Abstract
A new model for predicting the specific chemical exergy of municipal solid waste (MSW) is presented; the model is based on the content of carbon, hydrogen, oxygen, nitrogen, sulfur, and chlorine on a dry ash-free basis (daf). The proposed model was obtained from estimations of the higher heating value (HHV) and standard entropy of MSW using statistical analysis. The ultimate analysis of 56 different parts of MSW was used for the derivation of the HHV expression. In addition, 30 extra parts were used for validation. One hundred and seventeen relevant organic substances that represented the main constituents in MSW were used for derivation of the standard entropy of solid waste. The substances were divided into different waste fractions, and the standard entropies of each waste fraction and for the complete mixture were calculated. The specific chemical exergy of inorganic matter in the waste was also investigated by considering the inorganic compounds in the ash. However, as a result of the extremely low value calculated, the exergy of inorganic matter was ignored. The results obtained from the HHV model show a good correlation with the measured values and are comparable with other recent and previous models. The correlation of the standard entropy of the complete waste mixture is less accurate than the correlations of each individual waste fraction. However, the correlations give similar results for the specific chemical exergy, indicating that HHV has a greater impact when estimating the specific exergy of solid waste than entropy.
Introduction
The quantities of solid wastes increase as the population continues to grow throughout the world, whereas the available space for disposal decreases. If this waste is not properly treated and handled, it will pollute the air, land, ground water, and soil as well as has a negative impact on the hygienic conditions of the people [1]. A waste-to-energy (WTE) plant is one of the most robust and effective posttreatment options to decrease the volume of produced waste, reduce greenhouse gas emissions, and utilize the energy content in nonrecyclable waste for the production of electricity and heat, thereby reducing the dependence on fossil fuel.
For the efficient design of energy conversion processes, the chemical exergy and energy content of the fuel are basic properties to be considered to estimate the maximum available energy entering the system for performance analysis and optimization of the entire process. This can be done by detecting and evaluating quantitatively the thermodynamic imperfection (exergy loss) of the process under consideration, its main sources of loss and possible ways of improving such process can be indicated [2]. Estimating the chemical exergy of fuel is an important step when performing exergy analysis [3] in waste-to-energy plants. Exergy analysis is a method that uses the conservation of mass and energy together with the second law of thermodynamics. This analysis method is useful to achieve a more efficient energy-resource use because it enables the locations, types, and magnitudes of losses to be identified and to determine meaningful efficiencies [4].
However, because many solid fuels have unknown structures and chemical compositions, their exergy values cannot be calculated directly because of the lack of standard absolute entropy values [5]. Many models for the prediction and estimation of the chemical exergy of carbon-based fuels with complex bond interactions and unknown thermodynamics properties have been proposed based on the characteristics of the known homogeneous organic substances in the fuel. The first attempt was performed by Rant [6], involving the formulation of a semiempirical model to evaluate the availability (exergy) content of a structurally complicated material species. In that model, the chemical exergy of a fuel is evaluated from the computation of pure organic substances of known absolute entropies. Rant evaluated the ratios of the estimated chemical exergies and the higher heating values for seven gases and 12 liquid organic substances. Szargut and Styrylska [7] improved Rant′s correlation by considering the chemical composition of the fuels. They obtained correlation formulas to express the dependence between the ratio of the standard chemical exergy to the lower heating value using mass ratios of hydrogen, oxygen, nitrogen and sulfur to carbon that describe the chemical composition of the fuel. Due to lack of thermodynamic data, sulfur was not considered in their model for solid fuels, and their correlations were theoretically limited to Szargut′s reference environmental (R.E) model.
Using a model for estimating the thermodynamic properties of coal, char, tar, and ash, Eisermann et al. [8] approximated the standard entropy of coal by comparing the behavior of the standard entropies of a number of aliphatic and aromatic hydrocarbons as a function of several elemental ratios: H/(C + N), O/(C + N), N/(C + N) and S/(C + N). Shieh and Fan [9] estimated the specific chemical exergy of a structurally complicated material by adopting the concepts of the dead (or reference) state and the properties of the constituents in the material based on the first and second laws of thermodynamics. It was assumed that the entropy of a fuel is equal to the entropies of its constituent elements. This assumption is not accurate in many cases. Ikumi et al. [10] developed a method for estimating the entropies of coals based on the mole ratios of hydrogen, oxygen, nitrogen, and sulfur elements to the carbon element. Bilgen and Kaygusuz [11] used the entropy correlation proposed by Eisermann et al. [8] to improve the Shieh and Fan [9] model for the calculation of the chemical exergy of coals, and Stepanov [5] applied the entropy model developed by Ikumi et al. [10] to modify Shieh and Fan [9] to calculate the exergies of coke-oven gases of different metallurgical mills. These models are limited to coal fuels only because their constituent organic compounds have been derived from the standard entropies of the relevant organic substances of coals.
Song et al. [3] developed a model based on Shieh and Fan [9] to estimate the specific entropy of the organic matter in biomass used for the exergy calculations. Although their model showed a high accuracy and was simpler than the Szagut and Styrylskas correlation, it has a limited application, as it is only applicable to biomass. Song et al. [12] also proposed a model for estimating the entropy of solid fuels and then extended the Shieh and Fan [9] model using the major organic constituents of solid fuel for the prediction of the specific chemical exergy of solid fuels. However, they combined the higher heating value derived on a dry basis (db) with values of the standard entropy obtained based on a dry ash-free basis (daf) for the estimation of the chemical exergy. Furthermore, their model cannot be used for estimating the chemical exergy of substances containing elements other than C, H, O, N, and S and for combustible materials, such as certain categories of leather, plastic, and rubber, that are part of municipal solid waste. To the authors knowledge, no model has been found in the literature that is derived for predicting the chemical exergy of MSW.
The objective of this work is to propose a model for calculating the specific chemical exergy of MSW containing the C, H, O, N, S, and Cl from its elemental compositions on a dry ash-free basis.
Derivation of the Estimated Model
Municipal Solid Waste consists of a complex, heterogeneous mixture of organic and inorganic substances. The organic elements in MSW are mainly C, H, O, N, S, and Cl, which can be obtained from ultimate analysis, whereas the inorganics are commonly Si, Ca, K, P, Al, Mg, Fe, S, Na, Zn, Cu, Mn, and Cr, from which their oxides can be obtained from ash analysis data. Previous reports have shown that the influence of inorganic matters on the exergy value can be neglected in solid fuel as a result of their relatively small value [3, 12].
The standard chemical exergy of a substance that is not present in the environment can be evaluated by considering a reaction of the substance with other substances for which the chemical exergies are known [13]. The exact calculation of the chemical exergy of a material with complicated structures is difficult [14]; as a result, the standard chemical exergy of the substance in the environment is not readily available.
Standard chemical exergy of a substance
The chemical exergy of a substance is equal to the maximum amount of work that can be obtained from the substance by taking it to chemical equilibrium with the reference environment [15]. The standard exergy of a substance can be evaluated by considering an idealized reaction of the substance with other substances (generally reference substances) of known chemical exergies [16]. The known chemical exergies can be obtain from the table of standard chemical exergy based on Szarguts R.E model (Model II), as shown in Table 1. With consideration of the reversible reaction for chemical formation of a compound, Szargut et al. [2] expressed the standard chemical exergy of elements or compounds as
|
|
(1) |
| Substance | e0 (kJ/mol) | s0 (kJ/mol K) |
|---|---|---|
| ||
| CO2 | 19.87 | 0.214 |
| H2Ol | 0.95 | 0.070 |
| O2 | 3.97 | 0.205 |
| N2 | 0.72 | 0.192 |
| SO2 | 310.93 | 0.248 |
| SiO2 | 1.636 | 0.041 |
| HCl | 85.5 | 0.187 |
| CaO | 129.881 | 0.038 |
| K2O | 412.544 | 0.102 |
| P2O5 | 377.155 | 0.117 |
| Al2O5 | 4.479 | 0.051 |
| MgO | 62.417 | 0.027 |
| Fe2O3 | 17.656 | 0.087 |
| SO3 | 242.003 | 0.257 |
| Na2O | 296.32 | 0.075 |
| MnO | 122.390 | 0.060 |
| ZnO | 37.080 | 0.042 |
| Cr | 538.610 | 0.024 |
| Pb | 226.940 | 0.065 |
| As | 477.040 | 0.035 |
| Cd | 290.920 | 0.052 |
| Cl | 163.940 | 0.166 |
where , ni, and represent the standard Gibbs energy of formation, the mole fraction of component i in the mixture, and the standard chemical exergy of the constituent element i, respectively.
