Abstract
Background
Many of the factors that are known to alter left atrial (LA) contractility are present in patients with ischemic cardiomyopathy (ICM). Nevertheless, preservation of LA contractile function in this group of patients was reported in previous studies. The aim of this study was to assess the changes in LA size, geometry and contractile function in ICM.
Methods and results
60 subjects (age: 49 ± 12 years, 53% males) in sinus rhythm undergoing coronary angiography (CA), were enrolled in this study; 15 subjects as a control group, 30 patients with reduced ejection fraction (EF) and significant coronary artery disease; as the ICM group, and 15 with reduced EF and normal CA; as the dilated cardiomyopathy (DCM) group.
LA dimensions, volume, active emptying fraction (ACTEF) and eccentricity index (LAEi) as well as late diastolic velocities of the mitral annulus (a′) and LA free wall (A3) were measured.
Compared with the control group, ICM patients had larger LA volume and reduced ACTEF, a′ and A3, with no significant difference between patients with ICM and DCM in any of these parameters. LA eccentricity was, non-significantly, higher in both cardiomyopathy groups than in the control group.
Conclusions
Patients with ICM have increased LA volume and reduced LA contractile function in comparison with normal controls. LA enlargement, LA contractile dysfunction and LA geometric changes in ICM are similar to that occurring in DCM. LA size, contractile function and eccentricity may not be reliable in differentiating ischemic from idiopathic dilated cardiomyopathy.
Keywords
Left atrial function;Atrial eccentricity;Heart failure;Echocardiography;Ischemic cardiomyopathy
1. Introduction
Left atrial (LA) size and function are powerful predictors of outcome in heart failure (HF) patients with predominantly impaired systolic function, adding important clinical information, independent of any of its determinants [1]; [2] ; [3]. In addition to its prognostic value, LA size and function have been linked to exercise capacity and hospitalizations in patients with HF [3] ; [4]. These findings have drawn attention to the importance of LA size and function in patients with systolic heart failure.
Histological evidence of a high degree of fibrosis in the LA was found in patients with idiopathic dilated cardiomyopathy (DCM) but not in patients with old myocardial infarction despite similar degrees of left ventricular (LV) dysfunction. These findings suggested that atrial fibrosis in these patients may not have been related to mechanical overload of the left atrium but rather to LA involvement in the myopathic process [5]. These findings were further emphasized in a number of observational studies that reported LA contractile dysfunction in DCM and preservation in ischemic cardiomyopathy (ICM) [4]; [6] ; [7].
LA contractile dysfunction was, however, demonstrable in patients with coronary artery disease (CAD) even when the LV systolic function was preserved and LA was not enlarged [8].
With the progression of myocardial ischemia, and as the LV systolic and diastolic functions deteriorate, poor LV-ejection fraction (EF) and the presence of severe diastolic dysfunction causing a restrictive LV-filling pattern (RFP) become the most important determinants of LA contractile function in CAD patients [9]. LA contractile function is, thus, expected to be altered in patients with ICM (where LV systolic function is impaired and severe diastolic dysfunction {RFP} is common). We conducted this study primarily to assess LA size, geometry and contractile function in patients with ICM.
2. Methods
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the institutional review board. Sixty patients referred to Bab El-Sha'rya University Hospital for diagnostic coronary angiography (CA) and transthoracic echocardiography between May 2011 and June 2012, were enrolled. All patients provided written informed consent.
There were 30 patients with ICM and 15 patients with DCM. ICM was defined as a reduced EF (< 45%) and CA showing significant CAD proportionate with the degree of LV dysfunction (multivessel, left main or proximal left anterior descending artery stenosis), previous myocardial infarction or coronary revascularization [10]. DCM patients had an EF < 45% and normal CA.
In addition, 15 control subjects with normal EF, normal diastolic function and normal CA were included.
The exclusion criteria were: 1) atrial fibrillation; 2) conduction defects; 3) significant organic mitral or aortic valve disease and 4) a recent myocardial infarction (< 1 month).
