(Created page with " ==Abbreviations== AMT, Aortic mural thrombus;CTPA, Computed Tomography Pulmonary Angiography;DVT, Deep Venous Thrombosis ==Keywords== Aortic thrombus;Arterial thrombosis;...") |
m (Scipediacontent moved page Draft Content 118500705 to Haji et al 2016a) |
(No difference)
| |
Latest revision as of 11:31, 19 May 2017
Abbreviations
AMT, Aortic mural thrombus;CTPA, Computed Tomography Pulmonary Angiography;DVT, Deep Venous Thrombosis
Keywords
Aortic thrombus;Arterial thrombosis;Thromboembolism
1. Case report
In October 2015, a 45-year-old woman presented to our hospital by ambulance with a 10 day history of progressively increasing. Her past history was of Type-2 Diabetes Mellitus with known microvascular complications, hypertension and recent menorrhagia complicated by iron deficiency anaemia for which she had been commenced on norethisterone three weeks prior. Notably, she had a child delivered by caesarean section, had no previous history of venous thromboembolism and was an ex-smoker having quit 7 years prior.
At the time of ambulance arrival she was in severe respiratory distress with a respiratory rate of 44 and oxygen saturations of 85% on room air, hypertensive (210/110) and tachycardic (104 bpm). Her oxygen saturations improved with 10 L supplemental oxygen via mask but she remained tachycardic and hypertensive. Her chest was clear on auscultation and while she did have some leg pain there was no clinical suspicion of a Deep Venous Thrombosis (DVT).
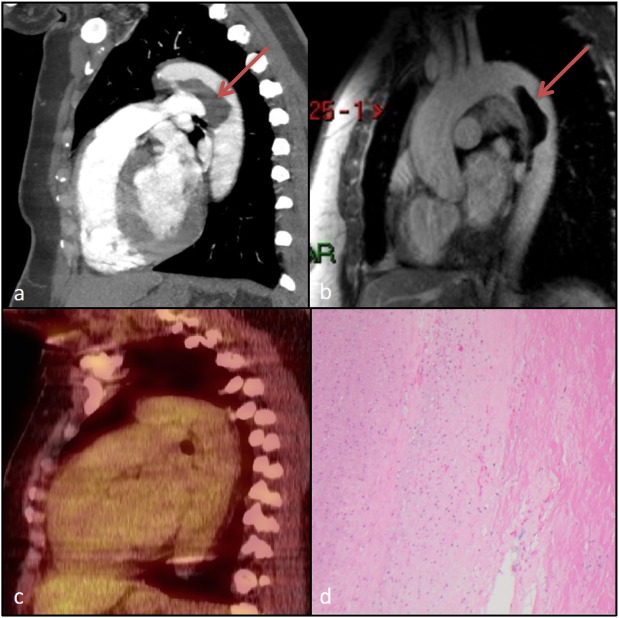
Given the high suspicion of pulmonary embolism a Computed Tomography Pulmonary Angiography (CTPA) was performed, which confirmed multiple bilateral pulmonary emboli. In addition there was an incidental lobulated filling defect measuring 2.2 × 2.1 × 5.6 cm within the descending thoracic aorta arising at the distal aortic arch (See Fig. 1 a). At this stage the differential diagnosis included an intramural thrombus, an angiosarcoma or a thrombosed dissection; the later diagnosis was believed to be less likely so heparin infusion was commenced.
|
|
|
Fig. 1. a) Sagittal section of computed tomography chest showing a feeling defect in the proximal to mid descending thoracic aorta most suggestive of an intramural thrombus. There is no evidence of atherosclerosis or dissection in the descending aorta. b) Sagittal section of MRI chest confirming an intra mural thrombus in the descending aorta. c) PET scan showing no pathological uptake demonstrated in the region of the thrombus within the descending thoracic aorta as correlated with the CT and MRI. d) Sections reveals intraluminal aortic mass is a massive atheroma characterized by a partially endothelialized paucicellular adjacent thickened intima and media with frequent foamy macrophages. There is no inflammation, no dissection and no malignancy. |
A limited thrombophilia screen was negative. On further investigation, peripheral Doppler ultrasound showed no DVT but a near occlusive thrombus in the right popliteal artery and a less significant stenosis in the mid peroneal artery. Further imaging was performed to better characterise the lesion. Magnetic Resonance Imaging was most suggestive of a bland thrombus without an underlying vessel abnormality and Positron Emission Tomography showed no pathological uptake within the lesion or elsewhere suggesting malignancy. See Fig. 1 b,c.