Calculation of the specific chemical exergy of municipal solid waste
For simplicity, suppose 1 kg of MSW (daf), expressed as CmHnNpOqCrSt, undergoes complete combustion at a standard state for the steady condition to produce carbon dioxide, water, nitrogen, hydrogen chloride, and sulfur dioxide as follows:
|
|
(2) |
All substances are assumed to enter and exit at the reference temperature, T0 = 298.15 K, and reference pressure, P0 = 101.325 kPa. The subscripts m, n, p, q, r, and t are the numbers of atoms of C, H, N, O, Cl, and S, respectively, in kmol/kg MSW or the molal compositions per kg of MSW expressed as:
|
|
(3) |
|
|
(4) |
|
|
(5) |
|
|
(6) |
|
|
(7) |
|
|
(8) |
where the elements in Equations (3)-(8) are expressed in wt% (daf). For the steady state, under the standard condition, the energy balance of the reaction in Equation (2) is given by
|
|
(9) |
The entropy balance is expressed as
|
|
(10) |
where W and Q are the work and heat transfer, respectively. Sgen is the entropy generated by the irreversibility in the reaction, and s0 and h0 represent the standard entropy and enthalpy, respectively.
Eliminating the heat transfer Q between Equation (9) and (10) gives the following:
|
|
(11) |
The maximum work, Wmax, will occur when there is no irreversibility in the system. Hence, Equation (11) can be expressed as
|
|
(12) |
or
|
|
(13) |
where represents the heat of reaction of the combustion process, which is equal to the negative higher heating value [9], that is
|
|
(14) |
then
|
|
(15) |
Assume that the reaction in Equation (2) at 298.15 K and 101.325 kPa is an adiabatic process with no irreversibility. The exergy balance equation, in absence of changes in the kinetic and potential energy for reacting systems, is given by
|
|
(16) |
where e represents the specific chemical exergy. Substituting Equation (15) into Equation (16), the specific chemical exergy of MSW (daf), eMSW, is presented as
|
|
(17) |
or
|
|
(18) |
where e0 is the standard exergy in kJ/mol and s0 is the standard entropy in kJ/mol K, as tabulated in Table 1. is the standard entropy of municipal solid waste, in kJ/K kg (daf), and HHVMSW is the higher heating value (HHV) of MSW, in kJ/kg (daf). The specific chemical exergy of MSW can be calculated once the standard chemical exergies of CO2, H2O(l), N2, O2, SO2, and HCl; the higher heating value; and the absolute entropies are known.
Estimating the higher heating value of municipal solid waste
In the absence of a measured value, the HHV of fuel can be estimated from their elemental composition [19, 20]. In this study, the HHV estimate was performed by considering 56 data points and 30 data points of MSW samples for the derivation and validation of the correlation, respectively; in addition, the chemical composition and HHV of each sample was collected from the published literature and presented in Tables A1 and A2 (Appendix 1). These data cover six categories of combustible MSW fractions, namely, food, wood, paper, textiles, plastics, and rubber waste [21, 22]. For the selection of a suitable model, 9 assumed algebraic expressions from previous work based on the correlation of the HHV and ultimate analysis of solid fuel (daf) were used, as shown in Table 2. Using regression analysis based on the generalized method of least squares [19] on the 56 data points, the constant terms of these algebraic expressions were evaluated. The correlation that has the least error and highest coefficient of determination, as described in 'Selection of the best correlation', was selected. The newly estimated correlation was compared with the experimental values of HHV and the results of previous models collected from the open literature, for further validation.
| S. No. | Assumed expression | Criteria for selection | Reference |
|---|---|---|---|
| |||
| 1. | HHV = aC + bH + cO + dN + eS + fCl | Assuming fuel HHV to be a linear function of it constituents. | Current model |
| 2. | HHV = aC + bH + cO + dN + eS | Based on Gumzs criteria | [19] |
| 3. | HHV = a0 + bH + cO + dN + eS + fCl | Based on Changs criteria | [23] |
| 4. | HHV = aC + b (H − O/8) + eS | Based on Dulongs criteria | [19] |
| 5. | HHV = aC + bH + cO + eS | Based on modified version of Dulongs criteria | [19] |
| 6. | HHV = a0 + aC + bH + cO2 | Based on Seylers criteria | [19] |
| 7. | HHV = a (C − (3/8) O) + b (3/8) O) + c (H − (1/6) O) + eS | Based on Steuers criteria | [19] |
| 8. | HHV = a (C − 0.75 (O/2)) + b (H − 0.125 (O/2)) + eS | Based Sumegis criteria | [19] |
| 9. | HHV = aC + bH + c ((N + O − 1)/8) + eS | Dulong-Berthelots criteria | [19] |
Estimating the standard entropy of municipal solid waste
Municipal solid waste (MSW) contains mainly organic polymers in plastics, wood, paper, textile, rubber, and food waste. The entropies of these polymers in the organic waste are estimated or evaluated by the entropies of their organic monomers structures as there is no significant difference between the entropies of the solid organic monomers and their polymers [24]. The difference ranges from 0.1 to 12.5% (Table 3).
| S. No. | Substance | S0 (J/mol K) |
|---|---|---|
| ||
| 1. | C6H11NO | 173.21 |
| (C6H11NO)n | 173.0 | |
| 2 | C4H4O4 | 157.2 |
| (C4H4O4)n | 151.4 | |
| 3 | C15H10N2O2 | 332.5 |
| (C15H10N2O2)n | 294 | |
| 4 | C13H24O2 | 401.9 |
| (C13H24O2)n | 351.6 | |
The standard entropy of MSW was derived from organic substances with known standard entropies. In this work, 117 samples of organic compounds relevant to MSW were collected from the published literature [3, 8, 10, 12, 17, 24] and tabulated in Table A3 (Appendix 1). The data points where selected based on the molecular structures of the organic substances that are associated or linked with the formation of larger molecular structure network of municipal solid waste. The organic compounds were grouped into the six categories of waste fractions, as previously used for the higher heating value, namely: food, plastic, textile, rubber, wood, and paper. This was accomplished by considering the molecular structures of the organic substances that can be found in each of the molecular structures of the waste fractions. For wood, it contains three major chemical components: cellulose, hemicelluloses, and lignin [25]. Each of the chemical structure of the wood constituents [26, 27] was studied and organic compounds (monomers) that can be made or found from these structures are selected. In the food, the main structural elements identified are proteins, carbohydrate and lipids [28]. The molecular structures of these food components [29, 30] were also investigated and organic monomers that are linked with the structure are selected. The same method was carried on chemical structures of plastic [31], textile [32, 33], and rubber [34] materials with identifications of biologically important molecules which form the building structure of their polymers. Based on the absolute entropies and elemental compositions of the selected organic substances, a first-order polynomial correlation was derived statistically for the standard entropy of the waste fractions and the mixture.
Selection of the best correlation
Three statistical parameters were used as evaluating parameters for both HHV and the standard entropy of MSW, which are computed as follows:
|
|
(19) |
|
|
(20) |
|
|
(21) |
where Zest and Zexp denote the estimated and experimental values, respectively. is the experimental average value. AAE is the average error of a correlation. A smaller error of correlation will occur when AAE is low, which indicates higher accuracy. ABE denotes the average bias error of correlation. A positive value of ABE indicates an overall overestimation, whereas a negative value implies an overall underestimation. The smaller the absolute value of ABE, the smaller the bias of correlation. R2 is used as a comprehensive parameter to measure the accuracy of the model. A higher R2 value means a better estimation and fitting [20]. These three parameters are the important statistical criteria and are primarily employed to assess correlations [3, 12, 19].
Specific chemical exergy of inorganic matter in municipal solid waste
Inorganic substances of waste materials are contained in the ash and obtained from complete combustion of solid fuel; ash is mainly contained in various metallic oxides and has a high thermal stability [35, 36]. The specific chemical exergy of inorganic matter in kJ/kg MSW was calculated from the major ash compositions data in Table 4 from a stoker-type incinerator [37] as follows [3, 12, 36]:
|
|
(22) |
| Component | Bottom ash, BA (wt%) | Fly ash, FA (wt%) |
|---|---|---|
| ||
| SiO2 | 37.8 | 2.47 |
| CaO | 20.79 | 44.5 |
| K2O | 0.85 | 3.01 |
| P2O5 | 3.63 | 0.26 |
| Al2O3 | 13.4 | 0.55 |
| MgO | 2.91 | 0.57 |
| Fe2O3 | 7.46 | 0.32 |
| SO3 | 1.01 | 1.61 |
| Na2O | 5.38 | 4.39 |
| ZnO | 0.52 | 2.25 |
| CuO | 0.51 | 0.096 |
| MnO | 0.17 | 0.04 |
| Cr | 0.63 | 0.008 |
| Pb | 0.22 | 0.51 |
| As | 0.021 | 0.062 |
| Cd | 0.0003 | 0.003 |
| Cl | 3.51 | 35.15 |
where ni represents number of moles of the component in inorganic matter, in mol/kg. and xi are the standard chemical exergy and mole fraction of components i in inorganic matter, respectively. R is the universal gas constant, 0.0083145 kJ/mol K, and A is the ash content of MSW in wt%.