2.1. Echocardiography
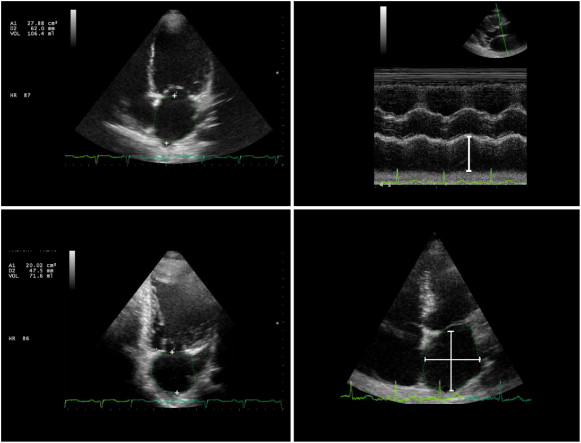
All patients were imaged in the left lateral position using an Esaote My Lab 5 Gold ultrasound system. Left atrial maximum volume (LA Vmax) was measured at LV end systole, LA minimal volume (LA Vmin) at LV end diastole and LA volume at the onset of atrial contraction (Vp) at the onset of electrocardiographic P wave from the apical four-chamber and apical two-chamber views using the biplane area–length method (Fig. 1) [11]. LA active emptying fraction ACTEF equals the difference between Vp and Vmin, divided by Vp i.e. ACTEF = (Vp-Vmin)/Vp [12] ; [13].
|
|
|
Fig. 1. Left; left atrial volume measured using biplane (apical four, upper, and two chamber, lower, views) area–length method. Right; left atrial antero-posterior (upper), superior–inferior and medio-lateral (lower) diameters. |
LA antero-posterior dimension (D1) was measured at end systole in the parasternal long axis view from the trailing edge of the posterior aortic wall to the leading edge of the posterior LA [11]. The superior–inferior (D2) and medio-lateral dimensions (D3) were measured in the apical four chamber view at end systole (Fig. 1). We used left atrial eccentricity index (LAEi = {2 × D2} / {D1 + D3}) to assess the changes in the LA shape [14].
Tissue Doppler velocities were measured in the apical four-chamber view at the septal side of the mitral annulus and mid-segment of LA free wall, using pulsed wave tissue Doppler. Peak velocity of septal mitral annulus at late diastole (a′) [15]; [16] ; [17] and atrial contraction peak velocity at the mid segment of the LA lateral wall (A3) [18] ; [19] were measured and used as markers of LA contractility.
To measure septal a′, the sample volume was placed on the atrial side of the mitral annulus at the basal inter-atrial septum from the apical four-chamber view [12]. To measure A3, sample volume of 4 mm was put at the mid segment of the LA lateral wall in apical four-chamber view. Special care was taken to place the sample volume away from the pulmonary vein entrance [18].
Left ventricular end-diastolic and end-systolic dimensions were measured and fractional shortening was calculated from the parasternal long axis view. Left ventricular end-diastolic and end-systolic volumes were measured and EF was calculated from the apical four-chamber and two-chamber views using modified biplane Simpsons method [11].
Mitral E-wave and A-wave velocities, E/A ratio, and E-wave deceleration time (DT) were also measured using pulsed wave Doppler with the sample volume put at the tips of the mitral valve leaflets. E-wave DT was measured as the interval in milliseconds (ms), from the peak of E-wave to an extrapolation of the deceleration to 0 m/s. Severe diastolic dysfunction/restrictive LV-filling was defined by the presence of the following criteria: E/A ratio > 2, DT < 160 ms and E/e′ > 15 [20].
Mitral regurgitation (MR) was semi-quantitatively assessed by color flow Doppler echocardiography. Four grades of regurgitant jet area were determined (1 = regurgitant jet area < 1.5 cm2; 2 = regurgitant jet area 1.5–3 cm2, 3 = regurgitant jet area 3–8 cm2 and 4 = regurgitant jet area > 8 cm2) [21].
2.2. Statistical analysis
Data are presented as the mean value ± standard deviation (SD) for continuous variables and as numbers and frequency percentages for categorical variables. Comparisons of continuous variables between groups were made using the unpaired t test, whereas categorical variables were compared by the Chi-square and Fishers exact tests.
Correlation between the LA volume and ACTEF was tested by Pearsons correlation coefficient.
The study had a power of 80% to detect a 15% difference in ACTEF between the groups with a SD of 10%, at α level of 0.05. A two-tailed probability (P) value of ≤ 0.05 was used to define a significant result.
3. Results
3.1. Baseline characteristics
ICM patients were older than patients with DCM and control subjects. The proportion of males was higher in ICM and DCM groups than in the control group. The prevalence of HTN was similar in the three groups.
