Abstract
The increasing incidence of Parkinson’s disease (PD) represents a significant public health challenge, highlighting the urgent need for more accessible and innovative treatments. Rocoto pepper (Capsicum pubescens), a South American chili pepper, has a unique composition of capsaicin, dihydrocapsaicin, antioxidants, and vitamins A and C. While previous studies have investigated the neuroprotective properties of several pepper species, the effects of Rocoto pepper on PD remain largely unexplored. The present study examines the effects of Rocoto pepper extract at varying concentrations on Parkinson’s disease using Caenorhabditis elegans (C. elegans) as a model organism. C. elegans were exposed to four different concentrations of Rocoto extract (0%, 4%, 8% and 16%) applied onto the surface of Nematode Growth Medium (NGM) and fed E. coli OP50. Over the course of one week, behavioral assays were conducted to monitor locomotion and touch sensitivity. By exploring an overlooked yet culturally rich crop, this study calls attention to a broader and more inclusive approach in the search for sustainable medical solutions. Future research would test the individual compounds found in the pepper to determine which component has the greatest neuroprotective effects and the mechanisms it underwent in dopaminergic pathways. Expanding the range of tested concentrations, increasing the sample size, and replicating trials would strengthen the reliability of the findings.
Introduction
Parkinson’s Disease
Parkinson’s disease (PD) is a progressive neurodegenerative disorder, which is characterized by the loss of dopaminergic neurons in the substantia nigra, leading to motor symptoms such as bradykinesia, rigidity, resting tremor, and postural instability (Poewe et al., 2017). In addition, PD has been affecting over 10 million people worldwide, and it also involves significant non-motor symptoms including cognitive decline, disturbances when sleeping, and autonomic dysfunction, which reduces the quality of life and complicates the treatments (Kalia & Lang, 2015). The lack of dopamine leads to symptoms like slow movement, muscular rigidity, involuntary shaking, and posture problems (Chaudhuri et al., 2006). There is still no well-established treatment that could slow the disease progression, but significant progress was made in developing new therapies to treat PD conditions (Poewe & Mahlknecht, 2009). Despite current pharmacological interventions, there is still no cure, and PD has been progressing over time (Olanow et al., 2009).
There are several factors that lead to the increase of the likelihood of the development of this PD. Although most cases of PD are known to be sporadic, genetic predisposition plays a role in some instances. Mutations in specific genes, such as PINK1, and PRKN, have been linked to familial forms of Parkinson’s disease. These genetic mutations can affect protein functions and lead to neural degeneration (Klein & Westenberger, 2012). Exposure to certain environmental toxins has also been associated with the increased risk of PD. Pesticides, herbicides and industrial chemicals have been implicated in the development of the disease. Rural living and well-water consumptions have furthermore been suggested as potential risk factors due to higher exposure to agricultural chemicals (Tanner et al., 2011). Parkinson's disease is very well known to have the most significant effects on people over the age of 60, and the likelihood of PD developing comes with the increase in age. This is thought to be due to cumulative effects of aging on neuronal health and resilience (Reeve et al., 2014). In many cases a factor to PD has also been discovered to be gender since it has been seen throughout studies that men are more likely to develop PD than women. This can be due to the hormonal differences, genetic factors and lifestyle variations can contribute to this disparity (Wooten et al., 2024).
Rocoto Pepper F
Rocoto peppers, scientifically known as Peruvian Capsicum Pubescens, are a unique and ancient variety of chili pepper native to the Andean region in South America, more specifically found in Peru and Bolivia. Historical evidence suggests that rocoto peppers have been cultivated for thousands of years, with their use being tracked back to pre-inca civilization (National Research Council, 1989). Rocoto peppers are a very big part of everyday diets in the Peruvian and Bolivian areas, being used as spices and as well as in many traditional dishes (Poewe & Mahlknecht, 2009). Rocoto peppers are characterized by their flower, purple and black seeds which are morphologically medium-sized apple shaped and bright red and for their tolerance to lower temperature environments (Yamamoto et al., 2013). One of the most important compounds is capsaicin, which gives the pepper its heat and may also help protect the body from disease. Scientists have found that capsaicin has anti-inflammatory and antioxidant properties, meaning it can help lower swelling in the body and protect cells from damage (Arslan et al., 2022)
Nutritionally, rocoto peppers are rich in vitamins A and C, as well as antioxidants and capsaicin, this compound has been extensively studied for its potential health benefits, including pain relief and metabolic enhancements (Materska & Perucka, 2005). Its pungency level ranges from 100,000 to 210,000 Scoville Hot Units (SHU), making them significantly hotter than jalapenos (López, 2003). Studies have shown that capsaicin may have antioxidant and anti-inflammatory properties, as well as possible benefits for metabolism and pain relief. In traditional medicine, rocoto chiles are sometimes used topically in moderation as a natural remedy for conditions such as arthritis or muscle pain (Pizzeria & Gastropub, 2025). Because of its strong bioactive properties, Capsicum pubescens is becoming a topic of interest in modern scientific research. Studies have shown that compounds in capsaicin may help protect cells from damage caused by oxidative stress and may even support brain health, making rocoto a promising candidate for studying effects on aging and neurodegenerative diseases in model organisms such as C. elegans (Reyes-Escogido et al., 2011).
