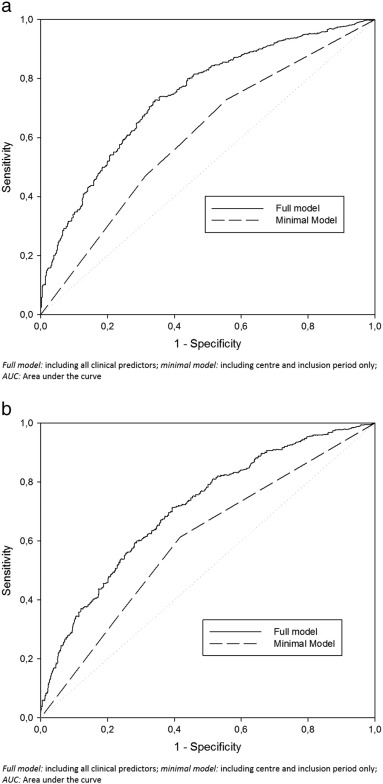
The ROC curves of the multivariable models as predictors for 5-year mortality are shown in Fig. 4a and b. The C-statistic for ICM is 0.70 and its difference from the corresponding minimal model including only center and inclusion period as factors is 0.12 (95%-CI 0.09–0.14). For the DCM group, the C statistic is 0.67 and its difference from the minimal model is 0.09 (95%-CI 0.06–0.12), which falls short of reaching a statistically significant difference from ICM (p = 0.17).
|
|
|
Fig. 4. Receiver operating characteristic curves for multivariable models as predictors of 5-year mortality according to the underlying etiology of heart failure. a: Receiver operating characteristic curves at 5 years post discharge for multivariable models as predictors of 5-year mortality in ICM patients. b: Receiver operating characteristic curves at 5 years post discharge for multivariable models as predictors of 5-year mortality in DCM patients. |
4. Discussion
In this present analysis we report differences in clinical predictors of mortality in a cohort of 2318 ambulatory CHF patients who were stratified according to the underlying cause of systolic CHF. The main finding of our study is that in CHF patients seen in daily clinical practice, with impaired systolic function under individually optimized pharmacotherapy, a comparably limited number of clinically-derived parameters are available to clinicians for prognostication of survival when DCM is the underlying etiology of the disease.
Interestingly, unlike in many other diseases, most clinical trials in CHF have investigated generalized populations of systolic dysfunction without incorporating potential effects of primary pathophysiological processes behind the syndrome. Yet with sufficient knowledge of preconditional heterogeneity of this systolic phenotype of CHF and with numerous aspects of personalized medicine (genomic, proteomic and metabolomic scores) on the horizon, it seems almost inappropriate to guide treatment in CHF without adequate accommodation of a decisive factor such as etiology.
Some previous trials have performed etiology-oriented analysis of long-term survival in CHF. Whereas the majority of these studies showed a clear benefit of non-ischemic origin of CHF [22]; [23]; [24] ; [25], Studies of Left Ventricular Dysfunction (SOLVD) and a large population-based study failed to demonstrate a negative influence of ischemic heart disease or previous myocardial infarction on survival [26] ; [27]. Reasons for these conflicting results are presumably diverse. They may partially lie in the incoherent composition of inclusion criteria in these trials or in the diagnostic accuracy achieved by the chosen imaging modality. Further, as the majority of trials which addressed the effect of disease pathogenesis on mortality were performed in the early 1990s, the percentage of patients who received BBL at the time of investigation ranged broadly between the respective studies. Consequentially, to enable comparisons of our present cohort study with results of these prior analyses including the observed mortality rates, the evolution in CHF treatment over the last fourteen years needed consideration. We therefore adjusted our regression analysis for the center in which the patient was treated (tertiary versus secondary center) and for the inclusion period. By this, we attempted to manage previously reported interactions of institutional settings and treatment patterns over time [28]; [29]; [30]; [31] ; [32]. To date, Frazier et al. have performed the largest available meta-analysis (11,719 patients) which focused on phenotypical issues of etiology and gender in CHF [25]. The authors showed that non-ischemic etiology was associated with longer overall survival and with longer time to the composite event of hospitalization or death. As mentioned earlier, data for the combined analysis by Frazier et al. were pooled from five randomized clinical trials, each of which evaluated the benefit of a specific pharmacotherapy in CHF and subsequently were subject to specific restrictive inclusion criteria. Thus, although their results may be revelatory by providing insights on a large number of patients with a maximum follow up period of 901 days, they may not broadly represent CHF patients under optimized pharmaco- and device therapy in the “real life” setting of outpatient care. To eliminate these uncertainties, we addressed the question on the level of daily clinical practice and searched for influential factors of reduced survival time and time to HTX in a multi-site registry with patients followed up to almost 15 years. As patients were cared for at specialized heart failure clinics over a period of many years, intense outpatient care including individually optimized pharmacotherapy were ensured. In regards to index clinical and demographic variables, the observations that patients with ICM were older at the time of diagnosis (on average 8 years older that in the group of patients with DCM) and that they presented with more severe clinical symptoms are of particular importance. Further, specific for patients in whom DCM was the underlying cause of CHF, were the findings that the proportion of women was higher and that although these patients more likely had concomitant COPD and a more severely impaired ejection fraction, they were less symptomatic and had higher exercise capacity. Concerning the notably prolonged survival time in patients with DCM in comparison to those with ICM, we confirmed the findings of Frazier et al. and of the majority of previous smaller trials which had all been performed before BBL and ACE-I/ARB were fully adapted in to clinical practice.