Compared with the control group, patients with ICM and DCM had significantly higher left ventricular end-diastolic dimension (LVEDD), MR grade and prevalence of severe diastolic dysfunction. LV ejection fraction (EF), septal mitral annular e′ velocity and E-wave deceleration time (DT) were significantly lower in ICM and DCM groups than in the control group. Table 1 shows the baseline clinical and echocardiographic characteristics of the patients and controls.
| ICM (n = 30) | DCM (n = 15) | Control (n = 15) | |
|---|---|---|---|
| Age (year) | 56 ± 8⁎§ | 40 ± 10 | 44 ± 13 |
| Male, n (%) | 22 (73)⁎ | 7 (47) | 3 (20) |
| Hypertension, n (%) | 16 (53) | 6 (40) | 5 (33) |
| LVEDD (cm) | 6.1 ± 0.8⁎ | 6.3 ± 0.9 | 4.8 ± 0.6 |
| LVEF (%) | 34 ± 8⁎ | 31 ± 8 | 62 ± 5 |
| Septal e′ (cm/s) | 6.5 ± 3⁎ | 5 ± 3 | 10.6 ± 3 |
| DT (ms) | 154 ± 59⁎ | 180 ± 70 | 212 ± 35 |
| Severe diastolic dysfunction, n (%) | 11 (35)⁎ | 4 (27) | 0 |
| MR grade | 1.8 ± 1⁎ | 1.9 ± 1.5 | 0.8 ± 0.8 |
Data are presented as mean ± SD or n (%).
LVEDD = left ventricular end-diastolic dimension, LVEF = left ventricular ejection fraction, e′ = mitral annular early diastolic peak velocity, DT = E-wave deceleration time, MR = mitral regurgitation, ICM = ischemic cardiomyopathy, and DCM = dilated cardiomyopathy.
⁎. P < 0.05; ICM vs control.
§. P < 0.05; ICM vs DCM.
3.2. LA volume and emptying fraction
LA maximum volume was larger in ICM patients than in controls (Table 2, P = 0.0001), but there was no significant difference between ICM and DCM groups (P = 0.41). LA active emptying fraction was lower in ICM patients than in the control group (P = 0.0046), but without significant difference between patients with ICM and DCM (P = 0.66).
| ICM (n = 30) | DCM (n = 15) | Control (n = 15) | |
|---|---|---|---|
| LAVmax (ml) | 78 ± 25⁎ | 85 ± 34 | 48 ± 13 |
| ACTEF (%) | 29 ± 13⁎ | 27 ± 10 | 41 ± 10 |
| Septal a′ (cm/s) | 8 ± 2.9⁎ | 7.6 ± 2.4 | 10.7 ± 2.9 |
| LA3 (cm/s) | 10.4 ± 5.1⁎ | 10.5 ± 4.3 | 17.3 ± 5.9 |
| LAD1 (cm) | 4.1 ± 0.7⁎ | 3.9 ± 0.8 | 3.2 ± 0.4 |
| LAD2 (cm) | 5.4 ± 0.9⁎ | 5.7 ± 0.6 | 4.6 ± 1.1 |
| LAD3 (cm) | 4.6 ± 0.9 | 4.6 ± 0.8 | 4.3 ± 0.7 |
| LAEi | 1.26 ± 0.2 | 1.36 ± 0.1 | 1.22 ± 0.3 |
Data are presented as mean ± SD.
LAVmax = left atrial maximum volume, ACTEF = left atrial active emptying fraction, a′ = mitral annular late diastolic peak velocity, LA3 = left atrial free wall velocity at atrial contraction, LAD1 = left atrial antero-posterior dimension, LAD2 = left atrial superior–inferior dimension, LAD3 = left atrial medio-lateral dimension, LAEi = left atrial eccentricity index, ICM = ischemic cardiomyopathy, and DCM = dilated cardiomyopathy.
⁎P < 0.05; ICM vs control.
3.3. Mitral annulus and LA free wall velocities at atrial contraction (Septal a′ and LA3)
As shown in Table 2, ICM patients had lower septal a′ and A3 than controls (P = 0.005 and 0.003, respectively), but no significant difference was noted between ICM and DCM patients (P = 0.67 and 0.93, respectively).