C. elegans
C.elegans is an organism that can be changed to model many different human-like diseases, making studying and experimenting on these diseases much easier. Caenorhabditis elegans, C. elegans for short, is a useful model organism in scientific research, especially to study aging, the nervous system, and diseases like Parkinson’s Disease. Even for its small size, it has a lot in common with humans at the genetic and cellular level. C. elegans is popular because it has a simple body made up of exactly 959 cells, including 302 nerve cells, which makes it much easier to study than complex animals like mice. Its entire body is see-through, so researchers can actually watch what’s happening inside the worm under a microscope in real time (Bargmann, 1998).
C. elegans grow fast, living only approximately 2 to 3 weeks, helping to see the results fast and run many experiments in a short period of time (Artal-Sanz et al., 2006). One important reason for the popularity of C.elegans is that they can be genetically modified to carry human genes, including those involved in diseases like Parkinson’s disease. For instance, some worms are made to produce human alpha-synuclein, which is the protein that builds up in the brain of people diagnosed with Parkinson’s disease. These worms start to show problems similar to what happens in human patients, like damage to dopamine-producing neurons and trouble moving (Lakso et al., 2003).
In the wild, C. elegans consumes a variety of soil bacteria. In laboratory conditions, they are typically fed Escherichia coli (E. coli), particularly the OP50 strain. This strain is used because it forms a thin lawn on agar plates, making it easy for the worms to consume and for researchers to observe them (Brenner, 1974).The bacterial diet provides essential nutrients required for the growth and reproduction of C. elegans. Studies have shown that different bacterial strains can affect the worm’s physiology and lifespan, highlighting the importance of diet in experimental outcomes (MacNeil et al., 2013).The simplicity of feeding C. elegans with bacteria like E. coli makes it an ideal model organism for genetic and developmental studies. Researchers can easily manipulate the bacterial diet to study various biological processes, including metabolism and aging (Cabreiro et al., 2013).
Previous Studies
Preceding research has highlighted the therapeutic potential of bioactive compounds found in various pepper species in combating neurodegenerative diseases such as Parkinson’s disease (PD) and Alzheimer’s disease. In 2014, a study investigated the inhibitory effects of piperine, the active compound in Piper nigrum (black pepper), on the enzyme monoamine oxidase B (MAO-B), which is implicated in PD progression. The study found that piperine possessed significant neuroprotective and antidepressant properties. Its ability to inhibit MAO-B, an enzyme responsible for degrading dopamine, suggests a protective mechanism by which it can help preserve dopaminergic signaling. Since dopamine is a key neurotransmitter for motor control, preserving its levels is critical in managing PD symptoms (Al-Baghdadi, 2014).
Another relevant study explored the effects of Capsicum annuum extract on neuronal cholinergic and purinergic enzyme activities in rotenone-intoxicated rat models of Parkinson’s disease. Rotenone is a neurotoxin that mimics Parkinsonian neurodegeneration. Treatment with ethyl acetate fractions of C. annuum significantly improved behavioral symptoms, such as memory decline, and demonstrated neuroprotective effects. The findings support the potential use of capsaicinoid-rich pepper extracts as part of a multifaceted therapeutic strategy in the management of Parkinson’s disease (Ogunruku + Ogunyemi, 2019).
While Rocoto pepper (Capsicum pubescens) has not yet been directly studied in relation to neurodegenerative diseases, a comprehensive 2015 phytochemical analysis of 32 C. pubescens varieties revealed notable levels of capsaicinoids, polyphenols, antioxidant capacity, and tocopherols. Although it displayed less phytochemical diversity compared to other domesticated pepper species, Rocoto still showed nutritionally robust properties, suggesting its potential value in health-related research. These findings support the hypothesis that Rocoto pepper could exhibit neuroprotective effects similar to those observed in other pepper species, particularly due to its rich content of compounds known to modulate oxidative stress and inflammation (Meckelmann et al., 2015).
Importance of Research
Parkinson’s disease is a growing global health concern, affecting over 10 million people around the globe. This number is projected to significantly increase due to the growing aging population; estimates suggest that the global prevalence of Parkinson’s disease would reach 25.2 million by 2050. Current treatments for PD, such as dopamine replacement therapies, offer temporary alternatives but do not slow down the disease progression or repair damaged neurons. This emphasizes the urge for identifying neuroprotective agents that can prevent or decelerate the neuronal degeneration (Luo et al., 2025).
Natural compounds from plants have gained increasing attention in recent years for their potential to combat neurodegenerative diseases. Peppers, in particular, have been studied for their high content of capsaicinoids and antioxidants, which may reduce oxidative stress, a key contributor to dopaminergic neuron loss in Parkinson’s disease (Blesa et al., 2015). However, while many common pepper species have been evaluated for those properties, Rocoto pepper remains mainly unexplored, despite its rich phytochemical composition and cultural significance in South America.
