Abstract
Objective
Elastofibroma dorsi (ED) is a rare, benign, soft tissue tumor typically located between inferior corner of scapula and posterior chest wall causing mass, scapular snapping, and pain. When classic symptoms and localization are present, it is diagnosed without biopsy and treated with marginal resection. This study retrospectively analyzed patients operated on for ED to evaluate presenting symptoms, tumor size, complications, and clinical results, and to suggest optimal treatments.
Methods
This study included 51 patients who underwent surgery for ED in 2 different clinics between 2005 and 2015. Patient age, gender, profession, side affected, symptoms, average duration of symptoms, and tumor size were researched. Radiological examinations of patients were evaluated. Patients with lesions larger than 5 cm in size were operated on. Postoperative complications, recurrence, and functional results were evaluated using Constant score and compared to preoperative values.
Results
A total of 61 operated lesions of 51 patients clinically and radiologically diagnosed with ED were retrospectively evaluated. Average length of time patient experienced symptoms was 11.21 months. Lesions in 19 (37.2%) patients were bilateral, 10 of which were symptomatic and larger than 5 cm in size, meeting indication for surgery. Average lesion diameter was 8.7 cm. Average follow-up was 26.89 months. Average of preoperative Constant score of 67.28 subsequently increased to 92.88 (p < 0.05). Seroma and hematoma were observed in 11.5% of patients.
Conclusion
Generally, good clinical results can be obtained with marginal resection without requiring a biopsy, considering classic complaints and radiological appearance of ED.
Level of evidence
Level IV, Therapeutic study.
Keywords
Complications ; Elastofibroma dorsi ; Marginal resection
Elastofibroma dorsi (ED) is a rare, benign, connective tissue tumor that grows slowly. It is frequently located between inferior corner of scapula and posterior chest wall. ED was first defined by Jarvi and Saxen in 1961 and is frequently observed in middle-aged and older females.1 ; 2 The most frequently observed symptoms of this tumor, which is typically located adjacent to periosteum of sixth, seventh, and eighth ribs deep in the latissimus dorsi, rhomboids, or serratus anterior muscles, are swelling, pain during shoulder movements, and snapping of the scapula.
Although friction between chest wall and inferior corner of scapula and development of trauma is most often held responsible, the actual etiology of ED is still being discussed.3 A study of autopsies revealed elastofibroma-like changes at a rate of 81%, which suggested that ED may be a result of aging.4 The fact that approximately half of cases were bilateral and that 30% of the patients had ED history in their families suggested genetic cause for the disease.5 Hernandez et al found deoxyribonucleic acid (DNA) sequence losses in 1p, 13p, 19p, and 22q chromosomes.6 Studies have shown an increase in the number of DNA copies on long arm of X chromosome.7 Imanishi et al stated that transforming growth factor β and basic fibroblast growth factor may play a role in development of ED.8 Taşlı et al reported that lesion may originate from primitive dermal mesenchymal cells due to factor XIIIa and CD34 positivity.9 All these histological findings have revealed that ED is a real neoplastic process rather than a degenerative disease or a result of aging of connective tissue.
Surgical treatment of ED is usually performed because of symptoms such as increasing swelling, pain with shoulder movement or in supine position, or snapping of the scapula.3 It is not a well-demarcated lesion, and although symptomatic healing is observed in majority of patients after marginal resection, complications such as seroma and hematoma may be observed.
Orthopedic literature consists of small case series and case reports about treatment of this rare tumor. Although some authors suggest marginal resection to relieve symptoms, due to its rarity, optimal treatment approach and postoperative care has not been clearly determined.3 ; 10 This study retrospectively analyzed patients operated on for ED to evaluate presenting symptoms, tumor size, complications, and clinical results, and to suggest optimal treatment.