3.4. LA geometry
Left atrial antero-posterior and the superior–inferior dimensions were longer in ICM patients than in the control group (Table 2, P = 0.0001 and 0.016, respectively). However, medio-lateral dimension showed no significant difference than in controls (P = 0.25). Similar geometric changes were noted in DCM patients (P values — ICM vs. DCM — for D1, D2 and D3 = 0.48, 0.24, 0.9). Compared with the control group, LA geometry was more elongated in the 2 cardiomyopathy groups, yet such increase in LAEi did not reach statistical significance even in the more elongated DCM group (P value for LAEi — DCM vs. control — = 0.27).
3.5. Comparison between cardiomyopathy patients with and without restrictive LV filling pattern – RFP – (Table 3)
When cardiomyopathy patients – from both ICM and DCM groups – with RFP (18 patients) were compared with cardiomyopathy patients with less severe diastolic dysfunction (non-RFP, 27 patients); no significant difference was noted in LVEF or MR (P = 0.2 and 0.15, respectively). However, those with RFP had a larger LA Vmax (P = 0.02) and a more depressed LA pump function than non-RFP patients (ACTEF: 25 ± 12 vs 33 ± 13%, P = 0.057; septal a′: 6.4 ± 2.6 vs 9.2 ± 2.1 cm/s, P = 0.001; A3: 8.1 ± 4 vs 12.4 ± 4.6 cm/s, P = 0.006).
| RFP (n = 18) | Non-RFP (n = 27) | P value | |
|---|---|---|---|
| LVEF (%) | 32 ± 9 | 35 ± 7 | 0.2 |
| MR grade | 2.1 ± 1.2 | 1.6 ± 1.2 | 0.15 |
| LAVmax (ml) | 89 ± 28 | 69 ± 24 | 0.02 |
| ACTEF (%) | 25 ± 12 | 33 ± 13 | 0.057 |
| Septal a′ (cm/s) | 6.4 ± 2.6 | 9.2 ± 2.1 | 0.001 |
| LA3 (cm/s) | 8.1 ± 4 | 12.4 ± 4.6 | 0.006 |
Data are presented as mean ± SD.
RFP = restrictive filling pattern, LVEF = left ventricular ejection fraction, MR = mitral regurgitation, LAVmax = left atrial maximum volume, ACTEF = left atrial active emptying fraction, septal a′ = septal mitral annulus peak velocity at late diastole, and LA3 = left atrial free wall velocity at atrial contraction.
4. Discussion
The present study showed that LA remodeling is frequent in patients with ICM. ICM patients have a significantly enlarged LA and a significantly reduced LA contractile function. This conclusion is supported by the concordance in three indices of LA booster pump function, namely; LA active emptying fraction, mitral annular and LA free wall velocities at atrial contraction. On the other hand, LA volume, geometry and atrial pump function, were not significantly different between ICM and DCM patients.
4.1. LA function in cardiomyopathy with impaired systolic function
Idiopathic dilated cardiomyopathy (DCM) is a diffuse process. Cardiomyocytes of both ventricles are involved and atrial function is also decreased. The typical gross pathologic picture is that of 4-chamber enlargement [22]. Unlike DCM, ischemic heart disease primarily affects the left ventricle. This key pathologic feature distinguishing ICM from DCM was further emphasized in a number of observational studies. Right ventricular function was significantly better in patients with ischemic cardiomyopathy as compared to patients with dilated cardiomyopathy in a magnetic resonance study [23].
Likewise, LA contractile function (estimated by active emptying fraction [7] and systolic deformation [4]) was shown to be more depressed in DCM than in ICM patients. Some authors concluded that LA contractility is preserved in ICM patients, showing no measurable impairment compared with normal controls [7].
The current study, however, showed that LA pump function is depressed in patients with ICM. It is likely that increased LA afterload, because of the elevated LV diastolic pressure [24], plays a major role in this process. Studies in both animals and humans have shown that LA size increases with LA pressure overload, with an associated initial gain in contractile shortening. However, with progressive dilatation of the LA which eventually leads to a threshold fiber length, atrial shortening and contractility begin to decline [12].
Impairment of LV diastolic performance starts early in CAD and progresses as negative LV remodeling ensues [25].
