Abstract
Hydrogen adsorption in porous nanostructured polyaniline (PANI) has been investigated to determine its potential as a hydrogen storage media. Residual gas analysis and thermogravimetric analysis were used to investigate the cyclical hydrogen adsorption in PANI. A hydrogen storage capacity of up to 3 wt % was obtained by charging the polymers at up to 3.0 × 106 Pa hydrogen at elevated temperature. Multiple charge and discharge cycles have been shown, with good retention beyond 106 sec.
Introduction
Considerable attention has been paid to hydrogen as a future clean fuel for a variety of applications such as fuel cells and clean vehicles, both for its strategic and environmental benefits. Hydrogen storage, however; is a major obstacle and a significant materials science challenge for successful implementation of hydrogen fuel cell technology in transportation applications [1]. Therefore, it is important to find a suitable material in which to store hydrogen safely and efficiently. Hydrogen adsorption/absorption in nanostructured polymers is a possible mechanism to be considered for storing hydrogen. The adsorption/absorption of hydrogen in nanostructured materials depends on a number of variables including the specific surface area, pore structure, pressure and temperature with the expectation that hydrogen uptake will be reversible with fast kinetics. The objective is to store a large amount of hydrogen at near room temperature and at safe pressures.
Various polymeric hydrogen storage media including carbon nanotubes [2-4], polyaniline (PANI) and polypyrrole (PPY) have been proposed as candidate storage systems due to their nanostructures and resultant abundance of physisorption and chemisorption sites [5-7]. However, a number of controversial reports have been made regarding hydrogen uptake in PANI and PPY based on various parameters such as surface morphology, charging temperature, charging duration, H2 pressure, etc. [5, 8, 9]. PANI is an organic conducting polymer whose conductivity can be increased by doping with protonic acids [8-10]. The acid treatment changes the texture of the polymer; which under certain conditions induces the formation of a microstructure with small nanosized pores (30 nm). This may be desirable for adsorption of small molecules like hydrogen [10].
In this work, we examine cyclical hydrogen storage and retention in PANI as a hydrogen storage medium. Adsorption/desorption measurements have been performed for fast charging and discharging of hydrogen. Residual gas analysis (RGA) was used to qualitatively measure the hydrogen stored in the polymers, and hydrogen uptake (wt %) was measured by thermogravimetric analysis (TGA) technique.
Experimental
Sample preparation
Polyaniline samples (Fig. 1) were prepared from conducting PANI powder (PANI emeraldine salt from p-toluenesulfonic acid) from Alfa Aesar. The powder was treated in 37% HCl solution for 24 h, and then dried in air for 24 h. The powder was then washed thoroughly with DI water and filtered followed by heating in vacuum at 90°C for 20 h to remove the water.
|
|
|
Figure 1. SEM micrographs of polyaniline nanopowder: (A) 3500 x; (B) 20,000 x. |
Charging
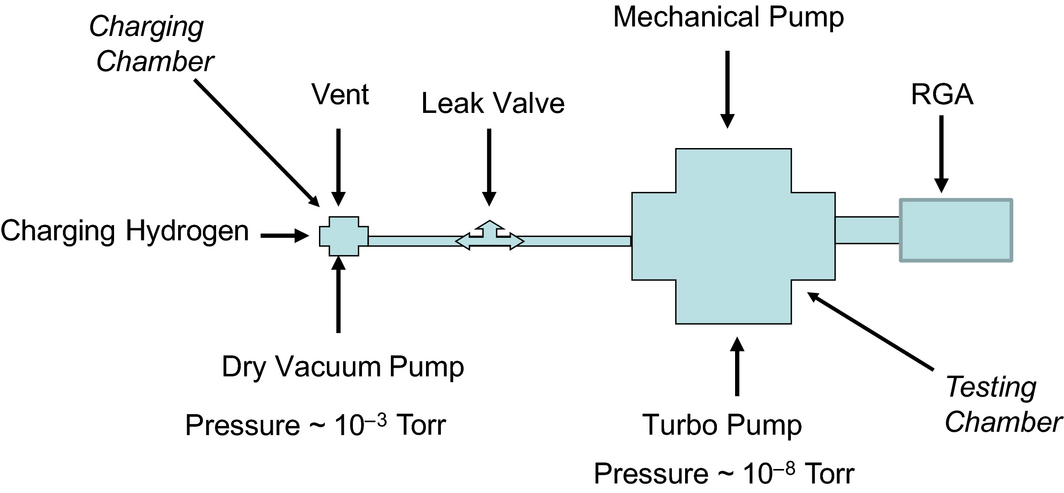