In conformity with previous trials which studied clinical risk factors in CHF, well-known associations with impaired outcome in CHF such as advanced age, EF ≤ 30%, NYHA functional class III/IV, renal insufficiency and hyponatremia sustained in our multivariable analysis of both sub-phenotypes. Other variables were associated with shorter survival: BMI < 25 kg/m2 and moderate/severe mitral regurgitation or with prolonged survival: female gender. However, it must be acknowledged that the 95%-CIs of these variables crossed unity and therefore they may have less clinical impact. We also discovered a consistent pattern between the two etiologies in regards to NTproBNP being the most strongly associated parameter followed by hyponatremia and NYHA functional class III/IV the second most strongly associated predictors of reduced survival. Diabetes, COPD, an increased resting heart rate of > 80 bpm, a wide QRS complex (> 120 ms)—LBBB in specific and atrial fibrillation failed to carry predictive power in patients with DCM in multivariable assessment. Interestingly, raised resting heart rate, an influential co-variable in heart failure which has attracted much notice in recent years [33]; [34]; [35] ; [36], was among the parameters which were independently associated with shorter survival time exclusively in patients with ICM yet not in those with DCM. Despite uniform electrocardiographical phenotypes, the differences in risk observed in this present cohort emphasize the diverging nature of rhythm disorders in ischemic and non-ischemic cardiac tissue and may partially explain differences in response to Cardiac Resynchronization Therapy (CRT) or antiarrhythmic agents.
It may attract attention that in the present study—in contrast to most of other large etiology based trials [37] ; [38]—the group of DCM patients is larger than the ICM cohort. As HELUMA is inter alia a registry containing data from a large specialized cardiomyopathy center, more than average numbers of DCM patients are included in the registry.
As stated previously, this analysis demonstrates that well known risk factors of CHF, which are commonly used to prognosticate survival of CHF patients in clinical daily routine, are especially conclusive in patients with ICM, whereas they are significantly less precise when used in with DCM patients.
The complexity of the pathophysiology of idiopathic DCM may explain the poorer performance of classical risk factors in comparison to ICM. Particular attention should be paid to atrial fibrillation and wide QRS complex (> 120 ms) as these risk factors were significant prognostic factor of long-term survival in ICM in our study, yet had no prognostic value in patients with DCM. In this context the lack of predictive significance of atrial fibrillation and wide QRS complex in DCM may be associated with their pre-existence without relevant LV dysfunction in DCM, whereas in ICM, atrial fibrillation and wide QRS complex most likely result from a previous ACS event [39] ; [40]. Interactions between genetic and non-genetic factors play a major role in DCM and in other heritable cardiomyopathies [41]; [42]; [43] ; [44]. Even in cases of presumed non-genetic DCM recent research points to underlying genetic predispositions [45] ; [46]. As of now, further insight is needed until we can fully understand how identified genetic variations and their modifiers translate into patients' phenotypic signatures.
Although the HELUMA heart failure registry includes a large amount of phenotypic information, we cannot account for additional clinical risk factors which were not evaluated in our registry yet possibly contribute to the differences found in our analysis. Our data provide new insights into etiology-specific differences of the systolic sub-phenotype of CHF in terms of the likelihood of rapid disease progression and mortality. With greater understanding of the syndromic heterogeneity of CHF and of its subphenotype-related variances in survival, more appropriate risk discrimination models for guidance in treatment and more refined therapeutic solutions to achieve delayed progression of systolic dysfunction may soon emerge for use in everyday clinical practice.