4.2. LA function in ischemic heart disease
Atrial contractile dysfunction appears early in ischemic heart disease (IHD), irrespective of previous myocardial infarction, co-existing systolic dysfunction, or severity of diastolic dysfunction [26]; [9] ; [8]. Yu et al. found that the peak regional atrial contraction velocities at atrial systole (VAa) measured at mid-level of the inter-atrial septum and the lateral LA in the apical four chamber view were drastically reduced in patients with IHD when compared with normal controls. Furthermore, each percent increase in ejection fraction correlated with an increase in VAa in LA by 0.06 cm/s, while the occurrence of a restrictive filling pattern was associated with a reduction of VAa in LA by 3.17 cm/s. A poor LV-ejection fraction and the presence of severe diastolic dysfunction (restrictive LV-filling pattern) were the most important determinants of LA contractile dysfunction in IHD [9].
Left ventricular diastolic dysfunction precedes systolic wall motion abnormalities in the ischemic cascade [25]. LA structural and functional remodeling have been proposed as a barometer of diastolic burden, and this, at least partially, explains the causal link between IHD and LA dilatation and dysfunction.
4.3. LA size and function as markers of LV properties
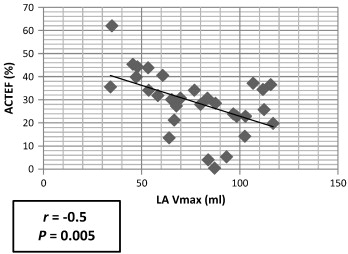
Left atrial volume, function and geometry have emerged as a comprehensive marker of left ventricular properties. LV mass, dimension, systolic and diastolic function were proved to be independent determinants of LA volume [2] ; [27]. LA volume has,also been identified as a major determinant of LA contractile function [27]. In our cohort of ICM patients, ACTEF was negatively correlated with LA Vmax (Fig. 2, r = − 0.5, P = 0.005).
|
|
|
Fig. 2. Correlation plot between left atrial maximum volume (LA Vmax) and left atrial active emptying fraction (ACTEF) in ischemic cardiomyopathy patients. |
Patients with impaired LV diastolic performance have increased LA volumes and decreased LA contractility that parallel the severity of diastolic dysfunction, such that patients with severe diastolic dysfunction (restrictive LV-filling pattern) have the largest atrial volumes and the worst LA booster pump function [28]; [29] ; [30].
In our cohort of cardiomyopathy patients, severe diastolic dysfunction was found in 18/45 patients (40%). This high prevalence of restrictive LV-filling was also noted in other observational studies (Table 4).
| Number of patients and etiology of cardiomyopathy included in the study | % of RFP | |
|---|---|---|
| Rossi et al. [2] | 337 (75% ischemic and 25% idiopathic) | 22% |
| Pratali et al. [31] | 116 (idiopathic) | 32% |
| Ito et al. [30] | 41 (mainly, idiopathic) | 32% |
| Yong et al. [32] | 40 (ischemic) | 48% |
RFP = restrictive filling pattern of left ventricular filling.
The high prevalence of severe diastolic dysfunction and the “elongated” geometry of the left atrium in cardiomyopathy patients suggested a major role of LV filling pattern on LA properties in this group of patients [14]. In order to test this assumption, our 45 cardiomyopathy patients (regardless of etiology) were divided into two groups according to the presence or absence of severe diastolic dysfunction (RFP). Both groups were comparable regarding LV EF and MR grade (Table 3).
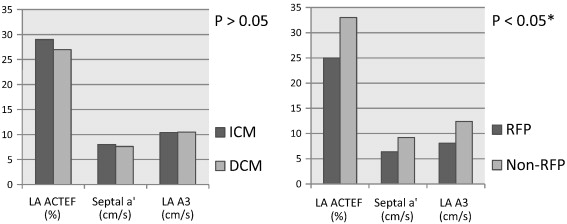
Categorizing patients with cardiomyopathy and reduced ejection fraction according to the severity of diastolic dysfunction, rather than according to etiology, identified two subgroups with significantly different LA properties, with larger LA size and more depressed contractile function in those with restrictive LV filling (Fig. 3).