Results and Discussion
Correlation based on the higher heating value
For the higher heating value of MSW, the correlation derived that showed the minimum error with a higher accuracy among the nine assumed correlations used in Table 2 was expressed as
|
|
(23) |
|
|
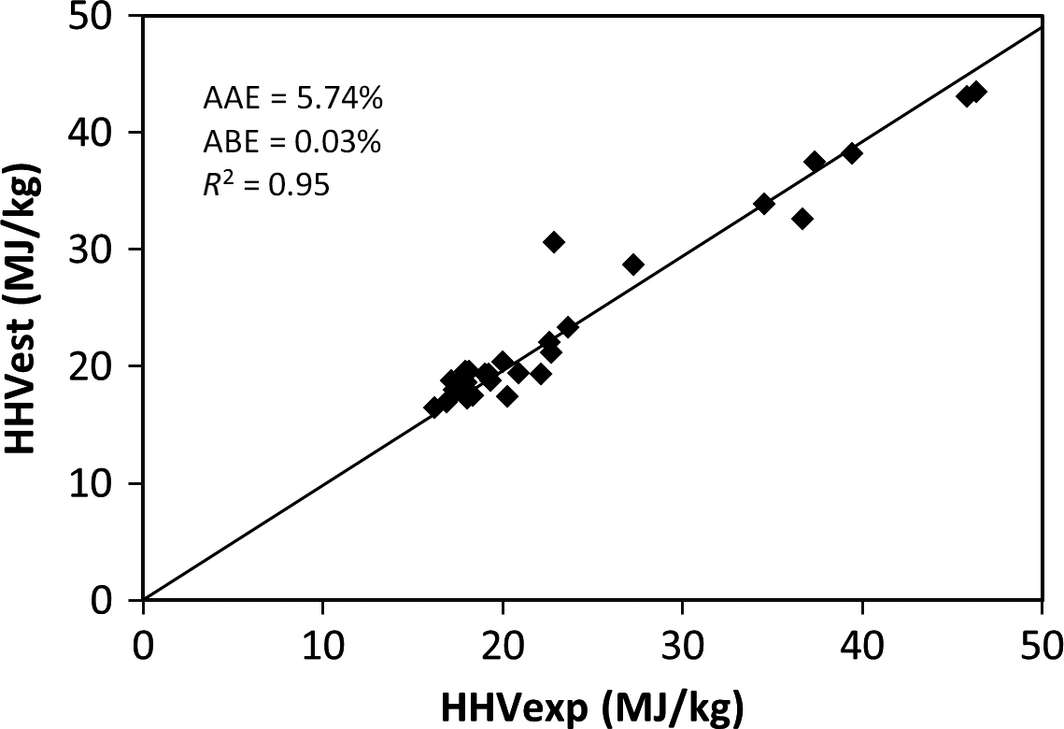
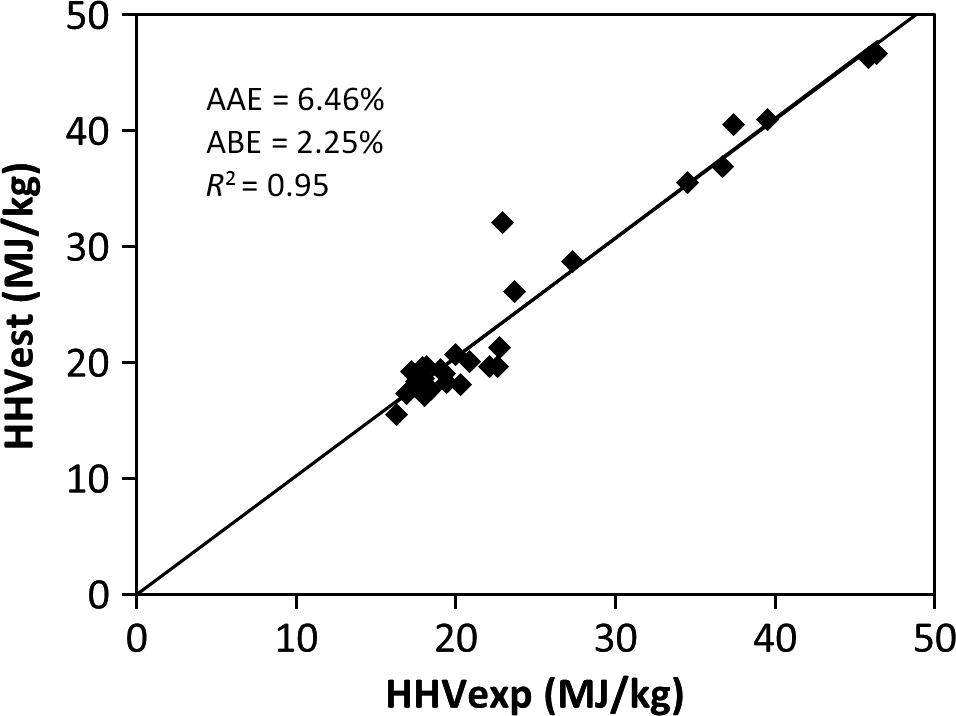
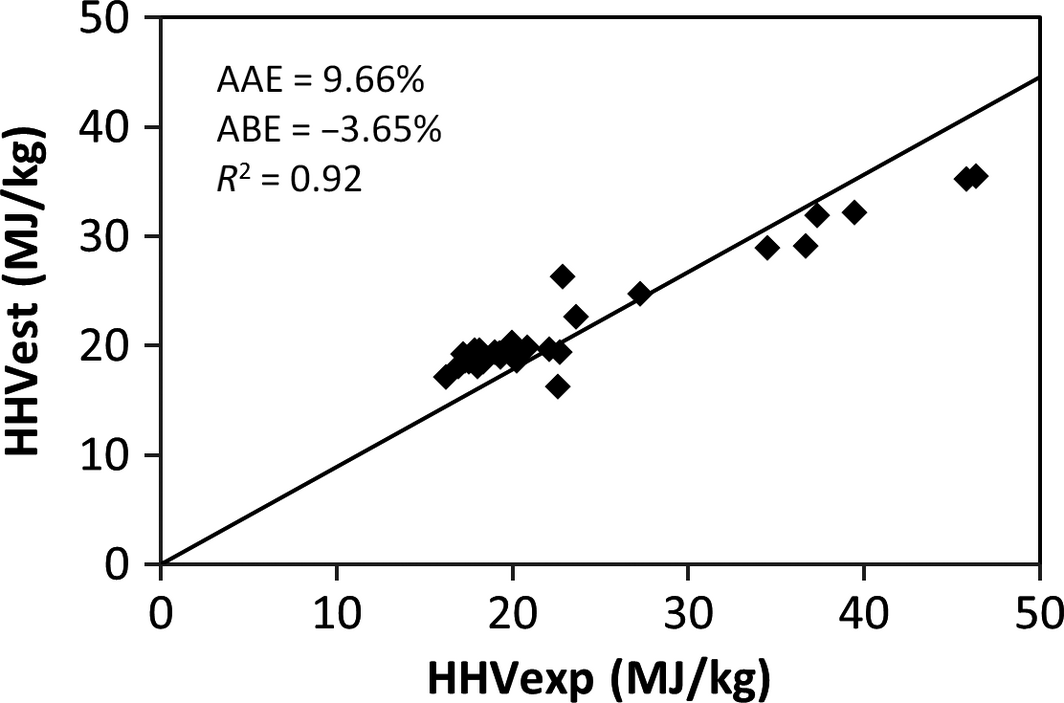
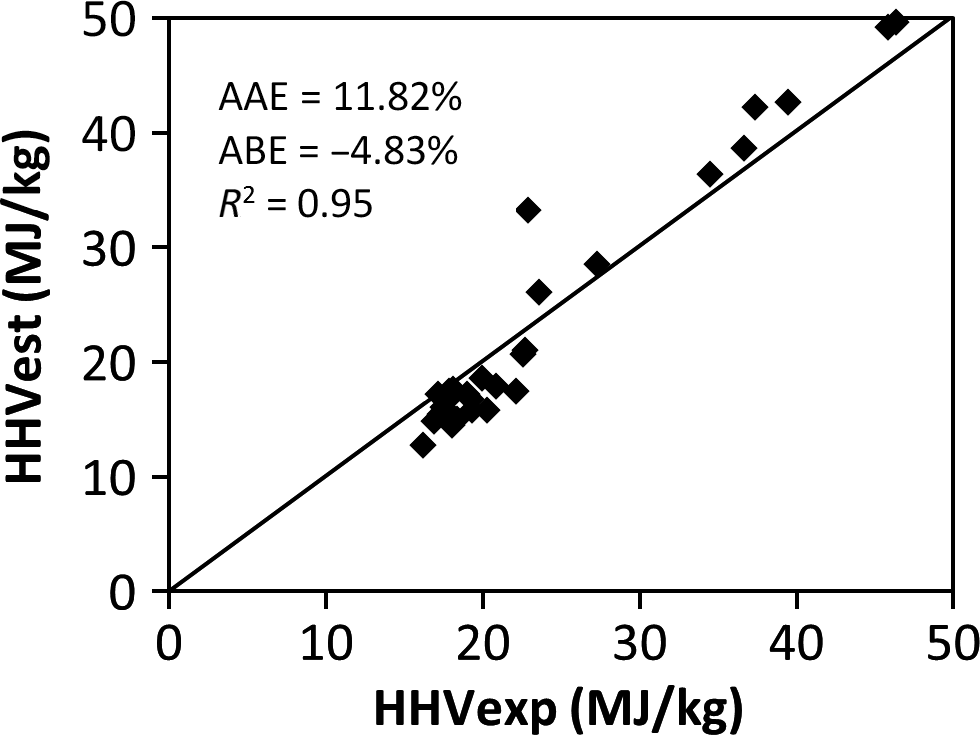
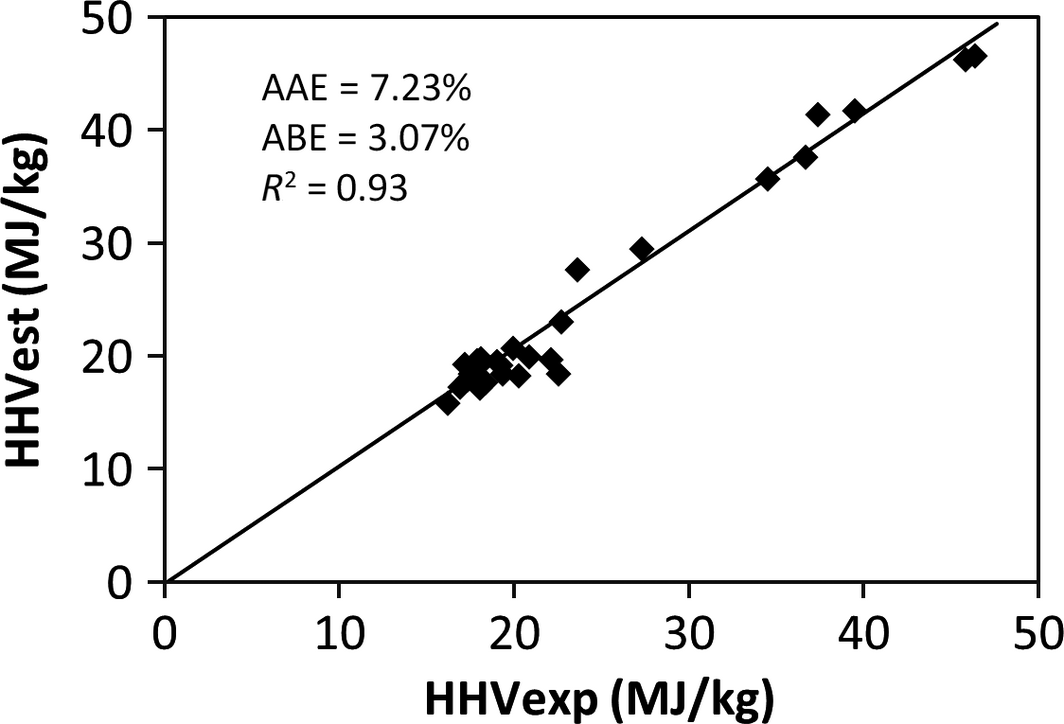
The results of the validation of the derived model and the comparison with published correlations using the experimental values of 30 samples of MSW in the different categories of food, wood, plastic, textile, rubber, and paper waste are shown in Table 5 and represented in Figures 1-5. Figures 1, 2, and 4 show the best correlation with experimental data (highest coefficient of determination), representing the model developed in this work, model by Channiwala and Parikh [19] and Dulongs correlation. However, the proposed model shows significantly better estimations when considering the errors (AAE and ABE) compared to the other models. This is not surprising, as these models have been derived from mixed solid fuel and coal. Figure 3 shows a correlation proposed by Sheng and Azevedo [20]. Although the correlation has a good coefficient of determination (R2 = 0.92), it has a higher error and underestimated the HHV. In addition, the correlation is limited to biomass. The correlation proposed by Chang (Table 5 and Fig. 5) has a considerable accuracy, with a coefficient of determination of R2 = 0.93. Although this correlation was derived from MSW, it overestimated the correlation and has a higher error value when compared with the present model.
| S No. | Name | Correlation (MJ/kg) | Application | AAE (%) | ABE (%) | R2 | Reference |
|---|---|---|---|---|---|---|---|
| |||||||
| 1. | Proposed Model | HHV = 0.364C + 0.863H − 0.075O + 0.028N − 1.633S + 0.062Cl | MSW | 5.738 | 0.032 | 0.95 | Current model |
| 2. | Channiwala and Parikh | HHV* = 0.3491C* + 1.1783H* + 0.1005S*− 0.1034O* − 0.0151N* − 0.0211A* | Mixed waste | 6.456 | 2.254 | 0.95 | [19] |
| 3. | Sheng and Azevedo | HHV* = −1.3675 + 0.3137C* + 0.7009H* + 0.0318 (100 − C* − H* − A*) | Biomass | 9.657 | −3.650 | 0.92 | [20] |