4.1. Limitations
Although data from the participating clinics were entered in a prospective manner, this present study is subject to the characteristic shortcomings of registries. Nevertheless, its clinical practise-based approach over a period of over 14 years also implies one of the main values of this present study. The all-comer population of CHF patients allows for long-term analysis of those, who for various reasons, would have been excluded by contemporary study protocols.
As this analysis is based on data provided by the HELUMA registry, in which solely CHF patients are enrolled, it is difficult to make a statement on a comparison between the two described collectives, ICM and DCM, and a healthy validation cohort. Furthermore, the registry contains two main endpoint entities, death of various reasons and heart transplantation. As there was no possibility to specify reasons for death due to the conception of the registry, we were unable to differentiate the cohorts according to important terms such as cardiac causes, sudden death, pump failure or other reasons. This is a serious limitation, which should be considered in future studies and registries in these patients.
There are some baseline parameters, which indicate a therapy not according to current guidelines, such as ICD/CRT implantation which was highly underrepresented in both collectives (30% in a high-risk cohort). There are multiplex reasons, as there are individual decisions being weighed for every single patient, such as common operability, especially because of limiting diseases, age, nutritional condition, etc. Finally, the evidence based need for ICD/CRT therapy may not have been appropriate for the individual patient and therefore—after carefully considering every single aspect—was not carried out.
An additional negative aspect of our longer term approach is the fact that we were unable to include blood levels of NTproBNP in patients who were treated at the secondary hospital before the year 1999 and results of 6-min walk tests into our multivariable model due the skewed availability of these data over the entire inclusion period. Besides this, the prognostic value of risk factors we presented may differ in female patients. Only 21.5% of the overall cohort was women and thus, sex-specific prognostic testing was not possible. Further, though outpatient care offered by the participating hospitals is offered to all patients, patient selection may be biased by referring physicians, who may have considered only certain patients in need of more intense care through specialized heart failure clinics. Since the participating hospitals are located in geographically close communities of Germany, the majority of included patients were mainly of white Caucasian origin. The results obtained from our study may therefore not be transferable across all ethnicities or be equally applied to all racial backgrounds. In this present analysis we did not evaluate the exact dosages of guideline adherent pharmacotherapy or censor patients who received an implantable cardioverter-defibrillator (ICD) or Cardiac Resynchronization Therapy (CRT). As these factors have shown to provide substantial advantages in survival, a general limitation may exist regarding the overall outcome. Lastly, as of this writing, the etiology-oriented predictors of mortality detected in our study have not been validated in other independent CHF populations. Only by such measures can we definitely confirm the generalizability of the derived prediction models.
4.2. Conclusion
This study demonstrates the marked disparity between the two main etiologies of CHF with regards to the prognostic value of clinically derived risk factors. Independent predictors for mortality were risk factors well known to be associated with reduced survival time in patients with CHF in general. However in multivariable analysis, the overall number of contemporary clinical variables associated with mortality in patients with DCM was lower, implicating a primary pathophysiology less modifiable through renowned risk factors. It may be assumed that traditional and routinely applied models predict long-term survival accurately for patients suffering from ICM. Although these models are transferred unaltered to DCM patients, there is no evidence of their precision in predicting mortality for this underlying etiology. In the future, the need to create separate predictive models for the various causes of CHF may occur to ensure reliable assessment of long-term survival for each etiology. The results of this study reveal the benefit of an etiology-specific multivariate prognostic model and improve accuracy of survival estimations in CHF.
Furthermore, there is a chance that based on the development of adjusted risk factor models, new pathways in treatment of CHF will open up.
Conflict of interest statement
The authors report no relationships that could be construed as a conflict of interest.
Acknowledgments
J. F. was supported by the Rahel Goitein-Straus Research Program of the Medical Faculty Heidelberg.
This project was supported by an unrestricted grant from the foundation Stiftung Institut für Herzinfarktforschung Ludwigshafen.
We acknowledge financial support by Deutsche Forschungsgemeinschaft and Ruprecht-Karls-Universität Heidelberg within the funding programme Open Access Publishing.