Powder samples of PANI of approximately 1 g each were loaded into a small hydrogen charging chamber as shown in Figure 2. The charging chamber of volume 15 cm3 is connected to a dry vacuum pump, a hydrogen cylinder to charge the polymer, and a transfer line with a leak valve connected to a measurement chamber containing a residual gas analyzer (RGA). The charging chamber is surrounded by a heater for varying the temperature at which hydrogen is charged into the polymer, as well as for undertaking temperature programmed desorption in the discharging of adsorbed hydrogen. The charging chamber is initially evacuated to 10−3 Torr, after which the polymer is heated to 150°C under vacuum for 2 h to fully desorb any species that may have been adsorbed during the PANI processing. The sample is then exposed to 99.999% pure hydrogen at pressures as high as 3 × 106 Pa for a certain time at a specific temperature. Following the exposure, the hydrogen is pumped out and the charging chamber is returned to a low vacuum condition (10−3 Torr).
|
|
|
Figure 2. Schematic of charging and testing system for hydrogen storage in polymers. |
Discharging
As shown in Figure 2 the charging chamber is connected to a larger gas measurement chamber through a leak valve so as to examine by thermal desorption spectroscopy (TDS) the amount of hydrogen loaded in the polymer. The base pressure in the gas measurement chamber to which the residual gas analyzer (RGA) system is connected, is ~1 × 10−8 Torr. With the RGA operated in the region of linear response, and the measurement chamber pumped at a constant speed, the output signal of the mass spectrometer at a given mass number, which is the ionization current I(t), is proportional to the desorption rate of that mass specie. Integrating the area under the ionization signal for given mass specie as a function of time or temperature yields a qualitative measure of the amount of the mass species. Thus, the species being desorbed from the polymer as it is heated in the charging chamber can be measured qualitatively. Of specific interest is hydrogen desorption. Any contributions to the hydrogen signal due to desorption from other parts of the chamber were normalized out through difference measurements. Since PANI is nominally stable up to ~200°C, the heating at a rate of 20°C/min was programmed to stop at 160°C. A quantitative measurement of the gas desorption from the polymers was done by calibrating the RGA through feeding the testing chamber with a known amount of hydrogen [9]. Additional calibration was also done by relating the area under the curve in RGA measurement to the corresponding wt% change in TGA measurement. The percentage of hydrogen uptake in the polymers was then calculated. Multiple charge–discharge cycles were preformed on the PANI so at measure hydrogen retention.
TGA set-up
Thermogravimetric analysis was used to directly measure the weight change of the polymer samples. A TGA/DSC1-1600C (Mettler Toledo, Columbus, OH, USA) system was used to measure weight loss from room temperature to 250°C also using heating and cooling rates of 20°C/min. The typical sample mass was about 10 mg and the accuracy of the equipment is 10−4 mg. The measurement was performed in open air without any air flow. TGA measurements required the removal of the loaded PANI sample from the vacuum charging/test system into laboratory air which presented the possibility of desorption of hydrogen from PANI in laboratory air. Such desorption was not seen to occur as shown by TGA measurements indicating essentially no difference in desorbed hydrogen amount from as-removed PANI, and PANI that was kept in laboratory air for >1 × 106 sec.