Patients and methods
With the permission of Çukurova University Faculty of Medicine ethical board, records of 61 surgically treated lesions of 51 patients diagnosed clinically and radiologically with ED at 2 dedicated tumor centers between 2005 and 2015 were retrospectively evaluated. Patient age, gender, profession, lesion side, symptoms, average duration of symptoms, and tumor size were analyzed, and radiological examinations were evaluated.
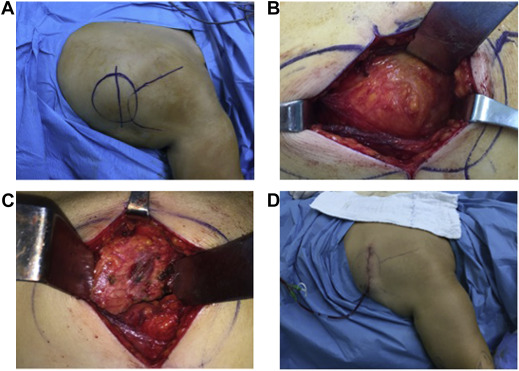
Surgical indications were defined as tumor larger than 5 cm in size and symptoms related to lesion. Marginal resection was performed by 2 senior orthopedic oncologists (authors 1 and 2) with the same exposure. Under general anesthesia, with patient in prone position, ipsilateral arm was draped freely to have better localization of lesion. Latissimus dorsi muscle was split with transverse incision over the lesion, and serratus anterior muscle was accessed. Hard, white tissue with uncertain margin that was firmly attached to serratus anterior and periosteum of ribs was separated using electrocautery, and marginal resection was performed. Suction drain was inserted after resection with careful control of hemorrhage (Fig. 1 ). Drain was kept in place for at least 48 h. Patients wore arm sling for 15 days after surgery. Subsequently, passive movements were initiated; active, full range of joint motion (ROM) was allowed in first postoperative month. Follow-up was conducted at 15 days, first month, third month, and at intervals of 3 months for the first 1.5 years after procedure. Recurrence was evaluated with clinical examination at each visit, followed by ultrasound (US) if there was suspected lesion.
|
|
|
Fig. 1. Surgical technique. (A) Mass is made clearly visible by abducting and elevating the arm in the prone position, and (B) is reached by passing through latissimus dorsi and serratus anterior muscles. (C) . Chest wall after resection. (D) Appearance after the wound was closed. |
Pre- and postoperative functional results were evaluated using Constant shoulder score, which is composed of 4 categories: pain, ability to perform activities of daily living, ROM, and strength of abduction. Both total score and category scores were evaluated statistically. Complications and recurrence rates were also analyzed.
Statistical analysis
Statistical analyses were performed using Statistical Package for Social Sciences software, version 16.0 (SPSS Inc., Chicago, IL, USA). Average and standard deviation were calculated for quantitative variables, and number of cases and rates were calculated for categorical variables. Differences between pre- and postoperative Constant score were analyzed using paired sample t-test. Chi-square test was used to investigate correlation between tumor size and complications. P value less than 0.05 was considered significant.
Results
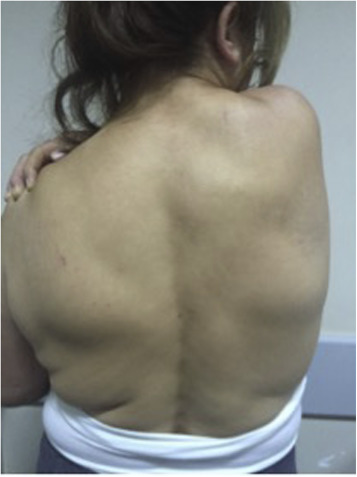
Study patients were 6 males (11.8%) and 45 females (88.2%). Average age was 55.33 ± 7.01 years (range: 41–69 years). Regarding professional occupation, 44 were housewives, 2 worked at desk jobs, and 5 were manual laborers. All of the lesions were located in inferior subscapular region, which is the typical location for ED. The most frequent presenting complaint was mass that could be clearly visualized with abduction and elevation of arm (90%) (Fig. 2 ), followed by pain (73.7%) during shoulder movement, and scapular snapping (65%). Average duration of symptoms was 11.21 ± 4.77 months (range: 4–26 months).