|
|
|
Fig. 3. Ischemic cardiomyopathy (ICM) and dilated cardiomyopathy (DCM) did not differ significantly in left atrial active emptying fraction (LA ACTEF), septal a′ or left atrial late diastolic velocity (LA A3). On the other hand, cardiomyopathy patients with restrictive filling pattern (RFP) had lower indices of LA contractility than those without RFP. *RFP vs Non-RFP; P value for ACTEF was 0.057. P value for septal a′ and LA A3 was < 0.05. |
5. Limitations
Small number of patients is a limitation, but the study had adequate power to detect at least a 15% difference in the primary parameter of interest, ACTEF, between the compared groups. The main finding of this study, that could be concluded upon, was that the LA pump function is significantly reduced in ICM patients compared with control subjects. Nevertheless, larger sample size may detect smaller differences (< 15%) between ICM and DCM groups in LA pump function parameters and between cardiomyopathy patients and controls in LA eccentricity. The use of multiple comparisons among several patients' groups is another downside. Given the small sample size, additional studies are recommended to examine LA contractility, particularly using novel echocardiographic markers of LA deformation, in a larger population.
Patients with ICM were significantly older than the other groups. However, it has been shown that aging is associated with an increase in active atrial contraction in response to increased LV stiffness [12] ; [33]. So, an even worse LA contractility in ICM group is expected if patients in this group were younger. Male gender was more found in the two cardiomyopathy groups than in controls. LA size tends to be larger in men than women, but this difference can be attributed to the variations in relative weight and body size [34]. When adjusted for atrial size, there are no sex related differences in LA function [35].
About one third of control subjects were hypertensive. This was inevitable in a cohort of patients referred for invasive coronary angiography. Most of these patients had a risk profile that justified invasive evaluation of their symptoms. However, those with any echocardiographic sign of diastolic or systolic dysfunction were excluded.
LA volume was not indexed to BSA. LA volume index is a more accurate measure of LA size [36].
The study did not investigate the molecular mechanisms that account for the changes in LA function, and additional research can shed light on this area.
6. Conclusion
LA is enlarged and its contractility is reduced in patients with ICM. The geometric and functional changes observed in left atria of patients with ICM are similar to the changes observed in patients with DCM. These findings contrast to previous reports that pointed to LA contractile function preservation in patients with ICM.
Larger LA with more depressed contractile function may indicate worse LV diastolic performance in patients with LV systolic dysfunction rather than indicating a certain etiology of cardiomyopathy.
Disclosures
The authors report no relationships that could be construed as a conflict of interest.
References
- [1] A. Rossi, P.L. Temporelli, M. Quintana, F.L. Dini, S. Ghio, G.S. Hillis, et al.; Independent Relationship of Left Atrial Size and Mortality in Patients with Heart Failure: an Individual Patient Meta-Analysis of Longitudinal Data (MeRGE Heart Failure); Eur J Heart Fail, 8 (10) (2009), pp. 929–936
- [2] A. Rossi, M. Cicoira, L. Zanolla, R. Sandrini, G. Golia, P. Zardini, et al.; Determinants and Prognostic Value of Left Atrial Volume in Patients With Dilated Cardiomyopathy; J Am Coll Cardiol, 40 (8) (2002), pp. 1425–1430
- [3] S.-H. Hsiao, K.-R. Chiou; Left atrial expansion index predicts all-cause mortality and heart failure admissions in dyspnoea; Eur J Heart Fail, 15 (11) (2013), pp. 1245–1252