Results and Discussion
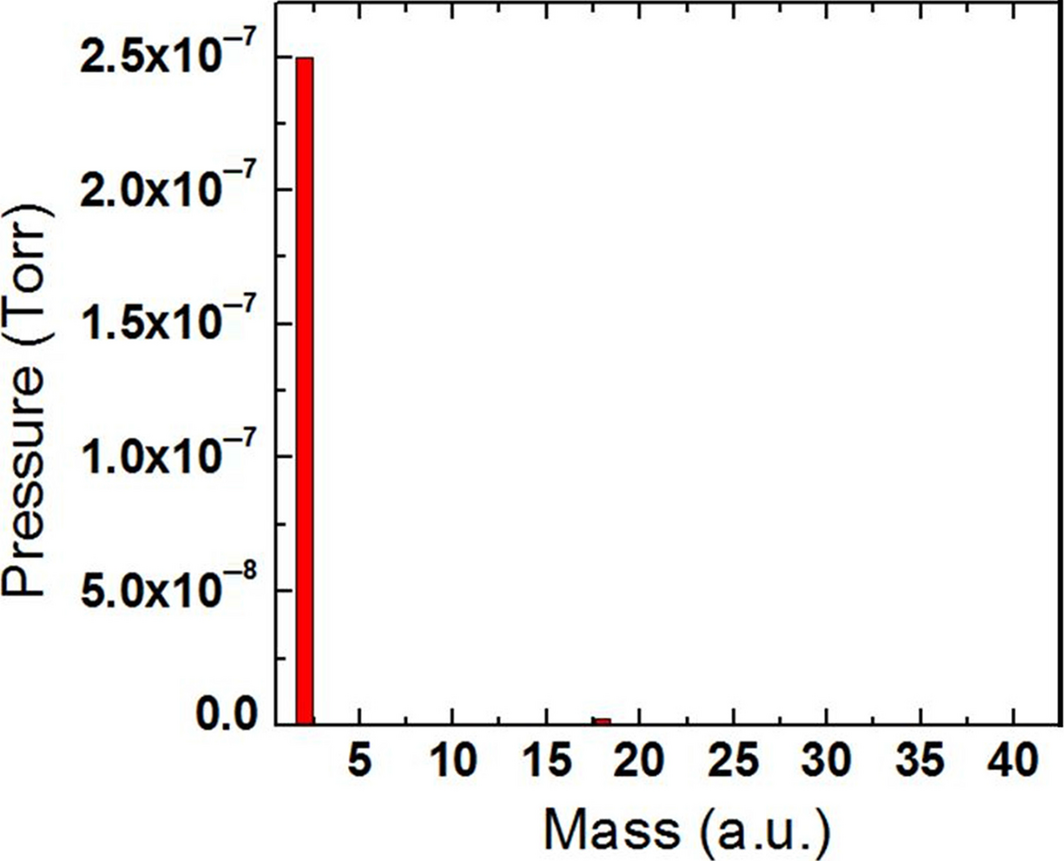
Thermal desorption spectroscopy was performed following the charging of the PANI samples. Figure 3 shows the mass spectrum of the charged PANI sample at a temperature of 120°C. Note that the mass spectrometer resolution was set so as to incorporate H and H2 into one histogram bar. The slight water vapor signal at 18 a.u. accounts for <2% of the total mass components observed during TDS.
|
|
|
Figure 3. Mass spectrometer histogram of desorbed species when hydrogen charged polyaniline was at a temperature of 120°C. |
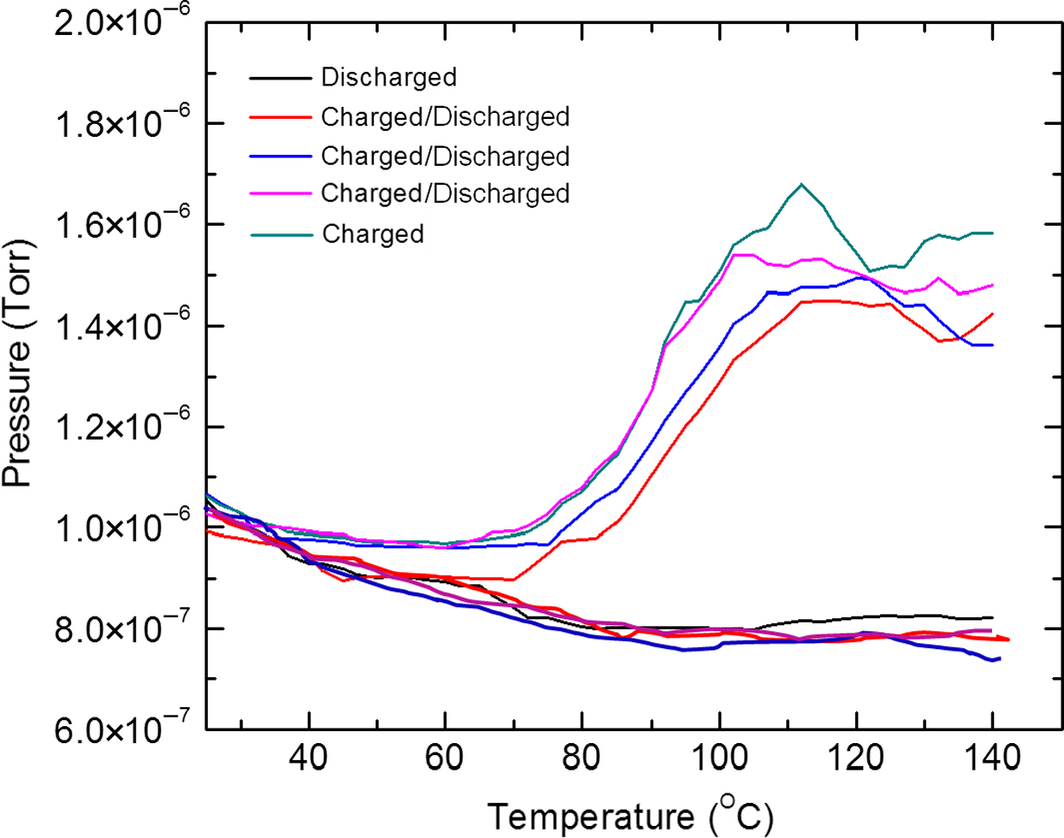
Figure 4 shows the TDS scans of a PANI sample undergoing a sequence of charge and discharge cycles. As noted, with the area under the desorption curve being directly proportional to the number of hydrogen molecules desorbed [9], the difference between the area of a charged PANI and the corresponding discharged PANI is an indication of the number of hydrogen molecules stored in the PANI. This yielded a hydrogen uptake value of about 2.5%. Good overlap is seen for the cycles which were extended to 12 with this number limited by personnel time and hydrogen use safety. Hence this data indicates that the PANI is rechargeable and may hold promise for hydrogen storage.
|
|
|
Figure 4. Hydrogen thermal desorption spectroscopy curves for cycling of hydrogen charging/discharging for polyaniline (PANI) charged at 3 × 106 Pa hydrogen pressure for 2 h at a PANI temperature of 80°C. |