| 4. | Dulong | HHV = 0.3383C + 1.443 (H − (O/8)) + 0.0942S | Coal | 11.822 | −4.832 | 0.95 | [19] |
| 5. | Chang | HHV = 35.8368 + 0.7523H − 0.2674S − 0.4654O − 0.3814Cl − 0.2802N | MSW | 7.234 | 3.067 | 0.93 | [19] |
|
|
|
Figure 1. Comparison between the experimental and the estimated HHV by the developed correlation. |
|
|
|
Figure 2. Comparison between the experimental and the estimated HHV by the Channiwala and Parikh [19] correlation. |
|
|
|
Figure 3. Comparison between the experimental and the estimated HHV by the Sheng and Azevedo [20] correlation. |
|
|
|
Figure 4. Comparison between the experimental and the estimated HHV by Dulongs correlation. |
|
|
|
Figure 5. Comparison between the experimental and the estimated HHV by Changs correlation. |
Standard entropy of municipal solid fuel
For the prediction of the standard entropy of the organic substance in MSW, a correlation in the form of the first-order polynomial was used. The five correlations derived for estimating the standard entropy of waste fractions and the mixture of waste were expressed as follows:
For Plastic waste
|
|
(24) |
|
|
With ABE, AAE, and R2 of 0.722, 7.314, and 0.7674, respectively.
For Textile/Rubber waste
|
|
(25) |
|
|
With ABE, AAE, and R2 of 0.714, 6.476, and 0.5457, respectively.
For Wood/Paper waste
|
|
(26) |
|
|
With ABE, AAE, and R2 of 0.329, 5.215, and 0.728, respectively.
For Food waste
|
|
(27) |
|
|
With ABE, AAE, and R2 of 0.414, 5.886, and 0.6922, respectively.
For Mixed waste
|
|
(28) |
|
|
with ABE, AAE, and R2 of 1.118, 8.293, and 0.5414, respectively.
Comparing the five equations obtained, the results show that the standard entropy correlations for each waste fraction in MSW are more accurate than the standard entropy correlation for the waste mixture. This is as a result of complicated mixture, heterogeneous molecule structure and variation in municipal solid waste chemical compositions and properties. Nevertheless, because the standard entropy of plastic, textile/Rubber, wood/paper, food, and waste mixture gave similar average values for the specific exergy of the MSW estimation of 24,359, 24,364, 24,426, 24,393, and 24,387 (kJ/kg), respectively, the correlation of the standard entropy of the waste mixture can be used for the derivation of exergy.