References
- [1] G.W. Dorn II, S.B. Liggett; Mechanisms of pharmacogenomic effects of genetic variation within the cardiac adrenergic network in heart failure; Mol Pharmacol, 76 (3) (Sep 2009), pp. 466–480
- [2] T.D. Karamitsos, J.M. Francis, S. Myerson, J.B. Selvanayagam, S. Neubauer; The role of cardiovascular magnetic resonance imaging in heart failure; J Am Coll Cardiol, 54 (15) (Oct 6 2009), pp. 1407–1424
- [3] A. Senthilkumar, M.D. Majmudar, C. Shenoy, H.W. Kim, R.J. Kim; Identifying the etiology: a systematic approach using delayed-enhancement cardiovascular magnetic resonance; Heart Fail Clin, 5 (3) (Jul 2009), pp. 349–367 [vi]
- [4] K.F. Adams Jr.; Pathophysiologic role of the renin-angiotensin-aldosterone and sympathetic nervous systems in heart failure; Am J Health Syst Pharm, 61 (Suppl. 2) (May 1 2004), pp. S4–S13
- [5] H.C. Volz, C. Seidel, D. Laohachewin, Z. Kaya, O.J. Müller, S.T. Pleger, et al.; HMGB1: the missing link between diabetes mellitus and heart failure; Basic Res Cardiol, 105 (6) (Nov 2010), pp. 805–820
- [6] M.J. Cox, H.S. Sood, M.J. Hunt, D. Chandler, J.R. Henegar, G.M. Aru, et al.; Apoptosis in the left ventricle of chronic volume overload causes endocardial endothelial dysfunction in rats; Am J Physiol Heart Circ Physiol, 282 (4) (Apr 2002), pp. H1197–H1205
- [7] A. Aleshin, R. Ananthakrishnan, Q. Li, R Rosario, Y. Lu, W. Qu, et al.; RAGE modulates myocardial injury consequent to LAD infarction via impact on JNK and STAT signaling in a murine model; Am J Physiol Heart Circ Physiol, 294 (4) (Apr 2008), pp. H1823–H1832
- [8] R. Vazquez, A. Bayes-Genis, I. Cygankiewicz, D. Pascual-Figal, L. Grigorian-Shamagian, R Pavon, et al.; The MUSIC Risk score: a simple method for predicting mortality in ambulatory patients with chronic heart failure; Eur Heart J, 30 (9) (May 2009), pp. 1088–1096
- [9] C. Zugck, C. Krüger, R. Kell, S Körber, D. Schellberg, W. Kübler, et al.; Risk stratification in middle-aged patients with congestive heart failure: prospective comparison of the Heart Failure Survival Score (HFSS) and a simplified two-variable model; Eur J Heart Fail, 3 (5) (Oct 2001), pp. 577–585
- [10] K.D. Aaronson, J.S. Schwartz, T.M. Chen, K.L. Wong, J.E. Goin, D.M. Mancini; Development and prospective validation of a clinical index to predict survival in ambulatory patients referred for cardiac transplant evaluation; Circulation, 95 (12) (Jun 17 1997), pp. 2660–2667
- [11] B.F. Voight, G.M. Peloso, M. Orho-Melander, R Frikke-Schmidt, M. Barbalic, M.K. Jensen, et al.; Plasma HDL cholesterol and risk of myocardial infarction: a Mendelian randomisation study; Lancet, 380 (9841) (2012 Aug 11), pp. 572–580
- [12] H. Schunkert, J. Erdmann, N.J. Samani; Genetics of myocardial infarction: a progress report; Eur Heart J, 31 (8) (Apr 2010), pp. 918–925
- [13] S. Ordu, H. Ozhan, R. Alemdar, M. Aydin, O. Caglar, H. Yuksel, et al.; Carbohydrate antigen-125 and N-terminal pro-brain natriuretic peptide levels: compared in heart-failure prognostication; Tex Heart Inst J, 39 (1) (2012), pp. 30–35
- [14] M. Volpe, P. Francia, G. Tocci, S. Rubattu, S. Cangianiello, M.A. Elena Rao, et al.; Prediction of long-term survival in chronic heart failure by multiple biomarker assessment: a 15-year prospective follow-up study; Clin Cardiol, 33 (11) (Nov 2010), pp. 700–707
- [15] M. Ishino, Y. Takeishi, T. Niizeki, T. Watanabe, J. Nitobe, T. Miyamoto, et al.; Risk stratification of chronic heart failure patients by multiple biomarkers: implications of BNP, H-FABP, and PTX3; Circ J, 72 (11) (Nov 2008), pp. 1800–1805