The TDS study of hydrogen charging-discharging in PANI has given semiquantitative results regarding the hydrogen uptake in the nanostructured polymer. However, due to the nonlinear response of the RGA at high pressures and the need for accurate calibration of the system, a more accurate quantitative measurement was best obtained using the TGA.
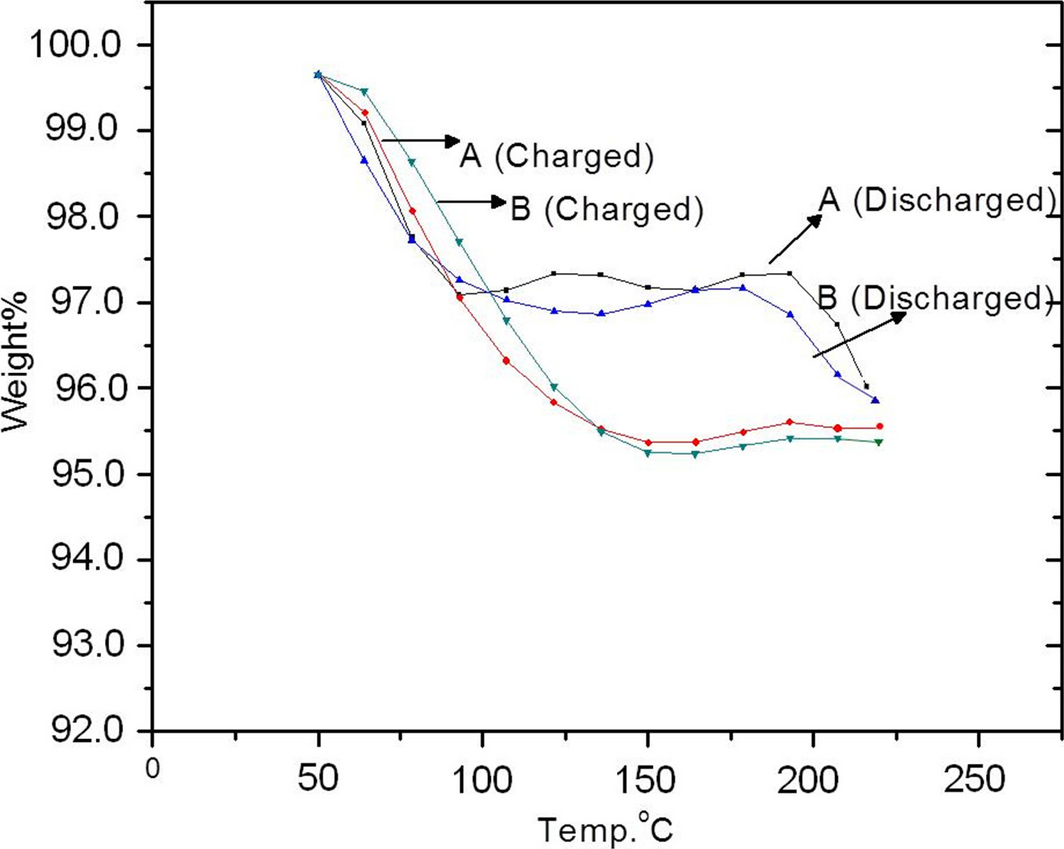
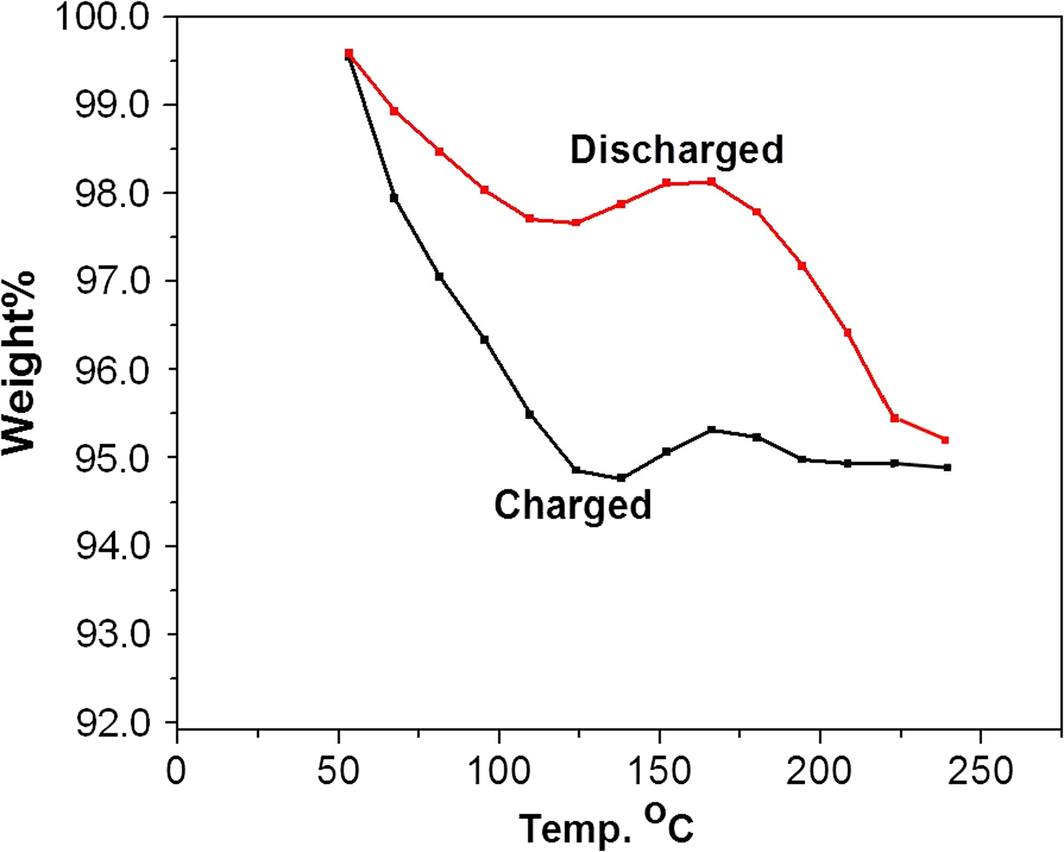
Thermogravimetric analysis measurements undertaken on PANI utilized two 1.6 g samples of PANI charged with hydrogen for 1.5 h at 80°C: one at 2.0 × 106 Pa and the other at 1.38 × 106 Pa. The samples were then discharged by heating accompanied by weight loss measurements of the sample. The TGA results for the PANI samples are shown in Figure 5 and first indicate little or no dependency of response on charging pressure within the limited pressure range used. Furthermore, an initial decrease in weight is observed upon the start of thermal desorption. This is attributed to adsorbed molecules coming off the PANI and is identical for both the charged and the discharged states. The discharged sample curves are essentially flat between ~100°C and ~200°C at around 97 wt%. The charged sample curves, however, indicate that PANI keeps loosing weight until ~150°C, at which point the curve becomes flat between 150°C and 200°C at ~95 wt%. Since the charged and discharged PANI are taken from the same sample, the extra weight loss in the charged PANI is attributed to the hydrogen loss (~2 wt %). Both charged and discharged curves show loss of mass above 200°C due to PANI decomposition. On the other hand, the TDS measurements for PANI using the RGA yielded a weight loss of ~2.5 wt%. This difference in comparison to the TGA measurements can be understood from the variability in charged/discharged states seen in Figure 4.