Specific chemical exergy of municipal solid fuel (daf) and specific chemical exergy of ash
By substituting Equation (3)–(8), (23), and (24)–(28) into Equation (18), along with the standard chemical exergy data from Table 1, the specific chemical exergy of solid waste on a dry ash-free basis can be expressed as follows:
For Plastic waste:
|
|
(29) |
For Textile/Rubber waste:
|
|
(30) |
For Wood/Paper waste
|
|
(31) |
For Food waste
|
|
(32) |
For mixed waste
|
|
(33) |
The minimum, maximum, and average specific exergy values of municipal solid waste calculated were 17,602, 43,396, and 24,387 in (kJ/kg), respectively. Although Equation (33) slightly underestimated the specific chemical exergy calculated by Equations (29) and (30), that is, an ABE of −0.139 and −0.113, respectively, and slightly overestimated the exergy estimated by Equations (31) and (32), that is, an ABE of 0.179 and 0.009, respectively, when compared, the coefficient of determination shows that Equations (29)-(32) are similar to Equation (33) (i.e., a value of 1 was achieved in all cases). This result indicates that Equation (33) can be used to estimate the specific exergy of municipal solid waste, that is, the HHV has more impact on the exergy.
The overall average ratio of the specific exergy of MSW developed with the higher heating value was obtained as 1.036, showing that the value of exergy is slightly higher than the HHV. The ratio of exergies to heating values obtained in this work is similarly when compared with Szargut and Styrylska [7] model with ratio of 1.047. As their methods were commonly used for evaluating the chemical exergy of solid fuels. This result indicates that the present model is reliable and accurate. However, the slight variation in the ratios is due to different types of fuel used.
The specific exergies of inorganic matter in MSW calculated from Equation (22) using the chemical ash composition data in Table 4 are 0.86 and 1.79 kJ/kg for bottom ash and fly ash, respectively. These values are very small when compared with the average specific chemical exergy values, 24,387 kJ/kg of MSW (daf) estimated, demonstrating that the specific chemical exergy of inorganic matter can be neglected.
Conclusions
Following the evaluations of the previous equations for estimating the specific chemical exergy of solid fuels, the present proposed models in this study were found to be more accurate when using municipal solid waste as a fuel. All other methods have either ignored the inclusion of chlorine from the elemental compositions of waste or have used other solid fuels with a limited amount of MSW. In this work, a simple method for estimating the specific exergy of municipal solid waste on (daf) from their ultimate analysis based on HHV, standard entropy, and exergy equation of reaction was proposed.
The higher heating values of the estimated MSW showed a good correlation and a higher accuracy compared with previous models. It is calculated as
|
|
The standard entropy of the estimated waste mixture has a rather low accuracy when compared with the waste fractions. However, the standard entropy can be used for the estimation of the specific chemical exergy of a solid, as it showed a similar result with the standard entropy of waste fractions; the standard entropy is expressed as
|
|
This result indicates that a higher heating value has more impact on the derivation of the specific chemical exergy of solid waste than entropy. In other words, the specific exergy of MSW mainly depends on the values of HHV.
Due to very low calculated values of specific chemical exergy of inorganic matter in MSW, the specific chemical exergy developed in this work is equal to the specific chemical exergy of the organic matter in MSW and is presented as
|
|
The results obtained demonstrate that the specific chemical exergy is always slightly higher than the highest heating value, indicating the validity and accuracy of the model.
The present correlation can be accepted for estimating the specific chemical exergy of MSW using the elemental compositions of the fuel within the range specified based on a dry ash-free basis. The model is applicable for the efficient modeling of a combustion system in a waste-to-energy plant.
Acknowledgments
The authors acknowledge the Nigerian Government and Michael Okpara University of Agriculture Umudike, Abia State, Nigeria, for supporting this work through TETfund Academic Staff Training and Development.
Conflict of Interest
None declared.
Nomenclature
AAE
average absolute error
A
ash content in the waste (%)
ABE
average bias error
E
chemical exergy (kJ)
e
specific chemical exergy (kJ/kg) or (kJ/mol)
FC
fixed carbon (%)
G
Gibbs energy (kJ/kg) or (kJ/mol)
H
enthalpy (kJ/kg)
HHV
higher heating value (kJ/kg)
MSW
municipal solid waste
P
pressure (kpa)
R2
coefficient of determination
S
entropy (kJ/K)
Sgen
entropy generated
s
specific entropy (kJ/kg K) or (kJ/mol K)
T
Temperature (K)
V
volatile matter (%)
Subscripts
ba
bottom ash
daf
dry ash-free basis
est
estimate
exp
experiment
fa
fly ash
f
formation
fo
food
ioc
inorganic compound
max
maximum
msw
municipal solid waste or mixed solid waste
o
standard state
pl
plastic
R
reaction
tr
textile/rubber
wp
wood/paper
Superscripts
0
reference state
Greek Symbols
∆
change
∑
summation
Appendix
| MSW groups, Subgroup and variety | Proximate analysis (wt %) | Ultimate analysis (wt %) | HHV (MJ/kg) | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | V | FC | C | H | O | N | S | Cl | |||
| |||||||||||
| Food waste | |||||||||||
| 1. Flour | – | – | – | 42.78 | 6.19 | 48.39 | 2.48 | 0.15 | – | 18.157 | [21] |
| 2. Rice | 0.40 | 84.42 | 15.18 | 45.97 | 6.35 | 45.74 | 1.67 | 0.25 | – | 18.213 | [22] |
| 3. Peanut shell | – | – | – | 53.6 | 6.70 | 38.3 | 1.20 | 0.20 | – | 20.598 | [21] |
| 4. Pea | – | – | – | 42.13 | 5.88 | 48.62 | 3.14 | 0.22 | – | 16.533 | [21] |
| 5. Scallion | – | – | – | 48.12 | 6.27 | 41.74 | 3.09 | 0.78 | – | 18.042 | [21] |
| 6. Potato | 3.15 | 79.52 | 17.33 | 44.41 | 5.33 | 47.82 | 1.81 | 0.64 | – | 17.656 | [22] |
| 7. Spinach | 15.97 | 65.26 | 18.77 | 47.58 | 6.48 | 43.93 | 1.57 | 0.43 | – | 20.326 | [22] |
| 8. Celery | 14.58 | 65.36 | 20.06 | 38.46 | 6.16 | 54.52 | 0.21 | 0.65 | – | 15.886 | [22] |
| 9. Pakchoi | 18.44 | 63.97 | 17.59 | 43.37 | 5.93 | 48.64 | 1.25 | 0.81 | – | 23.173 | [22] |