The investigation of the effects of Rocoto pepper extract on a C. elegans Parkinson’s disease model contributes to an underrepresented area of research, providing valuable insights into the neuroprotective potential of an overlooked food crop and evaluating whether its bioactive compounds can influence neuronal survival and behavior. If Rocoto demonstrates neuroprotective effects, it could open the door for future studies in more complex systems and support the development of natural-compound based therapies for Parkinson’s disease.
Furthermore, this research encourages a more sustainable and culturally inclusive approach to biomedical discovery. The exploration of native crops like Rocoto could promote biodiversity, regional agricultural development, and equitable innovation in global health.
Hypothesis
The aim of this study is to evaluate the effects of rocoto pepper at varying concentrations on Parkinson’s disease in C. elegans models. There is a lack of research on rocoto peppers and their potential effects on Parkinson's disease, but overall the pepper is very well known for its rich flavor and especially for its many nutritional benefits. This pepper has been used for centuries in South American countries for medicinal purposes as well as in many famous cuisines. Many studies show that people in South America, especially in places like Peru and Bolivia, have lower cases of PD compared to the US, and there could be a correlation between PD and the rocoto peppers due to this pepper being one of the native fruits in both Bolivia and Peru. Since rocoto peppers are widely used and considered important in parts of South America, there may be a correlation between eating them and reduced symptoms of PD. Furthermore it is anticipated that the higher concentrations of rocoto pepper extracts will show the greatest impact on the reduction of PD. The control group (C) will be used to assess the effects of the treated groups with rocoto pepper extract by comparing the different behaviors of Parkinson's disease C.elegans model.
Materials and Methods
Pre-Experiment
All material and equipment were sterilized before the experiment by autoclaving them using the scissors icon. Ethanol (#974454A), 5 NGM-135mL (#172520), and nutrient agar powder- 100 g (#842130) were ordered from Carolina biological beforehand. Gloves, goggles, masks, and aprons were worn at all times for safety precautions. The tables and tools were sterilized with 10% bleach before any actions were taken with the experiment. Glass pipettes, microcentrifuge tubes, and micropipettes were autoclaved to prevent contamination during the experiment. 50 mL of distilled water was measured and poured in a flask, which then was covered loosely using aluminum foil to prevent further contamination. The substance was autoclaved using the liquids option and was stored in the same bottle in which it was autoclaved, the bottle was labeled “Sterile water” and the date, sealed and stored in a clean area. The organisms were observed under a compound microscope which was provided by the lab.
Table 1. Experimental Setup
| Group | C. elegans strain | Concentration of the Rocoto Pepper Extract Treatment dissolved in ethanol (%) | Sample size | 100% Ethanol | Volume of
4, 8, 16% Rocoto Pepper Extract in 4% Ethanol (mL) |
| A | DDP1 | 0 | 2 | 0 | 0 |
| B | Wild type | 0 | 2 | 2.7 | 0 |
| C | Wild type | 0 | 2 | 0 | 0 |
| D | DDP1 | 0.5 | 2 | 0 | 1.25 |
| E | DDP1 | 1 | 2 | 0 | 1.25 |
| F | DDP1 | 2 | 2 | 0 | 1.25 |
Preparation of the Rocoto Pepper Extract Solutions
Two fresh Rocoto peppers were obtained. They were first washed, and the seeds and stems were carefully removed. The peppers were then dried using a paper towel and cut into slices with a clean, sterile scalpel. The pepper slices were placed on a tray and put into a dehydrator at 115. The dehydrator was left on overnight, and the peppers were checked every morning until they appeared dehydrated. The following step was done inside a fume hood while wearing a mask, goggles, and gloves to avoid inhaling the pepper powder. Once the peppers were fully dried, a mortar and pestle were used to grind them into a fine powder. If available, a clean blender or spice grinder was used instead. To prepare the extracts, three clean 50mL volumetric flasks and one 10mL graduated cylinder were ready for use. To make stock solution D (4% pepper, 4% ethanol), 2g of rocoto pepper were weighed using a scale. The pepper was transferred into a 50mL volumetric flask. 2mL of ethanol was measured using the graduated cylinder and added to the flask. The solution was brought to the 50mL mark using sterile water. The flask was inverted 25 times to ensure the solution was evenly integrated. For stock solution E (8% pepper, 4% ethanol), 4g of rocoto pepper were weighed out and transferred into a 50mL volumetric flask. Then 2mL of ethanol was measured out and added into the flask, which was then filled to the 50mL mark using sterile water. The solution was then mixed thoroughly. For stock solution F (16% pepper, 4% ethanol), 8g of rocoto pepper were weighed out, and 2mL of ethanol were added into the volumetric flask. The flask was then filled up to the 50mL mark with sterile water and the solution was then mixed thoroughly. These mixtures were then filtered by using the vacuum filtration method. The filtered liquids, which were the 4% extracts, were collected and stored in bottles. The bottles were covered with aluminum foil, since light degrades capsaicin, and kept in the refrigerator and labeled based on their concentrations respectively.