- [4] A. D'Andrea, P. Caso, S. Romano, R. Scarafile, S. Cuomo, G. Salerno, et al.; Association between left atrial myocardial function and exercise capacity in patients with either idiopathic or ischemic dilated cardiomyopathy: a two-dimensional speckle strain study; Int J Cardiol, 132 (3) (2009), pp. 354–363
- [5] K. Ohtani, C. Yutani, S. Nagata, Y. Koretsune, M. Hori, T. Kamada; High Prevalence of Atrial Fibrosis in Patients With Dilated Cardiomyopathy; J Am Coll Cardiol, 25 (5) (1995), pp. 1162–1169
- [6] Y. Wang, K. Takenaka, Y. Kuwada, M. Sonoda, T. Igarashi, F. Watanabe, et al.; Left Atrial Booster Pump Function in Patients with Ischemic Cardiomyopathy and Idiopathic Dilated Cardiomyopathy; J Am Coll Cardiol, 25 (2s1) (1995), p. 165A
- [7] F. Triposkiadis, I. Moyssakis, L. Hadjinikolaou, T. Makris, H. Zioris, A. Hatzizaharias, et al.; Left atrial systolic function is depressed in idiopathic and preserved in ischemic dilated cardiomyopathy; Eur J Clin Invest, 29 (11) (1999), pp. 905–912
- [8] Y.Y. Liu, M.X. Xie, J.F. Xu, X.F. Wang, Q. Lv, X.F. Lu, et al.; Evaluation of Left Atrial Function in Patients with Coronary Artery Disease by Two-Dimensional Strain and Strain Rate Imaging; Echocardiography, 28 (10) (2011), pp. 1095–1103
- [9] C.M. Yu, J.W. Fung, Q. Zhang, L.C. Kum, H. Lin, G.W. Yip, et al.; Tissue Doppler echocardiographic evidence of atrial mechanical dysfunction in coronary artery disease; Int J Cardiol, 105 (2) (2005), pp. 178–185
- [10] G.M. Felker, L.K. Shaw, C.M. O'Connor; A standardized definition of ischemic cardiomyopathy for use in clinical research; J Am Coll Cardiol, 39 (2) (2002), pp. 210–218
- [11] R.M. Lang, M. Bierig, R.B. Devereux, F.A. Flachskampf, E. Foster, P.A. Pellikka, et al.; Recommendations for chamber quantification; Eur J Echocardiogr, 7 (2) (2006), pp. 79–108
- [12] G.G. Blume, C.J. Mcleod, M.E. Barnes, J.B. Seward, P.A. Pellikka, P.M. Bastiansen, et al.; Left atrial function: physiology, assessment, and clinical implications; Eur J Echocardiogr, 12 (6) (2011), pp. 421–430
- [13] J.F. Ren, M.N. Kotler, N.L. DePace, G.S. Mintz, D. Kimbiris, P. Kalman, et al.; Two-dimensional echocardiographic determination of left atrial emptying, A noninvasive index in quantifying the degree of non-rheumatic mitral regurgitation; J Am Coll Cardiol, 2 (4) (1983), pp. 729–736
- [14] M. Naito, S. Nakao, A. Goda, M. Yuba, Y. Naito, M. Shimizu, et al.; Value of the measurements of left atrial geometry in patients with normalized or restrictive mitral flow velocity patterns; J Echocardiogr, 3 (3) (2005), pp. 109–117
- [15] B. Hesse, S.U. Schuele, M. Thamilasaran, J. Thomas, L. Rodriguez; A rapid method to quantify left atrial contractile function: Doppler tissue imaging of the mitral annulus during atrial systole; Eur J Echocardiogr, 5 (1) (2004), pp. 86–92
- [16] K.K. Poh, M.Y. Chan, H. Yang, Q.W. Yong, Y.H. Chan, L.H. Ling; Prognostication of valvular aortic stenosis using tissue Doppler echocardiography: underappreciation of late diastolic mitral annular velocity; J Am Soc Echocardiogr, 21 (5) (2008), pp. 475–481
- [17] R. Faludi, L. Tóth, L. Pótó, A. Cziráki, T. Simor, L. Papp; Correlation of B-type natriuretic peptide (NT-proBNP) levels and conventional and tissue Doppler echocardiographic parameters of left ventricular diastolic function in patients with hypertrophic cardiomyopathy; Orv Hetil, 146 (1) (2005), pp. 23–26
- [18] Y.T. Liu, R.J. Li, F. Fang, Q. Zhang, B.P. Yan, Y.Y. Lam, et al.; Left atrial function assessed by tissue Doppler imaging as a new predictor of cardiac events after non-ST-elevation acute coronary syndrome; Echocardiography, 29 (7) (2012), pp. 785–792
- [19] M. Perez-Paredes, M. Gonzalves, J.A. Ruiz Ros, D.M. Giménez, A. Carnero, A. Carrillo, et al.; Assessment of left atrial wall velocities by pulsed wave tissue Doppler imaging A new approach to the study of atrial function; Rev Esp Cardiol, 57 (11) (2004), pp. 1059–1065