|
|
|
Figure 5. Thermogravimetric analysis results from two samples of polyaniline charged with hydrogen at 80°C. (A) charged at 1.38 × 106 Pa; and (B) charged at 2.0 × 106 Pa both for a duration of 1½ h. |
The TGA results of Figure 5 indicate that at the low charging pressures used in this study (as compared to other studies [8]), the hydrogen uptake in PANI is essentially independent of the hydrogen charging pressure. A larger wt % of hydrogen absorption in PANI has been reported under much higher hydrogen charging pressures (9 × 106 Pa) [8], as well as at lower charging temperatures (to LN2 temperatures) [5, 10]. However, we do not consider low-temperature operation (e.g., LN2 temperature) as viable for future hydrogen storage applications, and hence did not study the low-temperature absorption/adsorption regime for the polymers. With respect to charging pressure our charging system limited our investigation to pressures below ~3 × 106 Pa.
The discussion as to whether hydrogen adsorption in PANI is physisorption or chemisorption has been ongoing for some time. Authors have indicated only low-temperature adsorption/desorption as a physisorption mechanism [5, 10, 11], whereas others have noted elevated temperature adsorption/desorption related to chemisorptions [6, 8, 9, 12]. Our data show adsorption of hydrogen in PANI at temperatures up to 80°C, and desorption at temperatures up to 200°C. Although we have not been able to define the specific adsorption pathway for hydrogen in PANI, such high desorption temperatures preclude a physisorption mechanism and point directly to chemisorptions as active in the hydrogen/PANI system.