Preparation of 4% Ethanol Solution
To prepare stock solution B (0% pepper, 4% ethanol), 2mL of ethanol was measured using a graduated cylinder and was then poured into the volumetric flask. Sterile water was then poured into the flask until the 50mL mark was reached. This solution was then poured into a sterile bottle and kept in the refrigerator and labeled “B” respectively.
Preparing Nematode Growth Medium
To begin the preparation of the growth medium, 12 Petri dishes with a diameter of 60 mm were obtained. 6 nematode growth agar, were ordered from Carolina Biological Supply, and prepared in bottles of 135 mL (#173520). With the liquid option on the autoclave, the bottles were placed in the autoclave and made into liquid to avoid any contamination that might occur. At first of the 12 petri dishes 2 were labeled with “A” on the bottom, this procedure was repeated with the groups “B”, “C”, “D”, “E” & “F”, followed by the respective dates for all plates making sure not to label the lids. In the Petri dish labeled “A” & “C”, there was no treatment since they were the control groups. Then, four 10mL beakers were obtained. Stock Solution D with the 4% pepper and 4% ethanol was obtained. With a micropipette, 1.25 mL (1250 μL) of the stock solution was pipetted, complete with 8.75 mL (8750 μL) of water. The mixture was pipetted up and down to incorporate and prepare the solution. Two plates previously labeled D were obtained, and 100 μL of the solution was then pipetted into the dishes. Using the Bunsen burner a cell spreader was flamed, allowed to cool down completely, and then used to spread the solution evenly. The same procedures were then repeated for stock solutions B, E, and F: 1.25 mL (1250 μL) of each stock solution was pipetted and completed with 8.75 mL (8750 μL) of water. The solution was mixed thoroughly and pipetted into the two labeled plates E and F respectively. For each set of plates pere coated with 100 μL of the prepared solutions, a spreader was flamed and allowed to cool down, and the solution was then spread evenly across the agar. After this process the plates were not flipped over.
Cultivation of E. coli OP50
Nutrient agar powder (#842130) was first obtained, using a scale, about 4.03 g of the powder was measured by scoping it using a scoopula on a weighing boat. The powder was transferred to a 300 mL Erlenmeyer flask. Using a graduated cylinder, 130 mL of deionized water was measured and poured in the same Erlenmeyer flask as the powder. The flask had to be gently swirled, however; for it to become a homogeneous solution it had to be autoclaved using the liquid option on the autoclave. 3 Petri dishes were obtained with a 100 mm diameter and was labeled “OP50” and the respective date on the bottom of the dish. Once the agar was autoclaved about 25 mL was carefully poured into each Petri dish working quickly to prevent any contamination. Afterwards, nutrient broth powder was obtained to cultivate E. coli OP50, the main source of nourishment for the nematodes. Using a scoopula about 2 grams of the powder was weighed on a weighing boat on a gram scale. Then the powder was added into a 300 mL erlenmeyer flask. A graduated cylinder was used to measure out 100 mL of deionized water then poured into the erlenmeyer flask. A piece of aluminum foil was loosely taped to the stem of the flask, and the solution was then autoclaved using the liquids option. Once it had been autoclaved and formed a homogeneous mixture, the tube in which the E. coli that was provided by the lab was retrieved. A bunsen burner was lit, and an inoculation loop was sterilized by flaming it. The loop was allowed to cool for a few seconds by touching the glass. The loop was then inserted into the tube to collect a small sample of E. coli, which was submerged into the broth solution and stirred. All of this was done near the flame for sterility. After the bacteria had been cultivated in the broth, the forceps were sterilized again by flaming. The aluminum foil was taped back over the flask to avoid contamination. The bacterial broth was incubated at 37 °C for one day to allow the bacteria to reproduce. Using a previously autoclaved micropipette with sterile tips, 100 μL of the prepared broth was pipetted. A cell spreader was flamed to disinfect it, then cooled by touching the border of the agar plate. The broth was then spread evenly over the agar surface to form a full bacterial lawn. The plates were incubated at room temperature. Backup plates were sealed with parafilm and stored at 4 °C.
Addition of the C. elegans into the plates
Before beginning the procedures, all materials and surfaces were sterilized and cleaned. The strain used in this experiment (DDP1) was distributed as a colony. A scalpel was carefully flamed and allowed to cool down for 10 seconds to ensure sterilization. The scalpel was used to cut a total of 12 agar chunks, which were then carefully picked up and placed onto the NGM plates labeled “B,” “D,” “E,” and “F.” This procedure was repeated for all 12 plates. The plates were incubated at 20 °C. For the plates labeled “A” and “C,” a chunk of worms from the “wild type” strain were added using the same method described above.