- [16] W.C. Levy, D. Mozaffarian, D.T. Linker, S.C. Sutradhar, S.D. Anker, A.B. Cropp, et al.; The Seattle Heart Failure Model: prediction of survival in heart failure; Circulation, 113 (11) (Mar 21 2006), pp. 1424–1433
- [17] P. Richardson, W. McKenna, M. Bristow, B. Maisch, B. Mautner, J. O'Connell, et al.; Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies; Circulation, 93 (5) (Mar 1 1996), pp. 841–842
- [18] B.J. Maron, J.A. Towbin, G. Thiene, C. Antzelevitch, D. Corrado, D. Arnett, et al.; Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention; Circulation, 113 (14) (Apr 11 2006), pp. 1807–1816
- [19] World Health Organization Worldwide prevalence of anaemia 1993–2005, 978-92-4-159665-7 (2008)
- [20] P.J. Heagerty, T. Lumley, M.S. Pepe; Time-dependent ROC curves for censored survival data and a diagnostic marker; Biometrics, 56 (2) (Jun 2000), pp. 337–344
- [21] H. Uno, T. Cai, M.J. Pencina, R.B. D'Agostino, L.J. Wei; On the C-statistics for evaluating overall adequacy of risk prediction procedures with censored survival data; Stat Med, 30 (10) (May 10 2011), pp. 1105–1117
- [22] F. Follath; Ischemic vs nonischemic heart failure--does etiology matter?; Ther Umsch, 57 (5) (May 2000), pp. 321–323
- [23] M.S. Nieminen, D. Brutsaert, K. Dickstein, H. Drexler, F. Follath, V.P. Harjola, et al.; EuroHeart Failure Survey II (EHFS II): a survey on hospitalized acute heart failure patients: description of population; Eur Heart J, 27 (22) (Nov 2006), pp. 2725–2736
- [24] C. Lourenço, F. Saraiva, H. Martins, R. Baptista, S. Costa, et al.; Ischemic versus non-ischemic cardiomyopathy–are there differences in prognosis? Experience of an advanced heart failure center; Rev Port Cardiol, 30 (2) (Feb 2011), pp. 181–197
- [25] C.G. Frazier, K.P. Alexander, L.K. Newby, S. Anderson, E Iverson, M. Packer, et al.; Associations of gender and etiology with outcomes in heart failure with systolic dysfunction: a pooled analysis of 5 randomized control trials; J Am Coll Cardiol, 49 (13) (Apr 3 2007), pp. 1450–1458
- [26] C.R. Benedict, G.S. Francis, B. Shelton, D.E. Johnstone, S.H. Kubo, P. Kirlin, et al.; Effect of long-term enalapril therapy on neurohormones in patients with left ventricular dysfunction. SOLVD Investigators; Am J Cardiol, 75 (16) (Jun 1 1995), pp. 1151–1157
- [27] D.S. Lee, P.C. Austin, J.L. Rouleau, P.P. Liu, D. Naimark, J.V. Tu; Predicting mortality among patients hospitalized for heart failure: derivation and validation of a clinical model; JAMA, 290 (19) (Nov 19 2003), pp. 2581–2587
- [28] J. Franke, C. Zugck, J.S. Wolter, L. Frankenstein, M. Hochadel, P. Ehlermann, et al.; A decade of developments in chronic heart failure treatment: a comparison of therapy and outcome in a secondary and tertiary hospital setting; Clin Res Cardiol, 101 (1) (Jan 2012), pp. 1–10
- [29] A.P. Maggioni, U. Dahlström, G. Filippatos, O. Chioncel, M.C. Leiro, J Drozdz, et al.; EURObservational Research Programme: the Heart Failure Pilot Survey (ESC-HF Pilot); Eur J Heart Fail, 12 (10) (Oct 2010), pp. 1076–1084
- [30] M. Komajda, F. Follath, K. Swedberg, J Cleland, J.C. Aguilar, A. Cohen-Solal, et al.; The EuroHeart Failure Survey programme–a survey on the quality of care among patients with heart failure in Europe. Part 2: treatment; Eur Heart J, 24 (5) (Mar 2003), pp. 464–474
- [31] J.G. Cleland, A. Cohen-Solal, J.C. Aguilar, R. Dietz, J. Eastaugh, F. Follath, et al.; Management of heart failure in primary care (the IMPROVEMENT of Heart Failure Programme): an international survey; Lancet, 360 (9346) (Nov 23 2002), pp. 1631–1639