| 10. Tangerine peel | 2.91 | 76.49 | 20.6 | 48.74 | 5.92 | 43.83 | 1.43 | 0.08 | – | 19.024 | [22] |
| 11. Banana peel | 10.85 | 64.38 | 24.77 | 35.8 | 4.79 | 54.93 | 4.37 | 0.10 | – | 18.385 | [22] |
| 12. Orange peel | 2.44 | 76.27 | 21.29 | 43.93 | 5.64 | 48.93 | 1.34 | 0.07 | 0.08 | 18.550 | [21] |
| 13. Rib | – | – | – | 52.92 | 8.83 | 25.63 | 2.29 | 0.32 | – | 17.277 | [21] |
| 14. Fish bone | 39.82 | 56.25 | 3.93 | 63.87 | 8.01 | 19.08 | 8.39 | 0.64 | – | 26.245 | [21] |
| 15. Food waste | 6.1 | 82.11 | 18.00 | 51.54 | 7.14 | 37.06 | 3.13 | 0.21 | 0.92 | 21.619 | [38] |
| Wood waste | |||||||||||
| 16. Poplar wood | 7.54 | 73.85 | 18.61 | 51.36 | 5.86 | 41.00 | 1.52 | 0.22 | – | 20.009 | [22] |
| 17. Poplar leaf | 15.69 | 68.74 | 15.57 | 49.54 | 5.24 | 43.30 | 1.32 | 0.59 | – | 19.986 | [22] |
| 18. Chinar leaf | 9.23 | 69.74 | 21.03 | 52.95 | 4.88 | 40.51 | 1.01 | 0.65 | – | 21.064 | [22] |
| 19. Gingko leaf | 11.62 | 73.19 | 15.19 | 41.35 | 5.54 | 50.88 | 1.36 | 0.87 | – | 17.289 | [22] |
| 20. Pine wood | 0.95 | 83.5 | 15.54 | 50.51 | 5.95 | 43.39 | 0.11 | 0.03 | – | 19.834 | [21] |
| 21. Sawdust | 0.42 | 81 | 18.58 | 49.42 | 7.26 | 42.92 | 0.39 | 0.01 | – | 21.267 | [21] |
| 22. Wood | 1.00 | 81.62 | 18.38 | 50.10 | 6.16 | 43.47 | 0.17 | 0.02 | 0.07 | 19.697 | [38] |
| 23. Wood chips | 1.95 | 82.66 | 15.4 | 49.54 | 6.21 | 44.06 | 0.12 | 0.04 | 0.03 | 19.544 | [21] |
| 24. Bamboo | 0.69 | 81.03 | 18.27 | 50.46 | 6.32 | 42.73 | 0.22 | 0.1 | 0.16 | 19.716 | [21] |
| 25. Leaves | 8.92 | 73.7 | 17.38 | 47.25 | 5.57 | 46.26 | 0.19 | 0.73 | – | 18.882 | [21] |
| 26. Pine needles | – | – | – | 52.57 | 6.3 | 40.44 | 0.54 | 0.16 | – | 20.843 | [21] |
| 27. King grass | 7.44 | 74.12 | 18.43 | 46.91 | 5.89 | 46.3 | 0.7 | 0.21 | – | 19.428 | [21] |
| Paper waste | |||||||||||
| 28. Blank printing paper | 10.69 | 79.33 | 9.98 | 45.12 | 5.31 | 48.91 | 0.38 | 0.28 | – | 15.127 | [22] |
| 29. Tissue paper | 0.52 | 90.47 | 9.01 | 45.18 | 6.13 | 48.32 | 0.25 | 0.11 | – | 17.340 | [22] |
| 30. Newspaper | 8.07 | 79.54 | 12.39 | 48.01 | 5.71 | 45.86 | 0.33 | 0.09 | – | 18.666 | [22] |
| 31. Magazine | 29.49 | 62.44 | 8.07 | 41.04 | 8.99 | 49.15 | 0.42 | 0.4 | – | 16.771 | [21] |
| 32. Writing paper | – | – | – | 43.66 | 5.84 | 50.16 | 0.16 | 0.18 | – | 13.69 | [21] |
| 33. Cardboard | 5.27 | 81.75 | 12.97 | 46.09 | 5.36 | 48.02 | 0.32 | 0.21 | – | 18.239 | [21] |
| 34. Carton | 7.22 | 83.95 | 8.82 | 48.97 | 6.14 | 44.52 | 0.21 | 0.16 | – | 18.430 | [21] |
| 35. Printing paper | 9.70 | 82.83 | 17.17 | 47.51 | 5.98 | 46.25 | 0.14 | 0.03 | 0.09 | 18.051 | [38] |
| 36. Packaging paper | 12.2 | 85.88 | 14.12 | 46.92 | 5.92 | 46.74 | 0.22 | 0.09 | 0.10 | 17.654 | [38] |
| Textile | |||||||||||
| 37. Absorbent cotton gauze | 0.14 | 94.85 | 5.01 | 46.74 | 5.69 | 47.23 | 0.27 | 0.08 | – | 14.664 | [22] |
| 38. Cotton cloth | 3.09 | 78.71 | 18.21 | 56.49 | 5.87 | 33.3 | 3.52 | 0.18 | 0.65 | 14.664 | [21] |
| 39. Wool | 1.24 | 84.76 | 14.00 | 60.07 | 4.24 | 31.48 | 2.65 | 1.55 | – | 21.183 | [21] |
| 40. Acrylic fiber | 0.14 | 75.25 | 24.61 | 66.78 | 5.2 | 7.31 | 20.26 | 0.45 | – | 29.812 | [21] |
| 41. Chemical fiber | – | – | – | 48.09 | 7.16 | 34.06 | 9.43 | 1.26 | – | 21.959 | [21] |
| 42. Polyester taffeta | 0.44 | 90.63 | 8.93 | 60.1 | 4.5 | 35.11 | 0.28 | 0.01 | – | 22.178 | [21] |
| 43. Terylene | 0.49 | 88.6 | 10.91 | 62.16 | 4.14 | 33.12 | 0.29 | 0.28 | – | 20.963 | [22] |
| 44. Textiles | 1.40 | 82.86 | 17.14 | 52.54 | 6.19 | 39.26 | 1.76 | 0.20 | 1.42 | 21.197 | [38] |
| Plastics waste | |||||||||||
| 45. PS | 0.04 | 99.57 | 0.39 | 86.06 | 6.27 | 1.93 | 5.73 | – | – | 38.946 | [22] |
| 46. LDPE | – | 99.98 | 0.02 | 85.98 | 11.20 | 2.61 | 0.21 | – | – | 46.480 | [22] |
| 47. HDPE | 0.18 | 99.57 | 0.25 | 85.35 | 12.70 | 1.90 | 0.05 | 0.14 | – | 46.444 | [22] |
| 48. PVC | – | 94.93 | 5.07 | 38.34 | 4.47 | – | 0.23 | 0.61 | 56.35 | 20.830 | [22] |
| 49. PET | 0.09 | 90.44 | 9.47 | 63.01 | 4.27 | 32.69 | 0.04 | – | – | 23.111 | [22] |
| 50. PE | 0.15 | 99.85 | – | 85.45 | 14.32 | – | 0.16 | 0.07 | – | 46.388 | [21] |
| 51. PP | 0.02 | 99.97 | 0.01 | 85.41 | 12.51 | 1.85 | 0.23 | – | – | 46.248 | [21] |
| 52. Packaging plastic | 3.90 | 95.21 | 4.79 | 75.75 | 9.78 | 12.00 | 0.35 | 0.03 | 2.08 | 26.951 | [38] |
| 53. Other plastic | 1.30 | 99.09 | 0.91 | 84.90 | 9.63 | 0.97 | 3.35 | 0.03 | 1.11 | 41.135 | [38] |
| Rubber waste | |||||||||||
| 54. Rubber | 8.36 | 84.77 | 6.86 | 77.72 | 10.12 | 7.42 | 0 | 2.66 | 2.08 | 25.474 | [21] |
| 55. Tire | 25.70 | 68.05 | 6.25 | 79.19 | 8.45 | 11.38 | 0.69 | 0.28 | – | 35.654 | [21] |
| Other combustibles | |||||||||||
| 56. Other combustibles | 20.40 | 90.83 | 9.17 | 70.48 | 8.79 | 17.53 | 1.63 | 0.83 | 0.74 | 32.161 | [38] |
| MSW groups, Subgroup and variety | Proximate analysis (wt %) | Ultimate analysis (wt %) | HHV (MJ/kg) | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | V | FC | C | H | O | N | S | Cl | |||
| |||||||||||
| Food waste | |||||||||||
| 1. Rice | 0.42 | 87.74 | 11.84 | 44.2 | 5.73 | 48.75 | 1.20 | 0.1 | 0.02 | 18.048 | [21] |
| 2. Potato | – | – | – | 42.09 | 6.5 | 49.06 | 2.12 | 0.23 | – | 16.912 | [21] |
| 3. Orange peel | 2.91 | 76.49 | 20.6 | 48.74 | 5.92 | 43.72 | 1.43 | 0.19 | – | 19.024 | [21] |
| 4. Rib | 38.22 | 61.56 | 0.23 | 51.61 | 6.38 | 31.91 | 9.48 | 0.69 | – | 22.716 | [21] |
| Wood waste | |||||||||||
| 5. Wood | 0.82 | 81.64 | 17.54 | 48.35 | 6.62 | 44.7 | 0.04 | 0.29 | – | 20.868 | [21] |
| 6. Wood chips | 3.45 | 81.5 | 15.05 | 49.03 | 5.69 | 44.98 | 0.22 | 0.07 | – | 19.255 | [21] |
| 7. Wooden chopsticks | 2.18 | 83.45 | 14.37 | 48.79 | 5.16 | 45.7 | 0.3 | 0.04 | – | 19.355 | [39] |
| 8. Bamboo | 1.79 | 81.36 | 16.84 | 51.42 | 6.01 | 41.92 | 0.36 | 0.29 | – | 19.974 | [40] |
| 9. Leaves | 9.43 | 74.32 | 16.25 | 47.18 | 5.61 | 46.35 | 0.18 | 0.68 | – | 20.278 | [21] |
| 10. King grass | – | – | – | 48.37 | 6.30 | 44.58 | 0.49 | 0.25 | – | 22.127 | [21] |
| Paper waste | |||||||||||
| 11. Newspaper | 5.43 | 85.04 | 9.53 | 45.24 | 7.17 | 47.1 | 0.25 | 0.23 | – | 17.204 | [21] |
| 12. Printing paper | 12.3 | 87.65 | 0.04 | 44.93 | 4.55 | 50.43 | 0.09 | – | – | 16.233 | [21] |
| 13. Cardboard | – | – | – | 46.71 | 5.31 | 47.35 | 0.32 | 0.32 | – | 18.367 | [21] |
| 14. Toilet paper | 0.52 | 90.47 | 9.01 | 45.18 | 6.13 | 48.32 | 0.25 | 0.11 | – | 17.337 | [21] |
| 15. Paper food cartons | 6.93 | – | – | 48.07 | 6.55 | 45.04 | 0.16 | 0.17 | – | 18.137 | [41] |