Transfer of E. coli OP50
The Bunsen burner was lit. A scalpel was carefully flamed until the blade glowed red-hot to ensure sterilization. It was then allowed to cool for 10 seconds. The previously prepared agar plate cultured with E. coli OP50 was taken, and the scalpel was used to cut an agar chunk as quickly as possible to prevent contamination. A total of 12 pieces of E. coli-containing agar were cut. The forceps were flamed and allowed to cool for a few seconds. They were then used to lift each agar chunk and transfer it onto an NGM plate. All plates were checked to ensure that each contained one piece of agar.
Gentle Touch Assessment
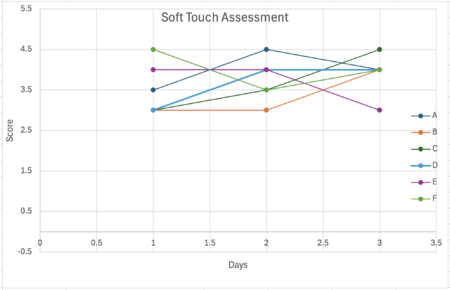
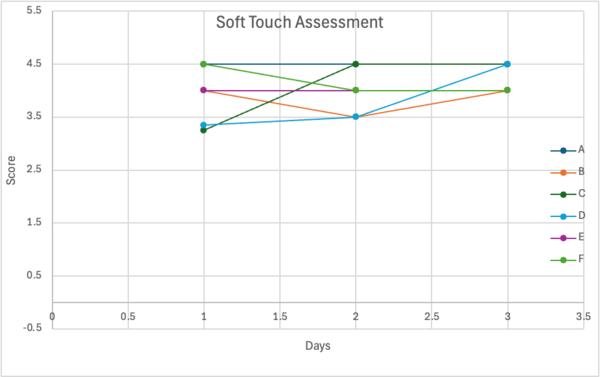
To begin the gentle touch assessment, an eyelash (about one centimeter long) was obtained and sanitized using 70 % isopropyl alcohol. Using a small piece of tape, the base of the eyelash was secured to the tip of a toothpick so it extended slightly beyond the tip. Before use the eyelash had to be sanitized once more to prevent contamination then the C. elegans was gently stroked about five times. When done with the strokes, the sit toothpick was then sanitized using sanitizing wipes and alcohol. In the morphological assay the data observed and recorded was how fast the C. elegans reacted in response to the gentle stimuli, this procedure was repeated every other day. Using a scale of 1-5 how fast the worms responded was assessed. Line graphs display the scale number that the C. elegans responded to over the span of 3 days.
Harsh Touch Assessment
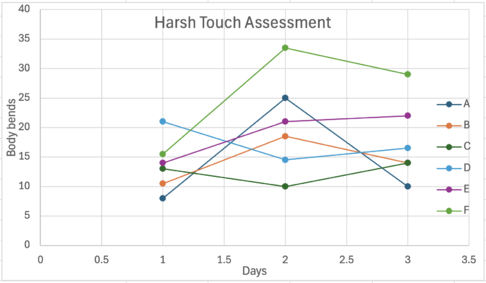
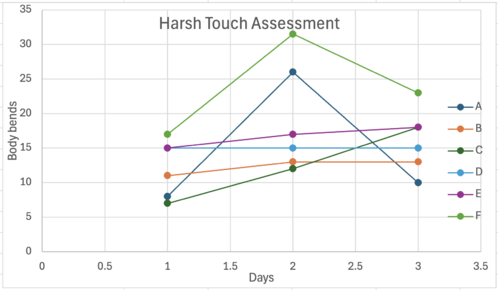
Before taking any action a metal worm probe was sterilized by using a bunsen burner, it was allowed to cool before usage. The metal worm probe was used to carefully touch the C. elegans. Once the C.elegans was in contact with the metal probe, they were observed on their response to the harsh touch assessment and the number of times the worm's thrashed was recorded. After the usage of the metal probe alcohol wipes were used to sanitize and to prevent any contamination after contact with the C.elegans. This procedure was repeated every day. For harsh touch response assays, the number of movements of the worms over a period of 20 seconds was recorded and shown by using a line graph.
Data Analysis
For harsh and gentle touch response assays, the different reactions were recorded and shown using line graphs.
In the morphological bending assay, worms were assessed for their ability to bend in response to a gentle and harsh stimulus. Using a scale from 1-5, the worms were assessed on the speed at which they exhibited a bending response. Line graphs were used to display the effects of the gentle stimuli on the reaction times of the worms across groups. The amount of time the worms thrashed to the harsh touch was recorded and assessed. Line graphs were used to display the number of movements exhibited by the C. elegans as a response to this harsh stimuli.
The significance of the harsh touch assessment was evaluated using a Negative Binomial Regression model. This statistical method allowed for the comparison of treatment groups while accounting for variability in the number of responses exhibited by C. elegans. This method was used since C. elegans are count-based and often overdisperse, making this model the most appropriate for analyzing differences between groups accurately.