Hydrogen loading into PANI may lead to microstructural changes in the material; hence SEM micrographs were taken of PANI after hydrogen loading. A PANI sample was charged with hydrogen under the conditions used for the sample in Figure 5, and was evaluated by SEM with the results shown in Figure 6. Under comparison to a virgin PANI sample (Fig. 1B) it can be seen that very little if any microstructural changes are seen in the hydrogen-loaded sample. Noting that the change in a hydrogen-loaded sample is approximately 3 wt%, microstructural changes may only be at the nanometer level which would not be possible to resolve under SEM.
|
|
|
Figure 6. SEM micrograph of polyaniline charged with hydrogen at 2.0 × 106 Pa for 1½ h. Compare with Figure 1B. |
Stability
Not only is hydrogen cycling important for a hydrogen storage medium, but hydrogen retention is of major interest for hydrogen storage. Samples of charged and discharged PANI were left for 3 weeks under standard temperature and pressure conditions in laboratory air to test the stability of the holding power for hydrogen by PANI. TGA measurements of hydrogen retention were then taken with results shown in Figure 7. The data of Figure 7 indicate that the hydrogen retention in PANI for the 3-week stored sample was measured at 3% which is essentially equivalent to that for a freshly loaded sample. Hence, good retention is seen for PANI over the 1.8 × 106 sec retention measurement period indicating that PANI is a good candidate for a long-term hydrogen storage medium.