| 16. Magazine stock | 29.26 | – | – | 46.55 | 6.56 | 46.44 | 0.16 | 0.30 | – | 17.967 | [41] |
| 17. Plastic-coated paper | 2.77 | – | – | 44.53 | 6.35 | 46.80 | 0.19 | 0.08 | – | 17.556 | [41] |
| Textile | |||||||||||
| 18. Cotton cloth | 1.52 | 84.53 | 13.95 | 46.51 | 5.8 | 46.98 | 0.43 | 0.28 | – | 17.699 | [22] |
| 19. Cotton | 1.45 | 86.7 | 11.85 | 46.19 | 6.12 | 47.07 | 0.54 | 0.08 | – | 17.500 | [21] |
| 20. Wool | – | – | – | 58.53 | 6.48 | 18.23 | 15.12 | 1.65 | – | 23.632 | [22] |
| 21. Shoe heel and sole | 30.09 | – | – | 76.13 | 10.14 | 11.10 | 0.72 | 1.92 | – | 36.676 | [41] |
| 22. leather | 10.1 | – | – | 66.74 | 8.90 | 12.79 | 11.12 | 0.44 | – | 22.892 | [41] |
| 23. Upholstery | 2.80 | – | – | 48.46 | 6.28 | 44.86 | 0.31 | 0.10 | – | 17.891 | [41] |
| Plastics waste | |||||||||||
| 24. PE | 0.15 | 99.85 | – | 85.45 | 14.32 | – | 0.16 | 0.07 | – | 46.388 | [42] |
| 25. PP | 0.16 | 99.84 | – | 84.3 | 14.44 | 1.05 | 0.18 | 0.03 | – | 45.842 | [21] |
| 26. PVC | 0.04 | 95.16 | 4.8 | 38.75 | 5.21 | – | 0.22 | – | 55.82 | 22.575 | [21] |
| 27. Polyurethane | 4.38 | 87.29 | 8.32 | 66.17 | 6.55 | 18.46 | 6.26 | 0.02 | 2.53 | 27.300 | [41] |
| 28. Plastic film | 6.72 | – | – | 72.05 | 10.42 | 16.96 | 0.49 | 0.08 | – | 34.519 | [41] |
| Rubber waste | |||||||||||
| 29. Rubber | 15.38 | 65.26 | 19.36 | 89.18 | 8.54 | – | 1.23 | 1.05 | – | 39.473 | [21] |
| 30. Tire | 19.27 | 63.11 | 17.61 | 88.56 | 8.52 | 0.88 | 0.75 | 1.29 | – | 37.364 | [21] |
| Name | Formula | S0 (kJ/kg K) |
|---|---|---|
| ||
| Food | ||
| 1. Allantoin | C4H6N4O3 | 1.233 |
| 2. Alloxan | C4H2N2O4 | 1.314 |
| 3. Arginine | C6H14N4O2 | 1.439 |
| 4. Asparagine | C4H8N2O3 | 1.322 |
| 5. Aspartic acid | C4H7NO4 | 1.279 |
| 6. Citric acid | C6H8O7 | 1.312 |
| 7. Creatine | C4H9N3O2 | 1.445 |
| 8. Cystine | C6H12N2O4S2 | 1.347 |
| 9. d-Glutamic acid | C5H9NO4 | 1.230 |
| 10. l-Lactic acid | C3H6O3 | 1.579 |
| 11. l-Phenylalanine | C9H11NO2 | 1.293 |
| 12. l-Proline | C5H9NO2 | 1.425 |
| 13. Maleic acid | C4H4O4 | 1.373 |
| 14. Malic acid | C4H6O5 | 1.199 |
| 15. Methionine | C5H11NO2S | 1.552 |
| 16. Phenanthrene | C14H14 | 1.207 |
| 17. Trytophan | C11H12N2O2 | 1.229 |
| 18. Tyrosine | C9H11NO3 | 1.181 |
| 19. Uric acid | C5H4N4O3 | 1.030 |
| 20. Valine | C5H11NO2 | 1.527 |
| 21. Xanthine | C5H4N4O2 | 1.059 |
| 22. Stearic acid | C18H36O2 | 1.531 |
| 23. Taurine | C2H7NO3S | 1.231 |
| 24. Urea | CH4N2O | 1.742 |
| 25. Hexadecanoic acid | C16H32O2 | 1.764 |
| 26. Adenine | C5H5N5 | 1.118 |
| 27. Creatinine | C4H7ON3 | 1.483 |
| 28. l-Serine | C3H7O3N | 1.419 |
| 29. l-Glutamine | C5H10O3N2 | 1.335 |
| 30. dl-Alanyl glycine | C5H10O3N2 | 1.460 |
| 31. Glycylglycine | C4H8N2O3 | 1.438 |
| 32. Alanine | C3H7NO2 | 1.450 |
| 33. Cysteine | C3H7NO2S | 1.402 |
| 34. Dimethyl sulfone | C2H6O2S | 1.509 |
| 35. d-Lactic acid | C3H6O3 | 1.593 |
| 36. Fumaric acid | C4H4O4 | 1.447 |
| 37. Guanine | C5H5N5O | 1.061 |
| 38. Gycine | C2H5NO2 | 1.379 |
| 39. Isoleucine | C6H13NO2 | 1.586 |
| 40. Leucine | C6H13NO2 | 1.586 |
| 41. l-Glutamic acid | C5H9NO4 | 1.279 |
| 42. 1-Hexadecanol | C16H34O | 1.864 |
| 43. Hypoxanthine | C5H4ON4 | 1.070 |
| 44. Glycolide | C4H4O4 | 1.354 |
| Plastic | ||
| 45. 1,3,5-Trioxane | C3H6O3 | 1.476 |
| 46. Benzophenone | C13H10O | 1.346 |
| 47. Biphenyl | C12H10 | 1.358 |
| 1.002 | 48. Hexachloroethane | C2Cl6 |
| 49. Diphenyl carbonate | C13H10O3 | 1.300 |
| 50. Diphenyl ether | C12H10O | 1.372 |
| 51. Diphenylcarbinol | C13H12O | 1.330 |
| 52. Polypropylene, isotatic | (C3H6)n | 1.662 |
| 53. Polypropylene, syndiotic | (C3H6)n | 1.798 |
| 54. Pyromellitic dianhydride | C10H2O6 | 1.087 |
| 55. Naphthalene | C10H8 | 1.306 |
| 56. Succinic acidic | C4H6O4 | 1.417 |
| 57. Cyanuric acid | C3H3N3O3 | 1.947 |
| 58. Acetamide | C2H5NO | 1.840 |
| 59. Durene | C10H14 | 1.166 |
| 60. Hexamethlenetetramine | C6H12N4 | 1.116 |
| 61. Triphenylene | C18H12 | 1.273 |
| 62. Hydroquinone | C6H602 | 1.182 |
| 63. Melamine | C3H6N6 | 1.251 |
| 64. Phthalic acid | C8H6O4 | 1.215 |
| 65. Phathalic anhydide | C8H4O3 | 1.405 |
| 66. Triethylenediamine | C6H12N2 | 1.947 |
| 67. 4,4′-diphenylmethane diisocyanate | C15H10N2O2 | 1.329 |
| 68. Polyisocyanurate | (C15H10N2O2)n | 1.175 |
| 69. Tridecanolactone | C13H24O2 | 1.893 |
| 70. Polytridecanolactone | (C13H24O2)n | 1.656 |
| 71. Polyvinylidene chloride | (C2H2Cl2)n | 0.894 |
| 72. Polyvinyl chloride | (C2H3Cl)n | 1.042 |
| 73. poly(1-butene), isotactic | (C4H8)n | 1.836 |
| 74. Polystyrene | (C8H8)n | 1.294 |
| Textile | ||
| 75. 3-Nitrobenzoic acid | C7H5NO4 | 1.227 |
| 76. 1,2-Diphenylethene | C14H12 | 1.39 |
| 77. Adipic acid | C6H10O4 | 1.504 |
| 78. 2-Methlnaphthalene | C11H10 | 1.547 |
| 79. Acenaphthene | C12H10 | 1.225 |
| 80. Anthracene | C14H10 | 1.164 |
| 81. 1,4-Benzoquinone | C6H4O2 | 1.506 |
| 82. Diphenylamine | C12H11N | 1.666 |
| 83. Pyrene | C16H10 | 1.112 |
| 84. Thiourea | CH4N2S | 1.523 |
| 85. Ammonium thiocyanate | CH4N2S | 1.842 |
| 86. 3-Nitroaniline | C6H6N2O2 | 1.276 |
| 87. Resorcinol | C6H6O2 | 1.341 |
| 88. Triphenylmethane | C19H16 | 1.277 |
| 89. Triphenylmethanol | C19H16O | 1.265 |
| 90. Isoquinoline | C9H7N | 1.324 |
| 91. Acridine | C13H9N | 1.161 |
| 92. 2-Nitrobenzoic acid | C7H5O4N | 1.247 |
| 93. 1,3-Phenylenediamine | C6H8N2 | 1.429 |
| 94. Dicyanodiamide | C2H4N4 | 1.538 |
| 95. ε-Caprolactam | C6H11NO | 1.531 |
| 96. Poly-ε-Caprolactam | (C6H11NO)n | 1.529 |
| 97. Polyglycolide | (C4H4O4)n | 1.304 |
| Wood | ||
| 98. l-Sorbose | C6H12O6 | 1.226 |
| 99. o-Cresol | C7H8O | 1.530 |
| 100. Oxalic acid | C2H2O4 | 1.220 |
| 101. p-Cresol | C7H8O | 1.547 |
| 102. Sucrose | C12H22O11 | 1.052 |
| 103. d-Mannitol | C6H14O6 | 1.309 |
| 104. Pentachlorophenol | C6HCl5O | 0.946 |
| 105. Galactose | C6H12O6 | 1.14 |
| 106. Phenol | C6H6O | 1.53 |
| 107. 2-Hydroxybenzoic acid | C7H6O3 | 1.29 |
| 108. Glucose | C6H12O6 | 1.161 |
| 109. Xylose | C5H10O5 | 0.956 |
| 110. 3-Hydroxybenzoic acid | C7H6O3 | 1.281 |
| 111. 4-Hydroxybenzoic acid | C7H6O3 | 1.272 |
| 112. Benzoic acid | C7H6O2 | 1.372 |
| 113. Catechol | C6H6O2 | 1.364 |
| 114. Lactose | C12H22O11 | 1.128 |
| 115. o-Hydroxybenzoic acid | C7H6O3 | 1.29 |
| 116. o-Hydroxybenzoic acid | C7H6O3 | 1.281 |
| 117. o-Hydroxybenzoic acid | C7H6O3 | 1.272 |
References
- Georgieva, K., and K. Varma. 1999. Municipal solid waste incineration. World Bank Technical Guidance Report. Washington D.C.