The significance of the soft touch assessment was analyzed using the Kruskal-Wallis test (a non-parametric alternative to one-way ANOVA). The Kruskal-Wallis test allowed for the determination of whether differences in response scores across treatment groups were statistically meaningful. This test was used since the data were not normally distributed, and this non-parametric test allows for comparing multiple treatment groups without needing normality.
P-value was used to determine whether there were significant differences among treatment groups compared to the control. The p-value ≤ 0.05 was considered statistically significant. The p-value reflects the probability that the observed differences could have occurred by chance if there were no real effect; a low p-value suggests that the treatment had a meaningful impact on behavior or survival.
To examine the changes and significance of the different tests and treatment groups, the z-score was used to show how far each data point was from the mean in units of standard deviation values. A z-value of 1 would indicate that the score is one standard deviation above the mean, while a z-value of -2 would indicate the score is two standard deviations below the mean. Using z-values allows for the standardization of data and comparing variations across treatments for both harsh touch and soft touch assessments.
Results
Figure 1. Trial 1 Soft Touch Assessment
Figure 2. Trial 1 Harsh Touch Assessment
Table 1. Statistical analysis of the Harsh Touch Assessment: Trial 1
| Group | Estimate | Std. Error | z-value | p-value | Significance |
| A | 2.419 | 0.183 | 13.196 | <0.001 | *** |
| B | 0.020 | 0.222 | 0.088 | 0.930 | |
| C | -0.106 | 0.226 | -0.470 | 0.638 | |
| D | 0.244 | 0.217 | 1.125 | 0.261 | |
| E | 0.307 | 0.215 | 1.423 | 0.155 | |
| F | 0.606 | 0.210 | 2.888 | 0.004 | ** |
| Day 2 | 0.388 | 0.152 | 2.550 | 0.011 | * |
| Day 3 | 0.229 | 0.154 | 1.485 | 0.138 |
Figure 3. Trial 2 Soft Touch Assessment
Figure 4. Trial 2 Harsh Touch Assessment
Table 2. Statistical analysis of the Harsh Touch Assessment: Trial 2
| Group | Estimate | Std. Error | z-value | p-value | Significance |
| A | 2.412 | 0.146 | 16.511 | <0.001 | *** |
| B | -0.157 | 0.179 | -0.878 | 0.380 | |
| C | -0.162 | 0.179 | -0.905 | 0.365 | |
| D | 0.042 | 0.172 | 0.245 | 0.806 | |
| E | 0.146 | 0.169 | 0.862 | 0.389 | |
| F | 0.494 | 0.160 | 3.087 | 0.002 | ** |
| Day 2 | 0.446 | 0.122 | 3.661 | 0.000 | *** |
| Day 3 | 0.288 | 0.125 | 2.304 | 0.021 | * |
Table 3. Statistical analysis of the Soft Touch Assessment
| Trial | Analysis | Test | χ² | df | p-value | Significance |
| 1 | Soft touch | Kruskal–Wallis | 3.1354 | 5 | 0.6791 | NS |
| 2 | Soft touch | Kruskal–Wallis | 4.3131 | 5 | 0.5053 | NS |
Discussion and Conclusion
Overall, the findings from this experiment suggest that Rocoto pepper extract may exhibit neuroprotective properties against Parkinson’s Disease in C. elegans. Across multiple trials, the increasing concentrations of the extract were associated with higher body-bend responses in the harsh touch assessment. This trend indicates improved motor function. The negative binomial regression further supported this pattern: Group F, which received the highest concentration of Rocoto extract, performed significantly better than Group A (the untreated DDP1 strain), where Parkinsonian symptoms were allowed to progress without intervention. These results align with previous research suggesting that plant-derived compounds, especially those with capsaicinoid-like activity, may mitigate neurodegeneration.
Despite these promising findings, several limitations must be acknowledged. Although the preparation of the extracts was conducted carefully, full sterilization could not be ensured because autoclaving would degrade important chemical components. Additionally, due to time constraints, the extract could not be incorporated directly into the NGM. Instead, it was applied onto the surface and allowed to absorb. This method may have caused the concentration to gradually decrease over time as it diffused into the agar. To reduce this effect, the trial period was kept relatively short (two weeks). Another limitation was that behavioral assays were conducted every other day rather than daily, reducing the granularity of the data and potentially obscuring short-term changes in motor function.
Future research should include a larger number of trials and test individual chemical components of Rocoto pepper to determine which compounds provide neuroprotection. The experimental period should also be extended to seven consecutive days with daily assays, allowing for clearer observation of behavioral trends over time. Furthermore, incorporating the extract directly into the NGM would help maintain consistent concentrations throughout the experiment. Finally, evaluating Rocoto pepper extract alongside standard Parkinson’s treatment, such as levodopa, would allow for direct comparison of efficacy. These improvements would strengthen the validity of the findings and help determine whether Rocoto pepper extract could serve as a potential supplementary therapy for neurodegenerative conditions.