- [20] S.F. Nagueh, C.P. Appleton, T.C. Gillebert, P.N. Marino, J.K. Oh, O.A. Smiseth, et al.; Recommendations for the evaluation of left ventricular diastolic function by echocardiography; J Am Soc Echocardiogr, 22 (2) (2009), pp. 107–133
- [21] K. Yoshida, J. Yoshikawa, Y. Yamaura, T. Hozumi, T. Akasaka, T. Fukaya; Assessment of mitral regurgitation by biplane transesophageal color Doppler flow mapping; Circulation, 82 (4) (1990), pp. 1121–1126
- [22] M. Zhang, F. Tavora, T. Huebner, J. Heath, A. Burke; Allograft pathology in patients transplanted for idiopathic dilated cardiomyopathy; Am J Surg Pathol, 36 (3) (2012), pp. 389–395
- [23] O. Grebe, K. Magnusson, U. Classen, S. Schlueter, B. Kurtz, E.G. Vester; Right ventricular function differs in idiopathic dilated versus ischemic cardiomyopathy; J Cardiovasc Magn Reson, 12 (Suppl. 1) (2010), p. M5
- [24] C. Stefanadis, J. Dernellis, P. Toutouzas; A clinical appraisal of left atrial function; Eur Heart J, 22 (1) (2001), pp. 22–36
- [25] A. Farzaneh-Far, V. Ariyarajah, C. Shenoy, J.-F. Dorval, M. Kaminski, Z. Curillova, et al.; Left Atrial Passive Emptying Function During Dobutamine Stress MR Imaging Is a Predictor of Cardiac Events in Patients With Suspected Myocardial Ischemia; J Am Coll Cardiol Img, 4 (4) (2011), pp. 378–388
- [26] Q. Zhang, G.W.-K. Yip, C.-M. Yu; Approaching regional left atrial function by tissue Doppler velocity and strain imaging; Europace, 10 (Suppl. 3) (2008), pp. 62–69
- [27] G. Valocik, P. Mitro, L. Druzbacka, I. Valocikova; Left atrial volume as a predictor of heart function; Bratisl Lek Listy, 110 (3) (2009), pp. 146–151
- [28] T. Ito, M. Suwa, Y. Otake, A. Moriguchi, Y. Hirota, K. Kawamura; Left ventricular Doppler filling pattern in dilated cardiomyopathy: relation to hemodynamics and left atrial function; J Am Soc Echocardiogr, 10 (5) (1997), pp. 518–525
- [29] S.G. Teo, H. Yang, P. Chai, T.C. Yeo; Impact of left ventricular diastolic dysfunction on left atrial volume and function: a volumetric analysis; Eur J Echocardiogr, 11 (1) (2010), pp. 38–43
- [30] A. Prioli, P. Marino, L. Lanzoni, P. Zardini; Increasing degrees of left ventricular filling impairment modulate left atrial function in humans; Am J Cardiol, 82 (6) (1998), pp. 756–761
- [31] L. Pratali, P. Otasevic, F. Rigo, S. Gherardi, A. Neskovic, E. Picano; The additive prognostic value of restrictive pattern and dipyridamole-induced contractile reserve in idiopathic dilated cardiomyopathy; Eur J Heart Fail, 7 (5) (2005), pp. 844–851
- [32] Y. Yong, S.F. Nagueh, S. Shimoni, K. Shan, Z.X. He, M.J. Reardon, et al.; Deceleration Time in Ischemic Cardiomyopathy; Circulation, 103 (9) (2001), pp. 1232–1237
- [33] F. Triposkiadis, K. Tentolouris, A. Androulakis, A. Trikas, K. Toutouzas, M. Kyriakidis, et al.; Left atrial mechanical function in the healthy elderly: New insights from a combined assessment of changes in atrial volume and transmitral flow velocity; J Am Soc Echocardiogr, 8 (6) (1995), pp. 801–809
- [34] J.D. Pearlman, M.O. Triulzi, M.E. King, V.M. Abascal, J. Newell, A.E. Weyman; Left atrial dimensions in growth and development: normal limits for two-dimensional echocardiography; J Am Coll Cardiol, 16 (5) (1990), pp. 1168–1174
- [35] K.T. Spencer, V. Mor-Avi, J. Gorcsan, A.N. DeMaria, T.R. Kimball, M.J. Monaghan, et al.; Effects of aging on left atrial reservoir, conduit, and booster pump function: a multi-institution acoustic quantification study; Heart, 85 (3) (2001), pp. 272–277
- [36] R.M. Lang, M. Bierig, R.B. Deverux, F.A. Flachskampf, E. Foster, P.A. Pellikka, et al.; Recommendations for chamber quantification: a report from the American Society of Echocardiographys Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology; J Am Soc Echocardiogr, 18 (12) (2005), pp. 1440–1463
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?