Given the high risk of systemic emboli a decision was made to proceed to surgical removal. An uneventful replacement of the descending aorta while on cardiopulmonary bypass was performed with an 85 mm × 30 mm × 25 mm thrombus removed. She was discharged home on warfarin.
An aortic mural thrombus (AMT) that develops in the absence of atherosclerotic or aneurysmal disease is a rare, incompletely understood, but clinically important diagnosis. It most commonly occurs in young women (mean age 47 years) with a history of smoking1. In most cases described in the limited literature available there was an undiagnosed hypercoagulable state. This highlights the need to exclude malignancy, inflammatory bowel disease, haematologic disorders and exogenous steroid use [1] ; [2] in these patients. In our case there was no active history of smoking, atherosclerosis wasn't seen on imaging, there was no malignancy and the thrombophilia screen was negative. However, several other factors may have contributed.
Firstly, it has been hypothesised that mild atherosclerosis not detected on imaging may be a site for thrombus formation [2]. Additionally, iron deficiency causes a hypercoaguable state [3] and the woman in our case was iron deficient secondary to menorrhagia. Bukharovich et al., [3] described 8 cases of AMT or proximal carotid thrombosis in young females with iron deficiency secondary to menorrhagia without atherosclerosis or an underlying thrombophilia. However the size of the AMT described above was significantly larger. No case reports are available linking norethisterone use to arterial thrombus formation. However norethisterone has been associated with an increased risk of venous thromboembolism in women with menorrhagia [4]. The timing of the symptoms with starting norethisterone also increases the possibility of norethisterone as a cause of the thrombus.
Despite limited research, AMT appears more commonly to occur in the transverse aortic arch but can occur in any section of the aorta between the ascending and abdominal aspects [5]. As a result of the highly pulsatile blood flow in the aorta, with periods of forward, zero and reverse flow, labile thrombi may develop [5]. Typically, as in our case, these occur at the end of the aortic arch on the inside bend of the vessel wall where the velocity of blood flow is lowest, with extension caused by radial growth and axial pulling of the thrombus by the blood flow [5]. Interestingly, in the case described above, the origin of the AMT indeed matched the majority of these found in the data, and was located in the distal end of the aortic arch. However in the contrast with the research, rather than developing in the direction of the flow, the proximal aspect of the AMT remarkably points against the direction of the flow.
Like the knowledge of pathophysiology, treatment regimes are also contentious. Current opinion varies between long-term anticoagulation, surgery, or a combination of both and is often reliant on patient characteristics [2]. A meta-analysis by Fayad et al. noted that up to 25% of patients initially treated with anticoagulation would go-on to require aortic surgery following recurrent peripheral arterial emboli or persistent AMT [2]. It was reported that these patients were also more likely to suffer from recurrent distal arterial emboli, persistent AMT and higher rates of limb amputation as a result of these complications [2]. For these reasons, it was suggested that patients at a higher risk of recurrence be treated with aortic surgery as an aggressive first-line treatment option. In contrast, Turley et al. [1] found that as our knowledge of the aetiology of AMT in relation to hypercoaguable states increased, so too did the indication for a conservative first-line approach. They cited improved success rates with the use of thrombolysis and long-term therapeutic anticoagulation with a significant reduction in the complication rate and length of hospital stay compared with surgical intervention1. In the setting of our patient who presented haemodynamically compromised with a symptomatic AMT complicated by distal arterial emboli, an aggressive treatment plan was carried out. The patient was discharged home on long-term anticoagulation and a follow-up plan to monitor for recurrence.
References
- [1] R.S. Turely, J. Unger, M.W. Cox, et al.; Aortic thrombus: should nonoperative management be first line?; Ann. Vasc. Surg., 28 (7) (2014), pp. 1610–1617
- [2] Z.Y. Fayad, E. Semaan, B. Fahoum, et al.; Aortic mural thrombus in the normal or minimally atherosclerotic aorta; Ann. Vasc. Surg., 3 (2013), pp. 282–290
- [3] I.F. Bukharovich, O. Wever-Pinzon, A. Shah, et al.; Arterial embolism caused by large mobile aortic thrombus in the absence of atherosclerosis associated with iron deficiency anemia; Echocardiography, 29 (2012), pp. 369–372
- [4] A. Sundstrom, H. Seaman, H. Kieler, L. Alfredsson; The risk of venous thromboembolism associated with the use of tranexamic acid and other drugs used to treat menorrhagia: a case–control study using the general practice research database; BJOG, 91-97 (2008)
- [5] D. Stoevesandt, C. Klempt, R. Scheubel, et al.; Embolic events caused by aortic thrombi: an underestimated entity?; J. Thromb. Thrombolysis, 35 (2013), pp. 223–227
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?