Importantly, Rocoto pepper has a unique composition of capsaicin, dihydrocapsaicin, antioxidants, and vitamins A and C. While previous studies have examined the neuroprotective properties of other pepper species, the effects of Rocoto pepper on Parkinson’s Disease remain largely unexplored. This novelty emphasizes the significance of the current findings.
Acknowledgements
We thank: Dr. D. Marmor, Dr. L. Wang, Ms. Babbar, Dr. B.A.B. Blackwell, Dr. J. Cohen, Dr. S. Lin, Mr. Z. Liang, Mrs. C. Rastetter, Ms. DePietro, and Ms. Macleod.
Funding: FLHS Science Research Program
References
Al-Baghdadi, O. B. (2014). Inhibition of Monoamine Oxidase by Derivatives of Piperine, an Alkaloid from the Pepper Plant Piper Nigrum, for Possible Use in Parkinson’s Disease [Master of Science, Kent State University].
Artal-Sanz, M., de Jong, L., & Tavernarakis, N. (2006). Caenorhabditis elegans: a versatile platform for drug discovery. Biotechnology Journal, 1(12), 1405–1418.
https://pubmed.ncbi.nlm.nih.gov/17109493/
Bargmann, C. I. (1998). Neurobiology of the Caenorhabditis elegans genome. Science, 282(5396), 2028–2033.
https://pubmed.ncbi.nlm.nih.gov/9851919/
Blesa, J., Trigo-Damas, I., Quiroga-Varela, A., & Jackson-Lewis, V. R. (2015). Oxidative stress and Parkinson’s disease. Frontiers in Neuroanatomy, 9, https://doi.org/10.3389/fnana.2015.00091
Brenner, S. (1974). The genetics of Caenorhabditis elegans. Genetics, 77(1), 71-94 .https://pubmed.ncbi.nlm.nih.gov/4366476/
Cabreiro, F., Au, C., Leung, K. Y., Vergara-Irigaray, N., Cochemé, H. M., Noori, T., … & Gems, D. (2013). Metformin retards aging in C. elegans by altering microbial folate and methionine metabolism. Cell, 153(1), 228-239 .https://pubmed.ncbi.nlm.nih.gov/23540700/
Chaudhuri, K. R., Healy, D. G., & Schapira, A. H. (2006). Non-motor symptoms of Parkinson's disease: diagnosis and management. The Lancet Neurology, 5(3), 235–245.
https://pubmed.ncbi.nlm.nih.gov/16488379/
Chege, P. M., & McColl, G. (2014). Caenorhabditis elegans: a model to investigate oxidative stress and metal dyshomeostasis in Parkinson’s disease. Frontiers in Aging Neuroscience, 6. https://doi.org/10.3389/fnagi.2014.00089
Chen, H., Zhang, S. M., Schwarzschild, M. A., Hernán, M. A., & Ascherio, A. (2005). Physical activity and the risk of Parkinson disease. Neurology, 64(4), 664–669. http://dx.doi.org/10.1212/01.WNL.0000151960.28687.93
Gardner, R. C., Yaffe, K., & Byers, A. L. (2014). Traumatic brain injury in later life increases risk for Parkinson disease. Annals of Neurology, 83(3), 455–461. https://doi.org/10.1002/ana.24396
Harrington, A. J., Hamamichi, S., Caldwell, G. A., & Caldwell, K. A. (2010). C. elegans as a model organism to investigate molecular pathways involved with Parkinson’s disease. Developmental Dynamics, NA-NA. https://doi.org/10.1002/dvdy.22231
Jadiya, P., Khan, A., Sammi, S. R., Kaur, S., Mir, S. S., & Nazir, A. (2011). Anti-Parkinsonian effects of Bacopa monnieri: Insights from transgenic and pharmacological Caenorhabditis elegans models of Parkinson’s disease. Biochemical and Biophysical Research Communications, 413(4), 605–610. https://doi.org/10.1016/j.bbrc.2011.09.010
Kaletta, T., & Hengartner, M. O. (2006). Finding function in novel targets: C. elegans as a model organism. Nature Reviews Drug Discovery, 5(5), 387–398.
https://pubmed.ncbi.nlm.nih.gov/16672925/
Klein, C., & Westenberger, A. (2012). Genetics of Parkinson’s disease. Cold Spring Harbor Perspectives in Medicine, 2(1), a008888. https://doi.org/10.1101/cshperspect.a008888
Lakso, M., Vartiainen, S., Moilanen, A. M., Sirviö, J., Thomas, J. H., Nass, R., ... & Wong, G. (2003). Dopaminergic neuronal loss and motor deficits in Caenorhabditis elegans overexpressing human α-synuclein. Journal of Neurochemistry, 86(1), 165–172.