|
|
|
Fig. 2. Mass is clearly seen on the left inferior corner of the scapula with abduction and elevation of the arm. |
Lesions of 19 (37.2%) of the patients were determined to be bilateral; 10 had symptomatic lesions larger than 5 cm in size and underwent surgery. Thoracic or shoulder magnetic resonance imaging (MRI) was used to diagnose 58 lesions, and 3 patients (3 lesions) were diagnosed using thoracic computed tomography (CT). Of the lesions, 35 were on the right side and 26 were on the left. Average lesion diameter was 8.7 ± 2.09 cm (range: 5–13 cm). Average follow-up time was 26.89 ± 12.87 months (range: 6–60 months).
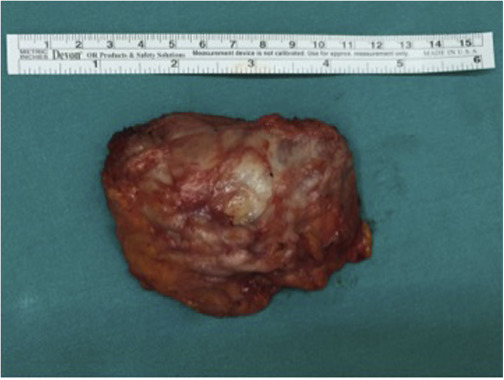
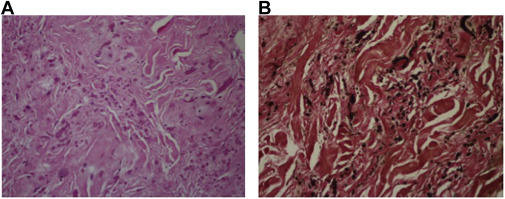
Macroscopically, ED is a hard tumor without a capsule and has uncertain boundaries that cannot be distinguished from the muscle and fat tissue around it (Fig. 3 ). Pathologically ED consists of abnormal elastic fibrils and fibroblasts spread in normal collagen fiber bundles (Fig. 4 ). None of the patients required blood transfusion.
|
|
|
Fig. 3. Macroscopic appearance of elastofibroma dorsi. |
|
|
|
Fig. 4. (A) Bent or globoid elastic fibers and rare fibroblasts are visible in imaging degenerated with eosinophilic thick collagen (H&E, ×200). (B) Extending and globoid elastic fibers observed using Verhoeff–van Giesons histochemical dye. |
Functional evaluation of shoulder was made using Constant score. Average Constant score improved from 67.28 ± 6.88 (range: 52–80) before the operation to 92.88 ± 4.72 (range: 78–100) at last follow-up (p < 0.05). Improvement in pain and activities of daily living categories were statistically significant (p < 0.05); no significant difference was found in ROM and abduction strength scores.
Recurrence was not observed in any of the patients. Seroma and hematoma were observed in 4 and 3 patients, respectively (11.5%). Six of these patients were treated only with aspiration and compressive dressing. One was re-operated on 15 days after initial operation and drainage was performed due to hematoma. There was no correlation between size of tumor and complications (p > 0.05).
Discussion
The literature about ED primarily consists of case reports and small series. The present study is 1 of the largest series with functional evaluation in orthopedic literature. Although ED is a rare, benign, soft tissue tumor, its actual incidence may be higher. Brander et al found incidence in normal population to be 2% in CT screenings.11 Rate of elastofibroma seen in the autopsy series was 11.2% in males and 24.4% in females older than 55 years of age.12 Majority of patients were housewives in the present series; 37.2% of cases showed bilateral ED, but only 19.6% underwent resection. Bilateral involvement of ED is reported between 12 to 73% of cases.13
Pain and snapping in scapula are the most frequent symptoms, accompanied by swelling. Hammoumi et al reported 93% pain, 51.3% functional limitation, and 31% clicking or lump in a series of 76 cases.14 Due to the variety of clinical signs, ED may be misdiagnosed as rotator cuff or cervical problem. Although in some cases it is detected during CT and MRI performed for another complaint, this deeply located lesion generally is not diagnosed unless it reaches a size larger than 5 cm. The sources of symptoms experienced at age when cervical vertebrae and shoulder problems are frequently observed should be evaluated carefully with a meticulous physical examination, and cases incidentally diagnosed with imaging methods should be separated.