|
|
|
Figure 7. Thermogravimetric analysis results for a polyaniline sample charged with hydrogen at 2.0 × 106 Pa and then discharged 3 weeks later (1.8 × 106 sec). |
Conclusion
Polyaniline (PANI) nanostructured polymer was charged at room temperature with hydrogen at ~3.0 × 106 Pa to examine the potential and the capacity of the conducting polymer for storing hydrogen. TGA and TDS results show a storage capacity of from ~2 to ~3 wt%. The charge/discharge cycling of the PANI were examined using TDS, and confirmed loading and unloading of the polymer samples for a number of cycles. Further it was shown that stored hydrogen remains intact in PANI for more than 3 weeks; a proof of storage stability. These data indicate the possible use of nanostructured PANI polymer for hydrogen storage, with hydrogen storage amounts at the 3% wt level.
Acknowledgments
The authors wish to acknowledge the support of the R.A. Welch Foundation through Grant E-632, the State of Texas through the Center for Advanced Materials, and the World Class University Program at GIST funded by the Ministry of Education, Science, and Technology through the National Research Foundation of Korea (R31-10026).
Conflict of Interest
None declared.
References
- Mori, D., and K. Hirose. 2009. Recent challenges of hydrogen storage technologies for fuel cell vehicles. Int. J. Hydrogen Energy34:4569.
- Liu, C., Y. Y. Fan, M. Liu, H. T. Cong, H. M. Cheng, and M. S. Dresselhaus. 1999. Hydrogen storage in single-walled carbon nanotubes at room temperature. Science286:1127–1129.
- Ye, Y., C. C. Ahn, C. Witham, B. Fultz, J. Liu, A. G. Rinzler, et al. 1999. Hydrogen adsorption and cohesive energy of single-walled carbon nanotubes. Appl. Phys. Lett.74:2307–2309.
- Bianco, S., M. Giorcelli, S. Musso, M. Castellino, F. Agresti, A. Khandelwal, et al. 2009. Hydrogen adsorption in several types of carbon nanotubes. J. Nanosci. Nanotechnol.9:6806–6812.
- Germaine, J., J. M. J. Freshet, and F. Svec. 2007. Nanoporous, hypercrosslinked polypyrroles: effect of crosslinking moiety on pore size and selective gas adsorption. J. Mater. Chem.17:4989–4997.
- Srinivasan, S. S., R. Ratnadurai, M. U. Niemann, A. R. Phani, D. Y. Gosmami, and E. K. Stefanakos. 2010. Reversible hydrogen storage in electrospun polyaniline fibers. Int. J. Hydrogen Energy35:225–230.
- Foreman, J. P., and A. P. Monkman. 2003. Theoretical investigation into the structural and electronic influences on the hydrogen bonding in doped polyaniline. J. Phys. Chem. A107:7604–7610.
- Cho, S. J., K. Choo, D. P. Kim, and J. W. Kim. 2007. H2 sorption in HCL-treated polyaniline and polypyrrole. Catal. Today120:336–340.
- Diaz-de Leon, M. J.2001. Proceeding of The National Conference on Undergraduate Research (NCUR), University of Kentucky.
- Lee, D., K. Char, S. W. Lee, and Y. W. Park. 2003. Structural changes on polyaniline/montmorillonite nanocomposites and their effects on physical[properties. J. Mater. Chem.13:2942–2947.
- Panella, B., M. Hirscher, and B. Ludescher. 2007. Low-temperature thermal-desorption mass spectroscopy applied to investigate the hydrogen adsorption on porous materials. Microporous Mesoporous Mater.103:230–234.
- Jurczyk, M. U., A. Kumar, S. Srinivasan, and E. Stefanakos. 2007. Polyaniline-based nanocomposite materials for hydrogen storage. Int. J. Hydrogen Energy32:1010–1015.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?