https://pubmed.ncbi.nlm.nih.gov/12807436/
Luo, Y., Qiao, L., Li, M., Wen, X., Zhang, W., & Li, X. (2025). Global, regional, national epidemiology and trends of Parkinson's disease from 1990 to 2021: findings from the Global Burden of Disease Study 2021. Frontiers in aging neuroscience, 16, 1498756. https://doi.org/10.3389/fnagi.2024.1498756
MacNeil, L. T., Watson, E., Arda, H. E., Zhu, L. J., & Walhout, A. J. M. (2013). Diet-induced developmental acceleration independent of TOR and insulin in C. elegans. Cell, 153(1), 240-252. https://pubmed.ncbi.nlm.nih.gov/23540701/
Materska, M., & Perucka, I. (2005). Antioxidant activity of the main phenolic compound isolated from hot pepper fruit (Capsicum annuum L.). Journal of Agricultural and Food Chemistry, 53(3), 1750-1756.
https://pubmed.ncbi.nlm.nih.gov/15740069/
Meckelmann, S.W., Jansen, C. Riegel D.W., Van Zonneveld M., Ríos, L., Peña, K.,
Mueller-Seitz, E. & Petz, M. (2015). Phytochemicals in Native Peruvian Capsicum pubescens (Rocoto). European Foods and Technology, 1-25. 10.1007/s00217-015-2506-y
Mendes, N.S., Coimbra, P.P.S., Santos, M.C.B., Cameron, L.C., Ferreira, M.S.L., Buera, M.P. & Goncalves, E.C.B.A. (2020). Capsicum pubescens as a functional ingredient: Microencapsulation and phenolic profilling by UPLC-MSE. Food Research International, 135. https://doi.org/10.1016/j.foodres.2020.109292
National Institute of Neurological Disorders and Stroke. (2023). Parkinson’s Disease: Challenges, Progress, and Promise | National Institute of Neurological Disorders and Stroke. Retrieved from www.ninds.nih.gov website: https://www.ninds.nih.gov/current-research/focus-disorders/parkinsons-disease-research/parkinsons-disease-challenges-progress-and-promise
National Research Council. (1989). Lost Crops of the Incas: Little-Known Plants of the Andes with Promise for Worldwide Cultivation. National Academy Press. https://nap.nationalacademies.org/catalog/1398/lost-crops-of-the-incas-little-known-plants-of-the
Ogunruku, O. O., & Ogunyemi, B. O. (2019). Pepper (C. annuum Linn) Extract Ameliorates Alterations to Neuronal Cholinergic and Purinergic Enzyme Activities in the Rotenone-intoxicated Rat Model of Parkinson’s Disease. Ife Journal of Science, 21(1), 249. https://doi.org/10.4314/ijs.v21i1.23
Olanow, C. W., Stern, M. B., & Sethi, K. (2009). The scientific and clinical basis for the treatment of Parkinson disease (2009). Neurology, 72(21 Suppl 4), S1–S136.
https://pubmed.ncbi.nlm.nih.gov/19470958/
Poewe, W., & Mahlknecht, P. (2009). The clinical progression of Parkinson’s disease. Parkinsonism & Related Disorders, 15, S28–S32. https://doi.org/10.1016/s1353-8020(09)70831-4
Poewe, W., Seppi, K., Tanner, C. M., Halliday, G. M., Brundin, P., Volkmann, J., ... & Lang, A. E. (2017). Parkinson disease. Nature Reviews Disease Primers, 3, 17013.
https://pubmed.ncbi.nlm.nih.gov/28332488/
Reeve, A., Simcox, E., & Turnbull, D. (2014). Ageing and Parkinson’s disease: why is advancing age the biggest risk factor? Ageing Research Reviews, 14, 19–30. https://doi.org/10.1016/j.arr.2014.01.004
Signorile, A., Ferretta, A., Pacelli, C., Capitanio, N., Tanzarella, P., Matrella, M. L., … Cocco, T. (2021). Resveratrol Treatment in Human Parkin-Mutant Fibroblasts Modulates cAMP and Calcium Homeostasis Regulating the Expression of Mitochondria-Associated Membranes Resident Proteins. Biomolecules, 11(10), 1511–1511. https://doi.org/10.3390/biom11101511
Tanner, C. M., Ross, G. W., Jewell, S. A., Hauser, R. A., Jankovic, J., Factor, S. A., … & Langston, J. W. (2009).Occupation and risk of parkinsonism: a multicenter case-control study. Archives of Neurology, 66(9), 1106–1113. https://doi.org/10.1001/archneurol.2009.195
Vozdek, R., Pramstaller, P. P., & Hicks, A. A. (2022). Functional Screening of Parkinson’s Disease Susceptibility Genes to Identify Novel Modulators of α-Synuclein Neurotoxicity in Caenorhabditis elegans. Frontiers in Aging Neuroscience, 14. https://doi.org/10.3389/fnagi.2022.806000
Wooten, G. F., Currie, L. J., Bovbjerg, V. E., Lee, J. K., & Patrie, J. (2004). Are men at greater risk for Parkinson’s disease than women? Journal of Neurology, Neurosurgery & Psychiatry, 75(4), 637–639. http://dx.doi.org/10.1136/jnnp.2003.017821
Document information
Published on 21/03/26
Submitted on 13/03/26
Licence: CC BY-NC-SA license