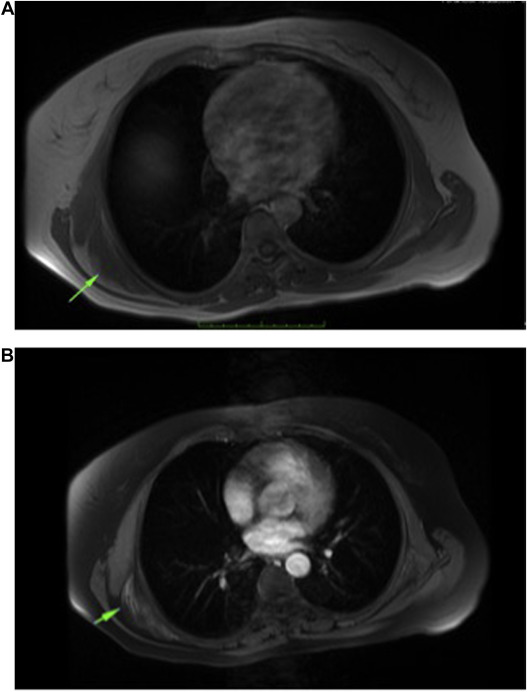
MRI was the most important diagnostic tool in this study (Fig. 5 ). Although lesion may be underdiagnosed radiologically, Malghem et al described a non-encapsulated lesion in typical location between fibrous tissue and fat tissue that showed an alternating pattern on MRI. While fibrous tissue showed low signal intensity in T1 and T2 sequences, fat tissue showed high signal intensity in T1 sequence. The areas that enhance or do not enhance can be seen in contrast series. Fat tissue is seen as lines in fibrous tissue parallel to chest wall.15 One study categorized ED into 3 subgroups based on MRI findings. Type A is inhomogeneous, fasciculated type; type B is inhomogeneous, non-specific type; and type 3 is isointense type with homogeneous muscle. Average positive predictive value and sensitivity were found to be 93.3% and 100%, respectively.16 US is 1 of the diagnostic modalities used in typical cases. A well-demarcated multilayer eco-signal is noted.17 Studies conducted using positron emission tomography/computed tomography (PET/CT) found that ED had a diffuse but slight 18F-fluorodeoxyglucose involvement. Studies found average standardized uptake value to be 2.29.18 Blumenkrantz found 1.66% ED in 1751 patients undergoing PET/CT.19
|
|
|
Fig. 5. (A) Axial T1 sequence shows isointense mass between scapula and thoracic wall. (B) Heterogenous contrast enhancement of lesion in T1 fat saturation sequence. |
The typical location of the tumor, long-term symptoms, and typical radiological findings, particularly MRI, are generally sufficient for ED diagnosis. Biopsy is generally not necessary with these findings. There is no evidence of malignant transformation of ED. Alberghini reported a case having high spindle cell sarcoma together with ED.20 Therefore, lipoma, hemangioma, and soft tissue sarcoma should be kept in mind for differential diagnosis in cases where there is rapid growth, significant contrast involvement, and atypical location.