- [32] J.G. Cleland, K. Swedberg, F. Follath, M Komajda, A. Cohen-Solal, J.C. Aguilar, et al.; The EuroHeart Failure survey programme– a survey on the quality of care among patients with heart failure in Europe. Part 1: patient characteristics and diagnosis; Eur Heart J, 24 (5) (Mar 2003), pp. 442–463
- [33] K. Swedberg, M. Komajda, M. Böhm, J.S. Borer, I. Ford, A. Dubost-Brama, et al.; Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study; Lancet, 376 (9744) (Sep 11 2010), pp. 875–885
- [34] M. Böhm, K. Swedberg, M. Komajda, JS Borer, I. Ford, A. Dubost-Brama, et al.; Heart rate as a risk factor in chronic heart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo-controlled trial; Lancet, 376 (9744) (Sep 11 2010), pp. 886–894
- [35] S.M. Kang, H. Won, N. Hong, J. Oh; Ivabradine and outcomes in chronic heart failure; Lancet, 376 (9758) (Dec 18 2010), p. 2069 [author reply 2069–2070]
- [36] J.C. Reil, F. Custodis, K. Swedberg, M. Komajda, J.S. Borer, I. Ford, et al.; Heart rate reduction in cardiovascular disease and therapy; Clin Res Cardiol, 100 (1) (Jan 2011), pp. 11–19
- [37] The CONSENSUS Trial Study Group; Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS); N Engl J Med, 316 (23) (Jun 4 1987), pp. 1429–1435
- [38] M. Komajda, M.C. Wimart, E. Thibout; The ATLAS study (Assessment of Treatment with Lisinopril and Survival); justification and objectives; Arch Mal Coeur Vaiss, 87 (Spec No 2) (Jun 1994), pp. 45–50
- [39] T.J. Wang, M.G. Larson, D. Levy, R.S. Vasan, E.P. Leip, P.A. Wolf, et al.; Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: the Framingham Heart Study; Circulation, 107 (23) (Jun 17 2003), pp. 2920–2925
- [40] D.L. Dries, D.V. Exner, B.J. Gersh, M.J. Domanski, M.A. Waclawiw, L.W. Stevenson; Atrial fibrillation is associated with an increased risk for mortality and heart failure progression in patients with asymptomatic and symptomatic left ventricular systolic dysfunction: a retrospective analysis of the SOLVD trials. Studies of Left Ventricular Dysfunction; J Am Coll Cardiol, 32 (3) (Sep 1998), pp. 695–703
- [41] N. Frey, H.A. Katus; Dilated cardiomyopathy as a genetic disease: molecular and clinical aspects; Internist (Berl), 49 (1) (Jan 2008), pp. 43–50
- [42] S. Waldmüller, J. Erdmann, P. Binner, G. Gelbrich, S. Pankuweit, C. Geier, et al.; Novel correlations between the genotype and the phenotype of hypertrophic and dilated cardiomyopathy: results from the German Competence Network Heart Failure; Eur J Heart Fail, 13 (11) (Nov 2011), pp. 1185–1192
- [43] R.E. Hershberger, J.D. Siegfried; Update 2011: clinical and genetic issues in familial dilated cardiomyopathy; J Am Coll Cardiol, 57 (16) (Apr 19 2011), pp. 1641–1649
- [44] E. Grunig, J.A. Tasman, H. Kucherer, W. Franz, W. Kubler, H.A. Katus; Frequency and phenotypes of familial dilated cardiomyopathy; J Am Coll Cardiol, 31 (1) (Jan 1998), pp. 186–194
- [45] F. Friedrichs, C. Zugck, G.J. Rauch, B. Ivandic, D. Weichenhan, M. Müller-Bardorff, et al.; HBEGF, SRA1, and IK: three cosegregating genes as determinants of cardiomyopathy; Genome Res, 19 (3) (Mar 2009), pp. 395–403
- [46] I. Portig, A. Sandmoeller, S. Kreilinger, B. Maisch; HLA-DQB1* polymorphism and associations with dilated cardiomyopathy, inflammatory dilated cardiomyopathy and myocarditis; Autoimmunity, 42 (1) (Jan 2009), pp. 33–40
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?