- Szargut, J., D. R. Morris, and F. R. Steward. 1988. Exergy analysis of thermal, chemical, and metallurgical processes. Hemisphere, New York, NY.
- Song, G., L. Shen, and J. Xiao. 2011. Estimating specific chemical exergy of biomass from basic analysis data. Ind. Eng. Chem. Res.50:9758–9766.
- Dincer, I., and M. A. Rosen. 2007. Exergy, energy, environment and sustainable development. Wiley, New York, NY.
- Stepanov, V. S.1995. Chemical energies and exergies of fuels. Energy20:235–242.
- Rant, Z.1961. Towards the estimation of specific exergy of fuels (in German). Allg. Wärmetech10:172–176.
- Szargut, J., and T. Styrylska. 1964. Approximate evaluation of the exergy of fuels (in German). Brennst. Wärme Kraft16:589–596.
- Eisermann, W., P. Johnson, and W. L. Conger. 1980. Estimating thermodynamic properties of coal, char, tar and ash. Fuel Process. Technol.3:39–53.
- Shieh, J. H., and L. T. Fan. 1982. Estimation of energy (enthalpy) and exergy (availability) contents in structurally complicated materials. Energy Sources6:1–46.
- Ikumi, S., C. D. Luo, and C. Y. Wen. 1982. Method of estimating entropies of coals and coal liquids. Can. J. Chem. Eng.60:551–555.
- Bilgen, S., and K. Kaygusuz. 2008. The calculation of the chemical exergies of coal-based fuels by using the higher heating values. Appl. Energy85:776–785.
- Song, G., J. Xiao, H. Zhao, and L. Shen. 2012. A unified correlation for estimating specific chemical exergy of solid and liquid fuels. Energy40:164–173.
- Bejan, A., G. Tsatsaronis, and M. Moran. 1996. Thermal design and optimization. John Wiley, New York, NY.
- Bilgen, S.2014. The estimation of chemical availability (Exergy) values for various types of coals in geographical regions of Turkey. Energy Sources36:830–842.
- Rivero, R., and M. Garfias. 2006. Standard chemical exergy of elements updated. Energy31:3310–3326.
- Kaygusuz, K.2009. Chemical exergies of some coals in Turkey. Energy Sources31:299–307.
- Standard thermodynamic properties of chemical substances. 2000. CRC Press. Available at http://www.update.uu.se/~jolkkonen/pdf/CRC_TD.pdf (accessed 3 July 2015).
- Kotas, T. J.1995. The exergy method of thermal plant analysis. Krieger Publishing Company, Malabar, FL.
- Channiwala, S. A., and P. P. Parikh. 2002. A unified correlation for estimating HHV of solid, liquid and gaseous fuels. Fuel81:1051–1063.
- Sheng, C., and J. L. T. Azevedo. 2005. Estimating the higher heating value of biomass fuels from basic analysis data. Biomass Bioenergy28:499–507.
- Zhou, H., A. Meng, Y. Long, Q. Li, and Y. Zhang. 2014. Classification and comparison of municipal solid waste based on thermochemical characteristics. J. Air Waste Manag. Assoc.64:597–616.
- Zhou, H., Y. Long, A. Meng, Q. Li, and Y. Zhang. 2015. Classification of municipal solid waste components for thermal conversion in waste-to-energy research. Fuel145:151–157.
- Kathiravale, S., M. N. M. Yunus, K. Sopian, A. H. Samsuddin, and R. A. Rahman. 2003. Modeling the heating value of municipal solid waste. Fuel82:1119–1125.
- Domalski, E. S., and E. D. Hearing. 1996. Heat capacities and entropies of organic compounds in the condensed phase. Volume III. J. Phys. Chem. Ref. Data25:1–525.
- Pandey, K. K.1999. A study of chemical structure of soft and hardwood and wood polymers by FTIR spectroscopy. J. Appl. Polym. Sci.17:1969–1975.
- Dietrich, F., and W. Gerd. 1983. Wood: chemistry, ultrastructure, reactions. De Gruyter, Berlin, pp. 66–181.
- Bledzki, A. K., V. E. Sperber, and O. Faruk. 2002. Natural wood and fiber reinforcement in polymers. Smithers Rapra, Shropshire, United Kingdom. pp. 4–18.
- Fundo, J. F., M. A. C. Quintas, and C. L. M. Silva. 2015. Molecular dynamics and structure in physical properties and stability of food systems. Food Eng. Rev.7:384–392.
- Wrolstad, R. E.2011. Food carbohydrate chemistry. Wiley-Blackwell, West Sussex, United Kingdom.
- Coultate, T. P.2002. Food: the chemistry of its components. Royal Society of Chemistry, Cambridge, United Kingdom, pp. 7–408.
- Brydson, J. A.1999. Plastics materials, 7th ed. Butterworth-Heinemann, Oxford, United Kingdom. pp. 205–813.
- Fan, Q.2005. Chemical testing of textile, 1st ed. Woodhead Publishing, Cambridge, England.
- Gordon, S., and Y. L. Hsieh. 2007. Cotton: science and technology. Woodhead Publishing, Cambridge, England.
- Simpson, R. B.2002. Rubber basics. Smithers Rapra, United Kingdom.
- Wang, S.2008. Application of solid ash based catalysts in heterogeneous catalysis. Environ. Sci. Technol.42:7055–7063.
- Song, G., L. Shen, J. Xiao, and L. Chen. 2013. Estimation of specific enthalpy and exergy of biomass and coal ash. Energy Sources35:809–816.
- Park, K., J. Hyun, S. Maken, S. Jang, and J. W. Park. 2005. Vitrification of municipal solid waste incinerator fly ash using Browns gas. Energy Fuels19:258–262.
- Hla, S. S., and D. Roberts. 2015. Characterisation of chemical composition and energy content of green waste and municipal solid waste from Greater Brisbane, Australia. Waste Manag.41:12–19.
- Xiao, G., M. Ni, Y. Chi, B. Jin, R. Xiao, Z. Zhong, et al. 2009. Gasification characteristics of MSW and an ANN prediction model. Waste Manage. (Oxford)29:240–244.
- Xiao, G., M. J. Ni, H. Huang, Y. Chi, R. Xiao, Z. P. Zhong, et al. 2007. Fluidized-bed pyrolysis of waste bamboo. J. Zhejiang Univ. Sci. A8:1495–1499.
- Meraz, L., A. Domínguez, I. Kornhauser, and F. Rojas. 2003. A thermochemical concept-based equation to estimate waste combustion enthalpy from elemental composition. Fuel82:1499–1507.
- Zheng, J., Y. Jin, Y. Chi, J. Wen, X. Jiang, and M. Ni2009. Pyrolysis characteristics of organic components of municipal solid waste at high heating rates. Waste Manage. (Oxford)29:1089–1094.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?