The limit for surgical indication is still being discussed; however, generally accepted indications for resection are symptomatic cases with lesions larger than 5 cm in size, as in present study. Some authors suggest resection of all symptomatic and asymptomatic cases in order to confirm diagnosis pathologically.3 As it is a slow-growing tumor and diagnosis can be made with typical localization and radiology, surgery is not indicated in asymptomatic lesions. Resection with scapulothoracic joint arthroscopy has been described for selected cases.21
After marginal resection, all complaints resolved totally and functional result was statistically improved. The most dramatic difference was seen in pain and daily living activity sections of Constant score. Abat et al reported significant improvement in pain and shoulder antepulsion.22 Parrat et al stated that visual analogue scale score improved from 4.6 to 2.4 postoperatively and Instability Classification (Stanmore) score of 78.1 after follow-up of 4.6 years. Forward flexion was the only movement found to be improved after surgery.10
The most frequently reported complications after resection of ED are hematoma and seroma. Nanago et al reported instance of hematoma as high as 43% and found that tumor diameter was related to development of hematoma.3 A very meticulous control of hemorrhage and keeping drain in place for sufficient length of time after resection in event of excessive hemorrhage in the chest wall can decrease these complications.22 There is no consensus in the literature about rehabilitation protocol after surgery. Nagano recommends immobilization for 1 week.3 We started passive ROM exercises after 15 days, which may seem to be quite a long time, but immobilization can help healing of muscle reattachments and can stabilize flaps, thereby helping to reduce complications.
No recurrence was observed in this series. Nagamine reported 0.06% recurrence in his series of 170 cases, and Lucaca reported 4.5% recurrence in his series of 71 cases.23 ; 24
The retrospective nature, and lack of randomization and control group are limitations of this study; however, large number of cases and long follow-up period make this study valuable.
In conclusion, ED is a rare, slow-growing, soft tissue tumor typically located in scapulothoracic region at lower corner of scapula. Generally, good clinical results can be obtained using marginal resection without requiring a biopsy, considering classic complaints and radiological appearance of ED. Attention should be paid to complications such as hematoma and seroma. Resection decision should be made based on discussion with patient and thorough evaluation of symptoms of each case.
Conflicts of interest
None declared.
References
- 1 O. Jarvi, E. Saxen; Elastofibroma dorse; Acta Pathol Microbiol Scand Suppl, 51 (Suppl. 144) (1961), pp. 83–84
- 2 J. Nishio, T. Isayama, H. Iwasaki, M. Naito; Elastofibroma dorsi: diagnostic and therapeutic algorithm; J Shoulder Elb Surg, 21 (2012), pp. 77–81
- 3 S. Nagano, M. Yokouchi, T. Setoyama, et al.; Elastofibroma dorsi: surgical indications and complications of a rare soft tissue tumor; Mol Clin Oncol, 2 (2014), pp. 421–424
- 4 G.D. Giebel, E. Bierhoff, J. Vogel; Elastofibroma and preelastofibroma- a biopsy and autopsy study; Eur J Surg Oncol, 22 (1996), pp. 93–96
- 5 J.S. Kumaratilake, R. Krishman, J. Lomax-Smith, E.G. Cleary; Elastofibroma: disturbed elastic fibrillogenesis by periosteal-derived cells? An immunoelectron microscopic and in situ hybridization study; Hum Pathol, 22 (1991), pp. 1017–1029
- 6 J.L. Hernandez, J.O. Rodriguez-parets, J.M. Valero, et al.; High resolution genome-wide analysis of choromosomal alterations in elastofibroma; Virchows Arch, 456 (2010), pp. 681–687
- 7 J. Nishio, H. Iwasaki, Y. Ohjimi, et al.; Gain of Xq detected by comparative genomic hybridization in elastofibroma; Int J Mol Med, 10 (2002), pp. 277–280
- 8 Imanishi A, Imanishi H, Yoshida Y, Okabayashi A, Tateishi C, Ikushima H, et al. Upregulation of TGF-B1 and basic FGF in elastofibroma: an immunohistochemical analysis. Med Mol Morphol . http://dx.doi.org/10.1007/s00795-015-0126-z .
- 9 F. Taşlı, E. Vardar, A. Argon, et al.; Histochemical and immunohistochemical characteristics of elastofibromas; Pol J Pathol, 65 (2) (2014), pp. 120–124
- 10 M.T.R. Parratt, J.R. Donaldson, A.M. Flanagan, et al.; Elastofibroma dorsi: management, outcome and review of the literature; J Bone Jt Surg Br, 92-B (2010), pp. 262–266
- 11 E.A. Brandser, J.C. Goree, G.Y. El-Khoury; Elastofibroma dorsi: prevelance in an elderly patient population as revealed by CT; Am J Roentgenol, 171 (1998), pp. 977–980
- 12 O.H. Jarvi, P.H. Lansimies; Subclinical elastofibromas in the scapular region in autopsy series; Acta Pathol Microbiol Scand A, 83 (1975), pp. 87–108
- 13 H. Pilge, T. Hesper, B.M. Holzapfel, P.M. Prodinger, M. Straub, R. Krauspe; Elastofibroma: clinical results after resection of a rare tumor entity; Orthop Rev, 6 (2014), pp. 5329–5333
- 14 M. El Hammoumi, A. Qtaibi, A. Arsalane, F. El Oueriachi, E.H. Kabiri; Elastofibroma dorsi: clinicopathological analysis of 67 cases; Korean J Thorac Cardiovasc Surg, 47 (2014), pp. 111–116
- 15 J. Malghem, V. Baudrez, F. Lecouvet, C. Lebon, B. Maldague, B. Vande Berg; Imaging study findings in elastofibroma dorsi; Jt Bone Spine, 71 (2004), pp. 536–541
- 16 F. Tamimi Marino, P. Sesma Solis, A. Perez Lara, J. Martinez Malo, M.L. Vazguez, F. Tamimi; Sensitivity and positive predictive value of magnetic resonance imaging in the diagnosis of elastofibroma dorsi: review of fourteen cases; J Shoulder Elb Surg, 22 (1) (2013), pp. 57–63
- 17 R. Haykir, S. Karaköse, A. Karabacakoğlu; Elastofibroma dorsi: typical radiological features; Australas Radiol, 51 (2007), pp. 95–97
- 18 N. Fang, Y. Wang, L. Zeng, et al.; Characteristics of elastofibroma dorsi on PET/CT imaging with 18F-FDG; Clin Imaging, 40 (2016), pp. 110–113
- 19 Y. Blumenkrantz, G.L. Bruno, C.J. Gonzales, M. Namias, A.R. Osorio, P. Parma; Characterization of elastofibroma dorsi with 18FDG PET/CT: a retrospective study; Rev Esp Med Nucl, 30 (2011), pp. 342–345
- 20 M. Alberghini, P. Bacchini, G. Pignatti, et al.; Histochemical and ultrastructural study of an elastofibroma dorsi coexisting with a high grade spindle cell sarcoma; Eur J Histochem, 48 (2004), pp. 173–178
- 21 G. Cakmak, T. Ergun, M.Ş. Şahin; Arthroscopic excision of elastofibroma dorsi at scapulothoracic joint: a surgical technique; Eklem Hastalık Cerrahisi, 25 (2) (2014), pp. 117–120 [Article in Turkish]
- 22 F. Abat, C. Alverez, L. Trullols, A. Peiro, S. Bague, I. Gracia; Elastofibroma dorsi: a 7-year follow-up of 37 cases and a review of the literature; Rev Esp Cir Ortop Traumatol, 56 (2012), pp. 295–300
- 23 N. Nagamine, Y. Nohara, E. Ito; Elastofibroma in Okinawa. A clinicopathologic study of 170 cases; Cancer, 50 (9) (1982), pp. 1794–1805
- 24 F. Lococo, A. Cesario, F. Mattei, et al.; Elastofibroma dorsi: clinicopathological analysis of 71 cases; Thorac Cardiovasc Surg, 61 (3) (2013), pp. 215–222
Document information
Published on 31/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?